TA-65 is a patented telomerase-activating supplement containing cycloastragenol, derived from Astragalus membranaceus root. A 2025 meta-analysis of eight randomized controlled trials involving 750 participants confirmed that TA-65 produces statistically significant telomere elongation (SMD=0.47, p<0.00001), with stronger effects in adults over 60. Based on published human clinical trial data, TA-65 has more RCT evidence than any other commercially available telomerase activator supplement. This guide, reviewed by Dr. Hector Valenzuela, PhD (UCLA doctoral research in telomere biology, multiple peer-reviewed publications), covers everything: the science, the studies, the honest limitations, and whether the TA-65 supplement is worth the investment.
TA-65 Supplement: The Complete Science Guide to Telomerase Activation
Quick Answer: Does TA-65 Work?
- Yes, for telomere maintenance: A 2025 meta-analysis of 8 RCTs (750 participants) found statistically significant telomere elongation (SMD=0.47, p<0.00001)
- Stronger in older adults: Effect size was nearly double in adults over 60 (SMD=0.63 vs 0.36)
- Immune benefits confirmed: 500-person double-blind RCT showed significant reduction in immunosenescent T-cells
- Excellent safety record: No serious adverse events across all 8 trials; 3 negative genotoxicity studies
- Independent validation: 2024 de Jaeger study (zero industry funding) replicated significant telomere lengthening at p=0.004
Does TA-65 work? Yes, for telomere maintenance. A 2025 meta-analysis of 8 randomized controlled trials in 750 participants confirmed statistically significant telomere elongation (SMD=0.47, p<0.00001), with stronger effects in adults over 60. Multiple separate RCTs confirm immune function improvements, and no severe adverse events were recorded across all 8 trials. The 2024 de Jaeger independent study (no industry ties, 6-month RCT, 100% completion rate) replicates the core finding and addresses concerns about industry funding bias directly.
Key Facts at a Glance
- Active compound: Cycloastragenol (patented TA Sciences extraction from Astragalus membranaceus)
- Human RCTs: 8 trials, 750 participants (2025 meta-analysis, PMID: 41286474)
- Telomere effect: Statistically significant elongation (SMD=0.47, p<0.00001); stronger in adults over 60
- Largest immune RCT: n=500 double-blind trial (Raffaele 2021) confirmed significant reduction in immunosenescent CD8+CD28- T cells vs. placebo
- Safety record: No severe adverse events across all 8 RCTs; 3 negative genotoxicity studies; no oncogenic signal over 12 months
- Dual mechanism: Telomerase activation + senolytic clearance (Zhang et al. 2023, PMID: 37047529; animal and cell culture studies only)
- Results timeline: Energy and focus: 4-8 weeks; measurable telomere changes: 6-12 months with objective testing
- Independent validation: de Jaeger 2024 (Nutrients, PMID: 39275278), zero industry funding, 100% participant completion, significant results
Dr. Ed Park was among the first 20 people in the world to swallow a capsule of the TA-65 supplement. What got him there: his father's cancer diagnosis, years spent studying why cells stop working correctly, and a single patented molecule with a mechanism no other supplement had yet addressed. He was a physician, not a hobbyist, and he was betting on the science before most people could spell "telomere."
What he found wasn't a magic pill. It was a specific, patented molecule extracted from Astragalus membranaceus root with precision that decades of research made possible. The TA-65 supplement now has more published human clinical trial data than any other commercially available cycloastragenol supplement, with an RCT evidence base that few dietary supplements in any category can match.
This article covers everything: the corrected science (including what the landmark 2011 Harley study actually found, which differs from how it has been described elsewhere), the 2025 research that strengthens the evidence base, accurate TA-65 dosage guidance, TA-65 price context, an honest look at what the data does and doesn't support, and how this compares to NMN and generic cycloastragenol. It addresses the honest criticism about industry funding directly, because you deserve that context before spending $200 on anything.
Sources and Methodology
This article synthesizes evidence from peer-reviewed journals including Cell Biology and Toxicology, GeroScience, Nutrients, Rejuvenation Research, The Journal of Immunology, and Cell. All cited studies are referenced by PubMed ID (PMID) or DOI where available. The 2025 meta-analysis was conducted using PRISMA guidelines. Effect sizes are reported as standardized mean differences (SMD) with confidence intervals. Studies were assessed for industry funding bias, which is explicitly disclosed throughout. Where evidence is limited to preclinical (animal or cell culture) research, this is clearly stated. Subjective user reports are labeled as anecdotal and distinguished from clinical findings.
Limitations acknowledged: 78% of included trials had industry funding. The independent 2024 de Jaeger study addresses this concern. Functional aging outcomes (frailty, pooled inflammation) did not reach statistical significance in meta-analysis. Long-term safety beyond 12 months has not been studied. Cognitive and bone health claims are based on preclinical research only.
Before you go further: Yes, TA-65 costs more than most supplements. Yes, the supplement industry has a credibility problem. Yes, there are $20 cycloastragenol products on Amazon. And yes, critics correctly note that 78% of TA-65 trials had industry funding. All of that is real. What's also real: the 2024 de Jaeger RCT was fully independent with no industry ties and produced statistically significant telomere lengthening at p=0.004. TA-65 is the only cycloastragenol product backed by an independent meta-analysis, 17 years of safety data, and a third-party COA for every batch. Generic cycloastragenol has zero published human RCTs. This article covers exactly what the evidence shows, including what it doesn't.
What Is the TA-65 Supplement? Understanding Cycloastragenol
TA-65 is a patented dietary supplement containing cycloastragenol, a compound derived from Astragalus membranaceus root that activates telomerase, the enzyme responsible for maintaining the protective caps (telomeres) at the ends of chromosomes. TA Sciences holds the patent on the extraction and standardization process. The clinical evidence base built around that specific process is what separates TA-65 from generic cycloastragenol.
Unlike antioxidants or general "anti-aging" compounds (and calling anything "anti-aging" somewhat misses the point, since the goal is to age well, not to pretend aging isn't happening), TA-65 works through a specific mechanistic pathway: telomerase activation. When telomerase is stimulated, it replenishes the nucleotide sequences at chromosome ends, potentially slowing the shortening that occurs with each cell division.
The Scientific Foundation: Telomere Attrition as a Hallmark of Aging
In 2023, a landmark review in Cell by Lopez-Otin et al. expanded the recognized hallmarks of aging from 9 to 12, explicitly listing telomere attrition as one and stating directly that "aging can be modulated by telomerase activation" ([Lopez-Otin et al., 2023]). When the most-cited aging researchers in the world put that in Cell, the target of TA-65's mechanism gets the scientific establishment's implicit endorsement.
Key Historical Research on TA-65 and Telomere Length
- Harley CB et al. (2011): Published in Rejuvenation Research, this observational study in TA-65-supplementing humans found that mean telomere length was maintained over one year, with a statistically significant reduction in the percentage of critically short telomeres. Critically short telomeres (not average length) are what trigger cellular senescence and dysfunction. ([Harley et al., 2011])
- Fauce SR et al. (2008): Published in The Journal of Immunology, this study demonstrated that cycloastragenol enhanced telomerase activity in antigen-specific human T-cells in ex vivo culture. ([Fauce et al., 2008])
- Dow CT & Harley CB (2016): A pilot study in Clinical Ophthalmology evaluated oral telomerase activator use in early age-related macular degeneration patients, noting preliminary observations warranting further investigation.
How TA-65 Works: Telomerase Activation Plus a Second Mechanism
TA-65 works through two mechanisms: (1) activating telomerase to slow telomere shortening during cell division, and (2) acting as a senolytic agent that selectively clears senescent "zombie cells" that accumulate with age. Most descriptions stop at the first. That's accurate but incomplete.
Mechanism 1: Telomerase Activation
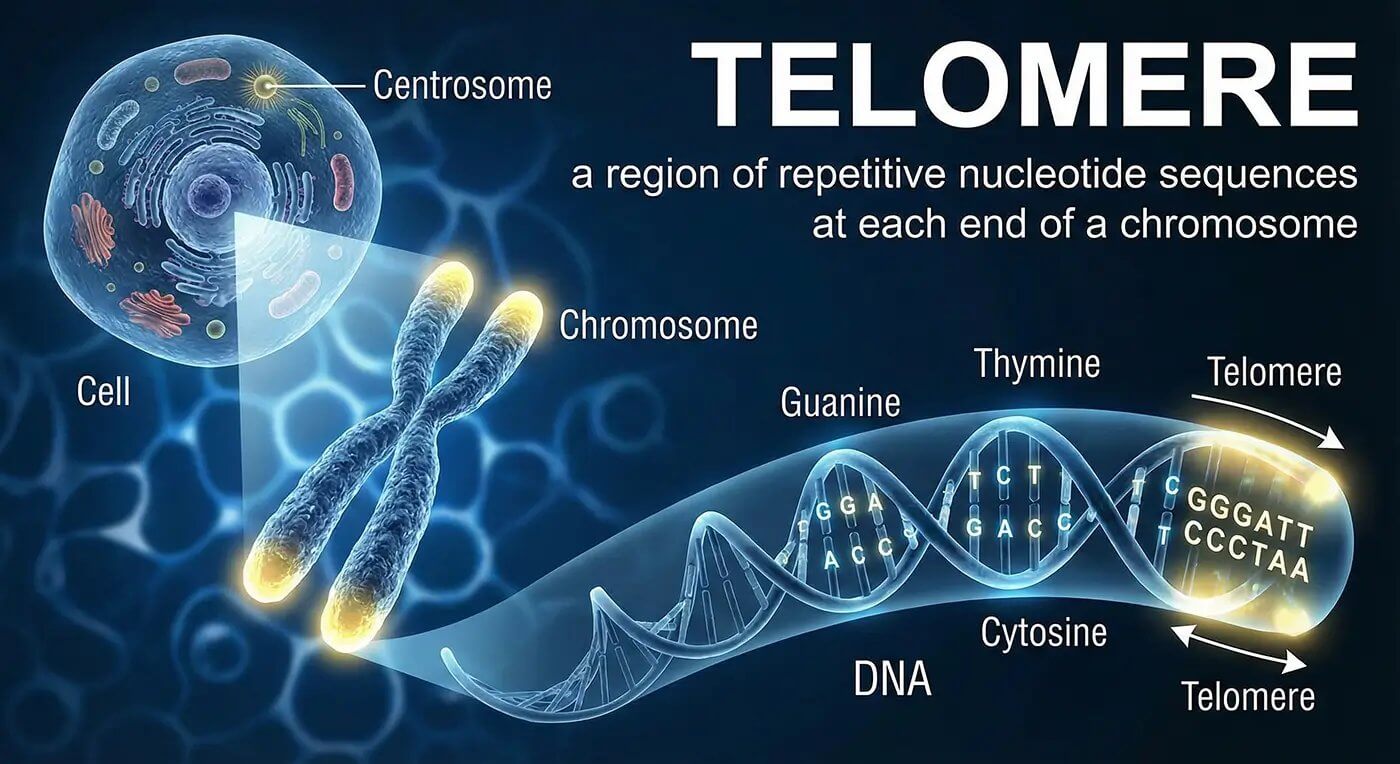
Your chromosomes end in protective caps of repetitive DNA sequences called telomeres (TTAGGG repeated thousands of times). Every time a cell divides, the replication machinery can't copy all the way to the very end of the chromosome. A small section is lost. Telomerase is the enzyme that adds those sequences back.
In most adult somatic cells, telomerase is largely silent. This is partly protective (active telomerase in the wrong context can fuel cancer), but it also means cells have no defense against progressive telomere shortening. Cycloastragenol appears to activate the telomerase gene (hTERT) at concentrations achievable with normal supplementation.
One nuance: the 2025 meta-analysis notes that some researchers debate whether TA-65's effects on immune telomere length reflect direct telomerase activation or a redistribution of immune cell populations toward younger cells with inherently longer telomeres. Both mechanisms could explain the observed data. The practical implication is the same: immune cells with longer telomeres. But the mechanism debate is real and ongoing.
The Hayflick limit describes how normal cells can divide only about 50 times before reaching senescence or death. Critically short telomeres are the trigger for that limit. Reducing the proportion of cells with critically short telomeres, as TA-65 has been shown to do, is the key biological goal. By age 60, the average person has lost roughly 20-40 base pairs of telomere length per year for decades. That's thousands of base pairs gone.
Mechanism 2: Senolytic Clearance of Zombie Cells
Senescent cells are cells that have stopped dividing but refuse to die. They sit in tissues secreting inflammatory signals (the senescence-associated secretory phenotype, or SASP) that damage neighboring healthy cells and accelerate systemic aging. "Zombie cells" is an apt description: dead to useful function, still causing problems.
Zhang et al. (2023, International Journal of Molecular Sciences, [Zhang et al., 2023]) identified cycloastragenol as a novel senolytic agent that selectively induces apoptosis in senescent cells via inhibition of Bcl-2 anti-apoptotic proteins and suppression of the PI3K/AKT/mTOR pathway. It also suppressed SASP, reducing the inflammatory signaling. In a traumatic brain injury-aged (TBI-aged) mouse model, two weeks of CAG treatment cleared senescent cell burden and improved age-related physical dysfunction.
This dual-action profile (telomerase activation plus senolytic clearance) is rare among natural compounds. Note: The senolytic findings are from in vitro cell studies and an animal model only. No human clinical trial has confirmed senolytic activity for cycloastragenol.
The Mitochondria Connection
Users frequently report improved energy as one of the first noticed effects. The mechanism may involve mitochondria. Research on related astragalus compounds shows that astragaloside IV (the precursor to cycloastragenol) enhances mitochondrial biogenesis and ATP production. Cycloastragenol's reduction of cellular senescence burden also indirectly supports mitochondrial function, since senescent cells are metabolically dysfunctional. The energy reports from users, common in weeks 4-8, may reflect this cellular cleanup rather than any direct stimulant effect. No published human trial has isolated the specific energy mechanism for TA-65.
Why You Can't Just Eat Raw Astragalus
Cycloastragenol is present in Astragalus membranaceus root at approximately 0.001 to 0.01 percent of dry weight. To get a meaningful supplemental dose, you'd need to consume quantities of raw root that would be physically impossible. And even if you committed to eating your way through an astragalus farm every morning (botanically ambitious and inadvisable), standard processing methods used in generic extracts fail to isolate and standardize the specific molecule. TA-65's patented extraction process concentrates and standardizes cycloastragenol specifically. Raw astragalus is not equivalent.
TA-65 Clinical Research: What 8 Human Trials Actually Show
The evidence base for TA-65 has grown substantially since the early Harley studies. A 2025 systematic review synthesized the entire body of human clinical trial data. Below is what it found, along with the individual trials that matter most.
The 2025 Meta-Analysis: 8 RCTs, 750 Participants
Su et al. (Cell Biology and Toxicology, November 2025, [Su et al., 2025]) conducted a PRISMA-guided meta-analysis of 8 randomized controlled trials with 750 participants, mean age 63.3 years. This is the largest synthesis of TA-65 clinical data ever published.
Key finding on telomeres: TA-65 induced statistically significant telomere elongation across all 8 trials (SMD = 0.47, 95% CI: 0.31-0.62, p<0.00001). The effect was stronger in adults over 60 (SMD = 0.63 versus 0.36 in younger adults, p=0.03). That's a moderate, robust, repeatedly confirmed effect.
The industry funding issue: 78% of included trials had industry funding. This is a legitimate criticism. The meta-analysis authors themselves flag it. The correct response is not to dismiss it but to note that the 2024 de Jaeger study (fully independent, no industry ties) replicates the significant telomere lengthening result. One independent RCT doesn't prove everything. But it substantially weakens the "all results are industry-influenced" argument.
Important nuance: The meta-analysis found that telomere elongation did not translate to statistically significant improvements in pooled frailty scores (p=0.15) or inflammatory markers across all studies (p=0.07). Individual trials show stronger inflammatory effects (see TACTIC below), but the pooled data requires honesty: changing a biomarker is not the same as changing a clinical outcome. The telomere data is solid. Functional aging claims need more evidence.
Safety: Across 8 RCTs and 750 participants, no severe adverse events were recorded. Mild GI effects (nausea 7.1%, abdominal discomfort 5.3%) were the most commonly reported issues. No oncogenic signal was detected over 12 months.
The 2024 de Jaeger RCT: Independent Science, Clean Data
de Jaeger et al. (Nutrients, September 2024, [de Jaeger et al., 2024]) ran a 6-month randomized, double-blind, placebo-controlled trial in 40 healthy adults with a mean age of 56. No industry funding. Every single participant completed the study. The astragalus-based supplement significantly lengthened median telomere length (p=0.01) and short telomere length (p=0.004). The placebo group showed no change. Zero adverse effects were reported.
Clean design, independent researchers, statistically significant on both median and short telomere length, zero safety signals. As supplement research goes, this is about as rigorous as it gets.
The Raffaele 2021 RCT: 500 People, Double-Blind, 9 Months
Raffaele et al. (OBM Geriatrics, 2021; [Raffaele et al., 2021]) ran the largest TA-65 trial ever conducted: a double-blind, placebo-controlled, multi-arm parallel RCT with 500 healthy subjects over 9 months. The question was whether TA-65 could move the needle on immune aging specifically.
It did. All TA-65 doses (100, 250, and 500 units daily) significantly decreased immunosenescent CD8+CD28- T cells compared to placebo. This is a key biomarker of immune aging: as these cells accumulate, the immune system becomes less flexible. In the placebo group, CD8+CD28- T cells actually increased by 4.38 cells per microliter over 9 months. In the 100-unit and 250-unit TA-65 groups, they decreased by approximately 28 cells per microliter. No serious adverse events were attributed to TA-65.
The TACTIC Trial: TA-65 in Heart Attack Patients
Bawamia et al. (GeroScience, August 2023, [Bawamia et al., 2023]) published the TACTIC trial, a randomized, double-blind, placebo-controlled Phase IIa pilot in 90 MI patients aged 65 and older. Participants received TA-65 (16mg daily, equivalent to two standard capsules) or placebo for 12 months following a myocardial infarction.
The primary endpoint (proportion of CD8+ TEMRA lymphocytes) did not differ significantly between groups. The secondary outcomes were striking. Total lymphocyte count increased by 285 cells per microliter in the TA-65 group (p<0.01), driven by increases in every major immune cell type. High-sensitivity CRP was 62 percent lower in the TA-65 group at 12 months (1.1 vs. 2.9 mg/L). TA-65 patients also experienced significantly fewer post-MI complications.
One trial doesn't prove a cardiovascular benefit, and the primary endpoint was not met. The honest characterization: TA-65 may support cardiovascular recovery and immune function in older MI patients. That framing is defensible. Claiming a proven cardiovascular benefit is not.
The Research Is Real. The Source Matters.
Across 8 randomized controlled trials, no other commercially available cycloastragenol product comes close to this evidence base. That data was built around the patented TA-65 molecule and extraction process.
Get the Supplement Behind 8 Human Clinical Trials at RevGenetics
Reviewed by Dr. Hector Valenzuela, PhD (UCLA Telomere Research alumnus). Third-party tested. 30-day price guarantee.
The Harley 2011 Study: What It Actually Found
The landmark 2011 Rejuvenation Research paper by Harley et al. ([Harley et al., 2011]) has been frequently mischaracterized as showing "significant increases in telomere length." That's not accurate. What the study actually found was maintenance of mean telomere length combined with a statistically significant reduction in the percentage of critically short telomeres. The distinction is biologically meaningful. Cells don't enter senescence because the average telomere is short. They enter senescence because at least one critically short telomere triggers the cellular alarm.
Ready to try TA-65? RevGenetics carries pharmaceutical-grade TA-65 supplements, independently verified and trusted since 2007.
Shop TA-65 Supplements →TA-65 Benefits: What the Science Actually Supports
1. Telomere Maintenance and Elongation
Human clinical studies, including the 2025 meta-analysis of 8 randomized controlled trials, confirm that TA-65 supplementation produces statistically significant telomere elongation in immune cells (SMD=0.47, p<0.00001). The effect is stronger in adults over 60. If you're in that age group and telomere maintenance is your goal, the data is as strong as supplement science gets.
2. Immune Function and Immune Cell Rejuvenation
T-cells and natural killer cells replicate constantly, which means their telomeres shorten faster than almost any other cell type in the body. The Raffaele 2021 trial (n=500, double-blind) demonstrated a significant decrease in immunosenescent CD8+CD28- T cells versus a placebo group that trended in the wrong direction. Two or more independent RCTs now support immune function claims for TA-65. These are human clinical findings, not extrapolations from cell studies.
3. Cardiovascular Support
The TACTIC trial produced striking secondary-endpoint findings in immune and inflammatory markers in post-MI patients. The pooled meta-analysis cardiovascular data was not statistically significant (p=0.07). That tension needs to be acknowledged honestly. TA-65 may support cardiovascular health, and the TACTIC secondary results are encouraging, but a single trial in a specific patient population doesn't establish a broad cardiovascular benefit.
4. Cognitive Function and Brain Health (Preclinical Only)
No human randomized controlled trial has tested TA-65 specifically for cognitive function. Preclinical research is emerging: Weng et al. (Phytomedicine, 2025, [Weng et al., 2025]) found that cycloastragenol enhanced microglial phagocytosis, reduced amyloid beta deposition, and improved cognition in 5xFAD Alzheimer's model mice. These findings are preclinical only and cannot be extrapolated to humans.
5. Bone Health Support (In Vitro Research)
Wu et al. (Phytotherapy Research, 2021, [Wu et al., 2021]) found that cycloastragenol protected osteoblasts from glucocorticoid-induced damage by activating telomerase. This is cell-level research only. No human clinical trial has confirmed bone health benefits.
6. Skin Health and Cellular Repair
Skin gets its own section because the data is genuinely interesting, even with appropriate caveats. See below.
TA-65 and Skin Health: What the Cycloastragenol Research Shows
Two published studies give skin health claims real scientific grounding, though both require context about what has and hasn't been tested in humans with oral TA-65 specifically.
Yang et al. 2023: Collagen, Hyaluronic Acid, and UV Protection
Yang et al. (Journal of Dermatological Science, July 2023, [Yang et al., 2023]) studied cycloastragenol in human dermal fibroblasts and HaCaT keratinocytes (the two main skin cell types). CAG significantly reduced UVB-induced matrix metalloproteinases (MMP-1, MMP-9, MMP-13), which are the enzymes that degrade collagen after sun exposure. CAG also increased Collagen I levels, enhanced hyaluronic acid, and upregulated skin hydration factors including filaggrin.
This is an in vitro study in human cells, not a clinical trial. It establishes strong mechanistic evidence for cycloastragenol's role in protecting skin collagen at the cellular level.
Weiss & Weiss 2014: Human Clinical Study with CAG-Containing Topical
Weiss RA et al. (Journal of Drugs in Dermatology, September 2014, [Weiss et al., 2014]) ran a 12-week open-label study in 20 subjects using a topical formulation containing cycloastragenol along with growth factors, peptides, and antioxidants. Significant improvement appeared across all 8 measured skin quality categories by week 12.
The formulation was multi-ingredient. Cycloastragenol can't be isolated as the sole active component from this study. No human RCT has tested oral TA-65 specifically for skin outcomes. Some users report improvements in skin texture and elasticity with consistent oral use. The cellular and topical human data support biological plausibility. The oral clinical connection hasn't yet been established.
Cellular Protection From the Inside Out
While most skin supplements address surface-level effects, TA-65 targets the cellular mechanisms that govern how skin cells age and replicate. Two published studies involving human skin cells support this angle.
TA-65 and Cancer: What Five Safety Studies Show
The cancer question is the most common concern about any telomerase activator or cycloastragenol supplement. Cancer cells hijack telomerase for unlimited, aberrant replication. Does activating telomerase in healthy cells risk the same thing?
What the Research Actually Shows
de Jesus et al. (2011, Aging Cell, co-authored by Harley CB and Blasco MA; [de Jesus et al., 2011]) studied TA-65 in aging mice and found that it elongated telomeres, improved glucose tolerance, and enhanced healthspan without increasing cancer incidence. That directly addressed the theoretical concern.
The genotoxicity record is equally reassuring. Three separate genotoxicity studies (bacterial reverse mutation assay, chromosomal aberration assay, erythrocyte micronucleus assay) all returned negative results. A carcinogenicity study in xenografted mice found no adverse effects on survival or tumor growth over four months.
The 2025 meta-analysis of 8 human RCTs found no oncogenic signal over 12 months of supplementation in 750 participants. That's 12 months of actual human data, not theoretical safety modeling.
The Honest Caveat
Long-term carcinogenic potential beyond 12 months hasn't been addressed in published studies. The safety record at standard supplemental doses for durations studied is excellent. What happens over decades of continuous use is genuinely unknown. Anyone with cancer history must speak with their oncologist.
TA-65 Side Effects: A Complete Safety Review
TA-65 has a genuinely good safety profile, backed by human clinical data across multiple trials. Here's what you need to know about TA-65 side effects before starting.
What the Clinical Trials Found
Across 8 RCTs involving 750 participants, no serious adverse events were recorded. The most commonly reported TA-65 side effects were mild gastrointestinal issues:
- Nausea: 7.1% of participants in clinical trials
- Abdominal discomfort: 5.3% of participants
- Mild digestive upset: Occasional reports, typically resolving when taken with food
Most users tolerate the supplement well when taken with a meal. Taking TA-65 on an empty stomach increases the likelihood of GI discomfort.
Safety Testing Record
- Three negative genotoxicity studies: Bacterial reverse mutation, chromosomal aberration, and erythrocyte micronucleus assays all returned negative
- No oncogenic signal: Detected over 12 months in any published trial
- NOEL established: No-observed-effect level of 150mg/kg body weight, providing a substantial safety margin above any normal supplemental dose
Who Should Not Take TA-65
Drug Interactions
No significant drug interactions have been documented in the published literature for TA-65 at standard supplemental doses. If you take prescription medications, consult your healthcare provider before starting any new supplement.
Looking for the real TA-65? RevGenetics is one of the few authorized TA-65 resellers. Get the authentic supplement backed by real telomere science.
View TA-65 Products →TA-65 Dosage: How Much Should You Take?
The standard TA-65 dosage is one 250-unit capsule (approximately 8mg purified cycloastragenol) taken daily with a fat-containing meal. Here's the complete breakdown of dosing options and what the clinical trials used.
The Two Main Products and Their Doses
| Product | Dose Per Capsule | Recommended Daily Use | Monthly Cost | Best For |
|---|---|---|---|---|
| TA-65 Standard (250-unit) | ~8mg purified cycloastragenol | 1 capsule daily | $200-$250 (~$6.67-8.33/day) | Adults 50+; serious longevity protocols |
| TA-65 Bioenhanced (100-unit) | Lower concentration, same molecule | 1 capsule daily | ~$100 (~$3.33/day) | First-timers; maintenance phase; budget-conscious |
Clinical trial dosing: The TACTIC trial used 16mg daily (equivalent to two standard capsules). The Raffaele 2021 500-person trial tested 100, 250, and 500 units, with all three doses showing significant immune effects. The Salvador 2016 RCT found that the 250-unit group gained an average 530 base pairs of telomere length over 12 months while the placebo group lost 290 base pairs.
Timing and Administration
Take TA-65 once daily with breakfast or your first substantial meal. Include healthy fats in that meal for best absorption, as cycloastragenol is a triterpenoid saponin and fat may support bioavailability. Morning dosing aligns with natural cellular repair rhythms.
Cycling Protocols
Clinical trials have used continuous daily dosing without cycling. Some biohackers use a 3-month-on, 1-month-off approach, but no published data specifically supports cycling over continuous use for TA-65. Consistent daily use over 6-12 months is more important than any specific cycling protocol.
TA-65 Before and After: What to Realistically Expect
If you're wondering about TA-65 before and after results, here's an honest timeline of what users typically experience and what the science can actually measure.
Subjective Changes: What You Might Feel
- Weeks 4-6: Many users report first noticeable improvements in energy and sleep quality
- Months 2-3: Workout recovery and immune resilience improvements commonly noted
- Months 5-6: Some users report improvements in skin texture and hair quality
These are self-reported experiences, not clinical guarantees. Individual results vary significantly. The lack of an immediate "feeling" doesn't mean nothing is happening at the cellular level.
Objective Changes: What Testing Can Measure
Measurable telomere length changes require 6-12 months of consistent use and objective testing. The de Jaeger 2024 RCT found significant telomere lengthening at 6 months under controlled conditions. For individual (non-controlled-trial) tracking, 12 months is a more reliable horizon.
Before starting: Get a baseline telomere test. This is your comparison point for everything that follows. No baseline, no data. Services like LifeLength (FISH-based, gold standard for precision) or TruDiagnostic (includes biological age) offer at-home collection.
After 12 months: Retest with the same service and same methodology. Normal age-related shortening runs approximately 20-40 base pairs per year. An increase of 0.2-0.5 kb, or even maintenance of length against expected decline, is a positive signal.
Setting Realistic Expectations
TA-65 isn't a fountain of youth. Nobody is going to confuse you with your college graduation photo after a year of supplementation. What the data supports: statistically significant telomere elongation, meaningful immune cell improvements, and a good safety profile. What the data doesn't support: dramatic visible anti-aging, disease reversal, or guaranteed functional improvements. The pooled data on frailty and systemic inflammation didn't reach statistical significance. Set your expectations at "subtle cellular support" rather than "obvious transformation."
Is TA-65 a Scam? An Honest Assessment
This is a high-search-volume question, so let's address it directly: Is TA-65 a scam?
The short answer: No. TA-65 has more published human clinical trial data than any competing telomerase activator supplement. The mechanism is real. The data is real. But the skepticism is understandable, and some of it is warranted.
Why People Ask This Question
- The price: $200-$250 per month is expensive for a supplement. That's a legitimate concern.
- Industry funding: 78% of TA-65 trials had industry funding. Critics correctly flag this as a potential source of bias.
- Supplement industry reputation: The broader supplement industry has credibility problems. Guilty by association is unfair but understandable.
- Overpromising competitors: Other "anti-aging" products make outrageous claims. TA-65 gets lumped in unfairly.
What the Evidence Actually Shows
- Real mechanism: Cycloastragenol activates telomerase. This is confirmed in laboratory and human studies.
- Real human data: 8 RCTs, 750 participants, meta-analysis in a peer-reviewed journal
- Independent validation: The 2024 de Jaeger study had zero industry funding and replicated significant telomere lengthening (p=0.004)
- Safety record: 17 years on the market, no serious adverse events in published trials
What TA-65 Is NOT
- A cure for aging
- A treatment for any disease
- A substitute for exercise, sleep, or good nutrition
- A guaranteed path to looking or feeling younger
The Honest Bottom Line
TA-65 isn't a scam. It's a real compound with a real mechanism and real human data. It's also not a miracle. The effects are moderate and primarily cellular. The price is high. Whether it's worth it depends on your goals, your budget, and how much you value the evidence base compared to cheaper alternatives with no published human research.
If you want the best-studied telomerase activator on the market with independent validation, TA-65 is it. If you want dramatic visible anti-aging on a budget, you're going to be disappointed by any supplement, including this one.
TA-65 vs NMN: Which Is Better?
This is one of the most common questions in the longevity supplement space. Here's the direct comparison.
Different Mechanisms, Different Targets
TA-65 directly addresses telomere maintenance through telomerase activation. It has 8 human RCTs supporting this specific claim. It also has emerging evidence as a senolytic agent (animal and cell studies only).
NMN (Nicotinamide Mononucleotide) boosts NAD+, a coenzyme critical for mitochondrial energy production, DNA repair, and sirtuin activation. NMN doesn't directly act on telomere length. Its human evidence base is growing but generally shorter in duration.
The Comparison Table
| Feature | TA-65 | NMN |
|---|---|---|
| Primary Mechanism | Telomerase activation + senolytic | NAD+ precursor, mitochondrial function |
| Human RCTs | 8 RCTs, n=750 (meta-analysis confirmed) | 7+ RCTs as of 2025, generally shorter duration |
| Telomere Effects | Directly addresses telomere length | No direct telomere mechanism |
| Cost Per Day | $3.33-$8.33 | $1.00-$3.00 |
The Real Answer: They're Complementary
If you're choosing only one: TA-65 has a deeper and more specific evidence base on the one outcome it claims (telomere maintenance), with 8 RCTs and a 500-person immune trial.
For most people serious about longevity, the question isn't "which one" but "can I do both." The combination makes biological sense. They work on different pathways with no known adverse interactions. Many longevity researchers use both, targeting different aspects of cellular aging simultaneously.
TA-65 from RevGenetics, the same telomerase activator studied in clinical trials. Third-party tested, no fillers.
Check Out TA-65 →TA-65 for Women: Hormones, Menopause, and Telomere Health
No TA-65 trial has specifically enrolled perimenopausal or postmenopausal women. But the science of hormones and telomeres makes this population particularly worth discussing.
Why Menopause Matters for Telomeres
Estrogen has a protective effect on telomere length. Research shows that higher estrogen levels correlate with longer telomeres in premenopausal women. The hormonal decline during perimenopause and menopause accelerates several aging pathways, and telomere attrition is among them.
Women going through menopause often report accelerated aging symptoms: changes in skin elasticity, energy levels, cognitive function, and immune resilience. While these are multifactorial, the telomere connection is biologically plausible.
What the Evidence Supports (and Doesn't)
The general RCT evidence on telomere maintenance applies to women as it does to men. The 2025 meta-analysis included both sexes. The mechanistic case for telomere support during hormonal transitions is reasonable.
What we don't have: Population-specific evidence for perimenopausal or postmenopausal women. No trial has specifically studied whether TA-65's effects differ during hormonal transition.
Practical Considerations
- Women with hormone-sensitive conditions (breast cancer history, fibroids, endometriosis) should consult their physician before starting TA-65
- The general safety profile applies equally to women, with the same contraindications
- Some women report skin and energy improvements that they attribute to TA-65, though these are anecdotal
If you're a woman in perimenopause or menopause considering TA-65, the evidence base is the same as for everyone else: strong on telomere maintenance, strong on immune function, unclear on population-specific effects during hormonal transition.
How to Track Your TA-65 Progress: Telomere Testing Guide
TA-65 is one of the rare supplements where you can actually measure whether it's working. Telomere length testing gives you objective "TA-65 before and after" data.
What Telomere Testing Measures
Most commercial tests measure leukocyte telomere length (LTL), the average telomere length in white blood cells. Some measure the percentage of critically short telomeres separately, which is arguably more important.
The two main testing methodologies:
- qPCR: Measures the ratio of telomere repeat copies to single-copy gene copies. Higher variability, better for population comparisons than individual tracking.
- FISH-based methods: Directly visualizes and measures telomere length. Gold standard for precision, especially for tracking small changes over time.
Current Telomere Testing Services
- LifeLength (Madrid, Spain): FISH-based measurement. Measures both mean telomere length and percentage of short telomeres below 3 kb. Widely considered the most scientifically rigorous option. Cost: $300-$500. Now offers at-home blood collection for US customers.
- TruDiagnostic (US-based): TruAge COMPLETE includes telomere length alongside epigenetic biological age testing. Popular in the US longevity community. $250-$350.
- Repeat Diagnostics (Vancouver, Canada): Flow-FISH based testing widely used in clinical research settings.
The Testing Protocol
- Baseline first: Get your baseline before starting TA-65. Not after two months in. Before you start.
- Wait 12 months: Telomere changes occur slowly. Six months is often not enough for individual tracking.
- Same service, same methodology: Comparing results from different labs or different methodologies is meaningless.
Start your baseline test today. LifeLength (FISH-based, gold standard) and TruDiagnostic (biological age + telomeres) both offer at-home collection. Order before your first capsule.
Adults Over 60 Get the Strongest Results: Who Else Benefits from TA-65?
The 2025 meta-analysis found that adults over 60 showed stronger telomere elongation effects (SMD=0.63 versus 0.36 in younger adults). But the data isn't limited to seniors.
Adults over 50: This is the core demographic. Telomere shortening accelerates with age, and the meta-analysis effect size is strongest here.
High-stress professionals and caregivers: The seminal 2004 Epel et al. study (PNAS) showed that women with the highest perceived stress had telomeres equivalent to women 10 years older. Chronic stress is one of the best-documented accelerators of telomere shortening.
Post-MI and cardiovascular patients: The TACTIC trial specifically enrolled MI patients aged 65 and older. The secondary-endpoint immune recovery data is the most specific human evidence for this group.
Serious biohackers tracking biological age: People using telomere testing services are well-positioned for TA-65. This is a supplement where objective measurement is possible.
Athletes with high training loads: Very high-volume training increases immune cell turnover and may be associated with accelerated telomere shortening. Anecdotal reports of improved recovery are common among athlete users.
TA-65 Reviews: What Long-Term Users Actually Report
The following TA-65 reviews represent self-reported user experiences. Individual results vary and these accounts don't constitute clinical evidence.
Dr. Ed Park: An Early Adopter's Perspective
Dr. Ed Park reports being among the first 20 people to take TA-65 in 2007. His journey into telomere science began with his father's cancer diagnosis, which led him to study aging mechanisms professionally. After five years on TA-65, he described his results as "remarkable" and built a practice around telomerase activation therapy.
Dr. Bill Andrews: Patient #1
When TA-65 launched in 2007, it cost $25,000 a year. Dr. Bill Andrews was Patient #1. He paid it. Andrews, the Sierra Sciences CEO who co-discovered the human telomerase gene, wasn't someone who stumbled onto the product through a late-night advertisement. He understood exactly what the compound was supposed to do, and when it became available, he was first in line regardless of the price. When the man who co-discovered the telomerase gene is Patient #1, that says something about how serious scientists view this compound.
Timeline of Commonly Reported Experiences
These patterns come from user self-reports and don't represent guaranteed outcomes:
- Weeks 4-6: Many users report first subjective energy and sleep improvements
- Months 2-3: Workout recovery and immune resilience improvements commonly noted
- Months 5-6: Skin texture and vitality improvements reported by some users
- Months 9-12: Measurable telomere length changes may be detectable on objective testing
TA-65 Price Guide: Costs, Savings and Value
TA-65 is priced at $200-$250 per bottle for the standard formula, working out to approximately $6.67-$8.33 per day. The Bioenhanced formula runs about $100 per month, or $3.33 per day.
For context: a single telomere length test from LifeLength runs $300-$600. A specialist longevity consultation can cost $400 or more. TA-65 at $6.67 per day is often the cheapest component of a serious longevity tracking protocol, not the most expensive one. (Though the telomere test is probably less awkward to explain on your credit card statement.)
Ways to Save
- Subscription / Auto-Delivery: 10% off every order
- Newsletter: 15-25% savings via exclusive deals. Subscribe at eepurl.com/epAfQ
- FSA/HSA: May be eligible for reimbursement. Check with your plan administrator.
- Bulk purchasing: Volume pricing available for multi-bottle orders
- Seasonal promotions: Up to 20% off during holiday events
Start Your Telomere Health Protocol
The research behind TA-65's benefits is cumulative, and so are the benefits themselves. Every month you delay is a month of compounding you don't get back.
Get cGMP-Manufactured TA-65 at RevGenetics
100% satisfaction guarantee. Third-party tested. Free shipping on qualifying orders. 30-day price guarantee.
Ready to try TA-65? RevGenetics carries pharmaceutical-grade TA-65 supplements, independently verified and trusted since 2007.
Shop TA-65 Supplements →Why Trust RevGenetics for TA-65?
RevGenetics has been in the longevity supplement market since 2007. That's 17+ years, which is ancient by supplement company standards. They source TA-65 directly from TA Sciences (the patent holder), so you get the same formulation used in the clinical trials, not a lookalike.
Quality Credentials
- Third-party tested, Certificate of Analysis available for every batch
- cGMP manufactured in an FDA-registered US facility
- Scientific review by Dr. Hector Valenzuela, PhD (UCLA Telomere Research alumnus)
- Direct sourcing relationship with TA Sciences (the patent holder)
- 100% satisfaction guarantee and 30-day price guarantee
- Free shipping on qualifying orders
The 8 human RCTs were built around the patented TA-65 formulation. If that research is why you're buying, make sure you're getting the product the research was actually conducted with.
Building a Complete Longevity Stack: Spermidine and TA-65
For those building a longevity protocol, spermidine and TA-65 represent two of the best-studied natural interventions that target distinct aging mechanisms. They work differently, and that's exactly why they complement each other.
What Spermidine Does
Spermidine is a naturally occurring polyamine found in natto (the fermented soybean paste that, if you've ever actually opened a package, goes a long way toward explaining why longevity supplements were invented), aged cheese, wheat germ, and mushrooms. It activates autophagy, the cellular cleanup process that removes damaged proteins and dysfunctional organelles. Spermidine levels fall with age.
What TA-65 Does That Spermidine Doesn't
TA-65 targets chromosomal telomere maintenance through telomerase activation, plus senolytic clearance of senescent cells. These are mechanisms spermidine doesn't address. The combination covers cellular cleanup (spermidine's autophagy) and chromosomal stability (TA-65's telomerase), two distinct fronts in the aging process.
Exercise as the Third Pillar
A 2025 meta-analysis published in Frontiers in Physiology ([Exercise Meta-analysis, 2025]) confirmed that exercise interventions increase both telomere length and telomerase activity in humans. TA-65 and regular exercise are synergistic, not competing interventions.
Practical Stack Protocol
- Morning: TA-65 (1 capsule with breakfast)
- Morning or evening: Spermidine (25mg with a meal)
- Optional additions: NMN/NR and resveratrol can be spaced several hours from TA-65
- Daily: Consistent moderate-to-vigorous exercise
Build the Complete Longevity Stack
TA-65 addresses telomere maintenance. Rapymine (spermidine) addresses cellular autophagy. Together they cover two distinct and complementary mechanisms of cellular aging.
Third-party tested. cGMP manufactured. Reviewed by Dr. Hector Valenzuela, PhD.
TA-65 Supplement: Frequently Asked Questions
What does the TA-65 supplement do?
TA-65 is a dietary supplement derived from Astragalus membranaceus that contains cycloastragenol, a compound studied for its ability to activate telomerase, the enzyme that maintains telomere length during cell division. A 2025 meta-analysis of 8 randomized controlled trials confirmed statistically significant telomere elongation in human subjects (SMD=0.47, p<0.00001). Research also confirms meaningful immune cell improvements in multiple human trials, including a 500-person double-blind RCT. TA-65 is not a drug and is not intended to treat any disease.
Does TA-65 actually work?
Based on the available human clinical evidence, yes, for its intended purpose of telomere maintenance and immune cell support. A 2025 PRISMA-guided meta-analysis of 8 randomized controlled trials (750 participants) found statistically significant telomere elongation with a moderate effect size. The 2024 de Jaeger independent RCT (no industry funding) replicated significant telomere lengthening. Immune function benefits are confirmed in multiple RCTs including a 500-person trial. Cardiovascular and functional aging claims require more evidence.
How long does it take to see results from TA-65?
Users often report subjective improvements in energy and focus within 4-8 weeks of consistent use. Measurable changes in telomere length require 6-12 months minimum and objective testing via services like LifeLength, TruDiagnostic, or Repeat Diagnostics. Individual results vary significantly.
Is TA-65 the same as cycloastragenol? How is it different from cheap Amazon products?
TA-65 contains cycloastragenol as its primary active compound, but it is not interchangeable with generic products. TA-65 uses a patented extraction and purification process developed by TA Sciences to deliver a standardized, verified dose. Generic cycloastragenol products on Amazon have variable purity, no standardization, and zero published human clinical trial data. The 8 human RCTs that form TA-65's evidence base were conducted specifically with the patented TA-65 formulation.
Is TA-65 safe for long-term use?
TA-65 has an excellent safety profile backed by human clinical data. Across 8 randomized controlled trials involving 750 participants, no severe adverse events were reported. Three independent genotoxicity studies returned negative results. No oncogenic signal was detected in any published trial over 12 months. Mild GI effects (nausea, abdominal discomfort) were the most commonly reported adverse events. Long-term safety beyond 12 months hasn't been established in published studies.
Is TA-65 a scam?
No. TA-65 has more published human clinical trial data than any competing telomerase activator supplement. The 2025 meta-analysis of 8 RCTs confirms statistically significant telomere elongation. The compound has a real molecular mechanism confirmed in both laboratory and human studies. Critics correctly note that 78% of trials had industry funding, but the 2024 de Jaeger independent study (no industry ties) replicates the core finding.
What are TA-65 side effects?
TA-65 has an excellent safety profile. Across 8 RCTs involving 750 participants, no serious adverse events were recorded. Mild GI effects (nausea 7.1%, abdominal discomfort 5.3%) were the most commonly reported issues. Most users tolerate the supplement well when taken with food. Three independent genotoxicity studies returned negative results.
What is the recommended TA-65 dosage?
The standard TA-65 dosage is one 250-unit capsule (approximately 8mg purified cycloastragenol) taken daily with a fat-containing meal, preferably breakfast. The TACTIC trial used 16mg daily (two capsules). The Bioenhanced 100-unit version is available for first-timers or maintenance dosing at a lower cost.
How does TA-65 compare to NMN? Which is better?
TA-65 and NMN target different aspects of cellular aging and are more complementary than competing. TA-65 directly addresses telomere maintenance through telomerase activation, with 8 human RCTs behind it. NMN boosts NAD+, supporting mitochondrial energy and DNA repair enzymes. If forced to choose one, TA-65 has a deeper evidence base for telomere-focused aging support. Most serious longevity researchers use both.
Can women in perimenopause or menopause benefit from TA-65?
No TA-65 trial has specifically enrolled perimenopausal or postmenopausal women. However, estrogen has a protective effect on telomere length, and hormonal decline during menopause accelerates telomere attrition. The mechanistic case for telomere support during this transition is reasonable. The general RCT evidence applies to women as it does to men.
What is the TA-65 price?
TA-65 standard formula costs $200-$250 per bottle (30 capsules), working out to approximately $6.67-$8.33 per day. The Bioenhanced formula costs about $100 per month ($3.33/day). Subscription orders receive 10% off. Newsletter subscribers get 15-25% off through exclusive deals.
Where can I buy authentic TA-65?
RevGenetics.com is the primary recommended source for authentic, third-party tested TA-65. Purchasing directly ensures access to the satisfaction guarantee, subscription discounts, and price guarantee. TA-65 is not sold at GNC, Whole Foods, or Costco. Be cautious of Amazon listings that may not carry the authentic TA Sciences-licensed formulation.
17 Years of Human Clinical Evidence: The TA-65 Research Timeline
| Year | Milestone |
|---|---|
| 2007 | TA-65 becomes commercially available; Dr. Bill Andrews among first patients at $25,000/year. RevGenetics founded. |
| 2008 | Fauce et al. (J Immunol): cycloastragenol enhances telomerase in human T-cells ex vivo ([Fauce et al., 2008]) |
| 2011 | Harley et al. (Rejuvenation Research): telomere maintenance and reduced critically-short telomeres ([Harley et al., 2011]); de Jesus et al. (Aging Cell): no increased cancer incidence in mice ([de Jesus et al., 2011]) |
| 2016 | Salvador et al. RCT: significant telomere lengthening in TA-65 250U group vs. placebo (PMC5178008) |
| 2021 | Raffaele et al.: double-blind RCT in 500 subjects; significant reduction in immunosenescent T cells ([Raffaele et al., 2021]) |
| 2023 | TACTIC Trial: 62% CRP reduction in post-MI patients ([Bawamia et al., 2023]); Zhang et al.: CAG identified as senolytic ([Zhang et al., 2023]); Lopez-Otin (Cell): telomerase activation confirmed as aging modulator ([Lopez-Otin et al., 2023]) |
| 2024 | de Jaeger et al. (Nutrients): independent RCT (no industry funding), significant telomere lengthening at p=0.004 ([de Jaeger et al., 2024]) |
| 2025 | Su et al. meta-analysis: 8 RCTs, 750 participants, SMD=0.47 telomere elongation (p<0.00001) ([Su et al., 2025]); Exercise meta-analysis confirms exercise increases telomere length and telomerase ([Exercise Meta-analysis, 2025]) |
The TA-65 Supplement: Bottom Line
After 8 randomized controlled trials, a 2025 meta-analysis of 750 participants, and 17 years of safety data, the picture on TA-65 is unusually clear for the supplement category. The TA-65 supplement produces statistically significant telomere elongation in humans. It reduces immunosenescent T-cell burden in a 500-person double-blind trial. It has no oncogenic signal in any published study. The 2024 de Jaeger independent RCT (zero industry ties) replicates the core finding. That's not a marketing claim. It's the published record.
What TA-65 doesn't do: it's not a cure for aging, not a treatment for any disease, and not a substitute for exercise, sleep, or good nutrition. The functional aging claims from the meta-analysis (frailty, systemic inflammation across all studies) aren't yet statistically significant in pooled data. The science supports telomere maintenance and immune cell rejuvenation. The broader functional claims need more time and more trials.
The TA-65 dosage that the evidence supports is one standard 250-unit capsule daily with a fat-containing meal, at a cost of approximately $6.67-$8.33 per day. The TA-65 price is real, and so is what you're buying: the only commercially available cycloastragenol supplement backed by an independent meta-analysis, 17 years of safety data, and third-party COA verification for every batch.
If you're past 50, tracking your biological age, and looking for the supplement with the deepest human evidence base on the specific mechanism of telomere maintenance, TA-65 belongs at the top of your list. Get your baseline telomere test first. Start your protocol. Come back in 12 months with data.
Ready to Start?
RevGenetics offers the authentic TA Sciences-licensed TA-65 supplement with third-party COA verification, cGMP manufacturing, 100% satisfaction guarantee, and 30-day price guarantee.
Get Authentic TA-65 at RevGenetics, Free Shipping on Qualifying Orders
Reviewed by Dr. Hector Valenzuela, PhD (UCLA Telomere Research alumnus). The evidence has been building for 17 years. The research is not waiting for you.