Telomere Research Breakthrough: How Scientists Achieved 24% Life Extension in Mammals
Revolutionary telomere research has unlocked extraordinary possibilities for extending healthy lifespan. Scientists have successfully demonstrated that activating telomerase, the enzyme responsible for maintaining telomeres, can extend median lifespan by up to 24% in mammals while improving multiple health markers. This groundbreaking discovery opens new doors for understanding how telomeres influence aging and longevity.
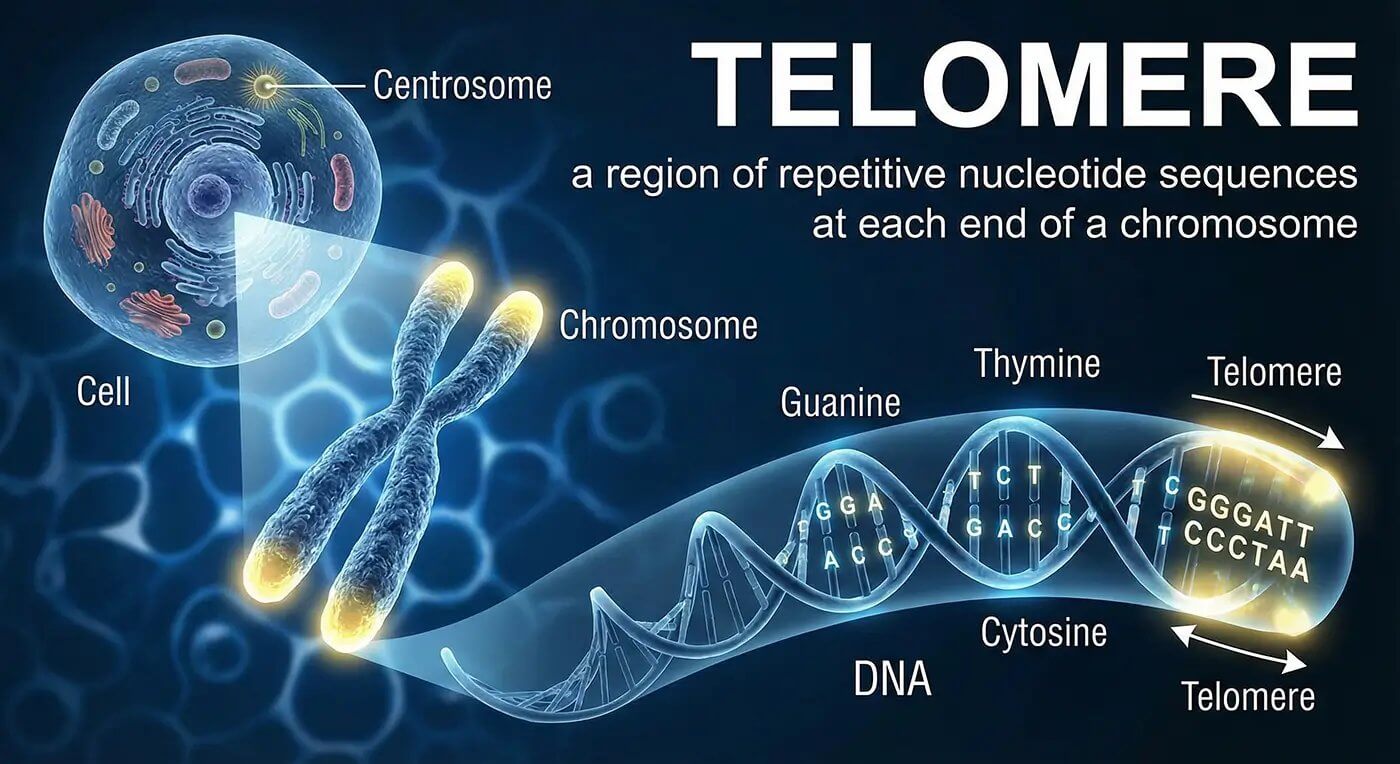
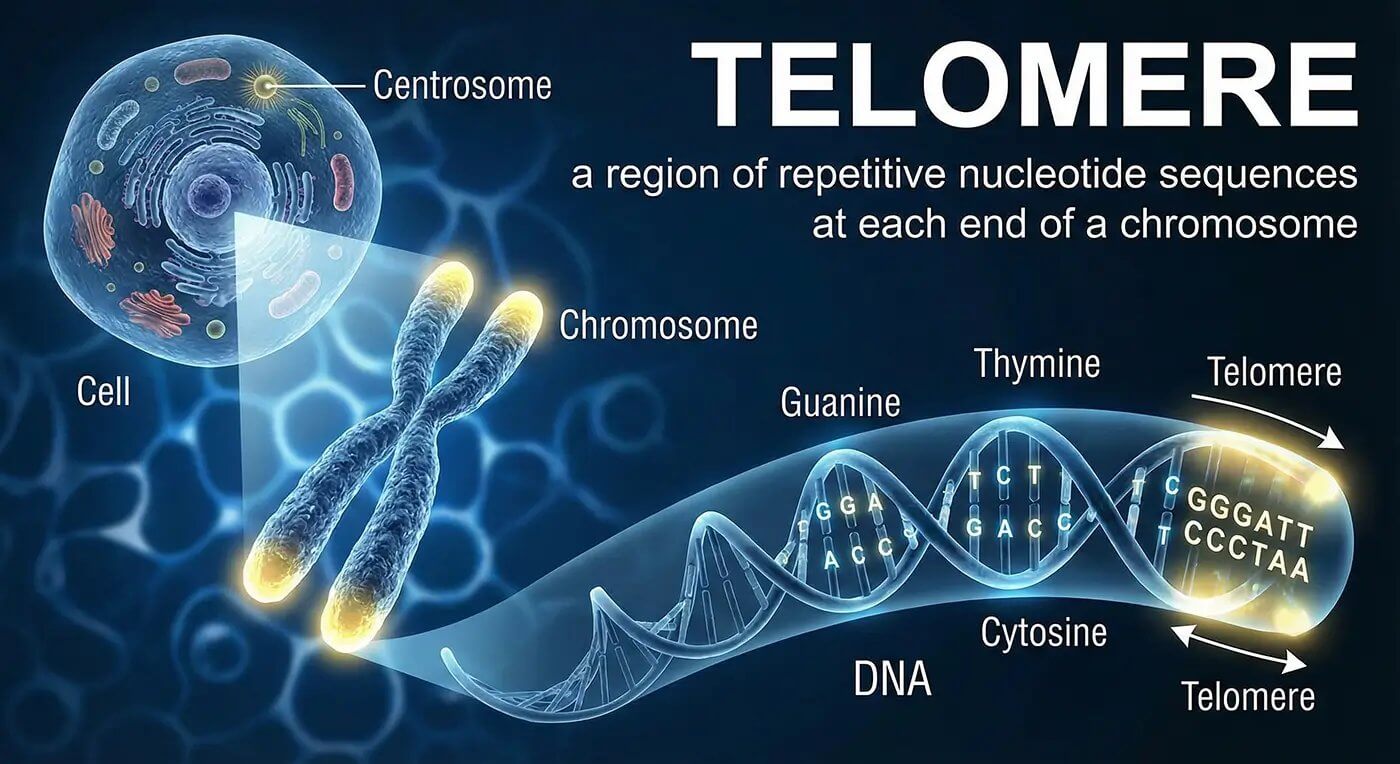
Understanding Telomeres: The Cellular Timekeepers
Telomeres are protective structures located at the ends of chromosomes, often compared to the plastic tips on shoelaces that prevent fraying. These specialized DNA sequences play a crucial role in cellular aging and overall health. Each time a cell divides, telomeres naturally shorten, eventually triggering cellular senescence when they become critically short.
The connection between telomere length and biological aging has captivated researchers worldwide. Longer telomeres are associated with improved cellular function, enhanced disease resistance, and extended healthspan. This relationship has made telomere biology a focal point for longevity research and potential therapeutic interventions.
The Landmark Telomere Immortalization Discovery
The journey toward understanding telomere potential began over a decade ago with a paradigm-shifting experiment. Researchers discovered that adding an extra copy of the gene responsible for telomerase activity could effectively halt cellular aging in human cells. These modified cells remained normal in every measurable way yet demonstrated remarkable resistance to the typical aging process.
This initial breakthrough sparked widespread interest in telomere research across the scientific community. The implications were profound: if cellular aging could be halted or reversed at the molecular level, what possibilities might exist for whole organisms? This question drove researchers to explore telomerase activation as a potential intervention for age-related decline.
From Cells to Organisms: The Telomere Translation Challenge
While the cellular findings were encouraging, translating these results to living organisms presented significant challenges. Scientists needed to determine whether telomerase activation could provide similar benefits at the organismal level without triggering unwanted side effects. Multiple research teams worldwide began investigating various approaches to safely enhance telomerase activity in animal models.
The complexity of whole-organism biology meant that researchers had to consider numerous factors beyond simple telomere lengthening. Questions about tissue-specific effects, immune system responses, and potential cancer risks required careful investigation. Each study contributed valuable insights to the growing body of telomere science.
Breakthrough Study: Telomere Gene Therapy in Aging Mice
The most compelling evidence for telomere-based life extension came from a groundbreaking study published in EMBO journal. Led by renowned scientist Maria Blasco, the research team employed an innovative approach using viral vectors to deliver the mouse telomerase gene directly to living animals.
What made this study particularly remarkable was its focus on already-aged subjects. Rather than starting with young mice, the researchers injected middle-aged and elderly mice with the telomerase gene therapy. This approach more closely mimicked potential human applications, where treatments would likely begin after aging has already progressed.
Extraordinary Results: Life Extension and Health Improvements
The results exceeded expectations. Middle-aged mice experienced a 24% increase in median lifespan, while elderly mice showed a 13% extension. These improvements weren't merely about living longer; the treated mice exhibited comprehensive health benefits that transformed their quality of life.
Key health improvements observed in telomerase-treated mice included:
- Enhanced insulin sensitivity - improving metabolic health and reducing diabetes risk
- Reduced osteoporosis - maintaining stronger bones throughout extended lifespan
- Improved neuromuscular coordination - preserving mobility and cognitive function
- Better overall fitness parameters - maintaining youthful vigor in advanced age
- Improved tissue regeneration - faster healing and recovery capabilities
Addressing the Cancer Concern: Telomere Safety Profile
One persistent concern about telomerase activation has been its potential link to cancer development. Since cancer cells often exhibit high telomerase activity, researchers worried that artificially boosting telomerase might increase cancer risk. The Blasco study provided crucial reassurance on this front.
Remarkably, the telomerase gene therapy did not increase cancer incidence in the treated mice. This finding suggests that properly regulated telomerase activation can provide anti-aging benefits without triggering malignant transformation. The distinction appears to lie in the controlled, physiological activation of telomerase versus the dysregulated expression seen in cancer cells.
How Telomere Extension Works: The Molecular Mechanisms
Understanding how telomere extension produces such dramatic effects requires examining the molecular mechanisms involved. Telomerase acts as a specialized reverse transcriptase, adding repetitive DNA sequences to chromosome ends. This process counteracts the natural telomere shortening that occurs with each cell division.
When telomeres maintain their length, cells can continue dividing without triggering senescence pathways. This sustained cellular renewal capacity translates to improved tissue function, enhanced regenerative potential, and delayed onset of age-related diseases. The systemic benefits observed in the mouse studies reflect this fundamental cellular rejuvenation.
The Telomere-Mitochondria Connection
Recent research has revealed intricate connections between telomere biology and mitochondrial function. Longer telomeres correlate with improved mitochondrial efficiency, leading to better cellular energy production and reduced oxidative stress. This relationship helps explain the wide-ranging health benefits observed with telomerase activation.
Current Telomere Research: Progress Toward Human Applications
While direct gene therapy remains years away from human application, researchers are exploring multiple avenues to harness telomere science for health benefits. Current approaches include:
- Small molecule telomerase activators - compounds that naturally boost telomerase expression
- Lifestyle interventions - diet, exercise, and stress reduction strategies that support telomere health
- Nutraceutical approaches - supplements targeting telomere maintenance pathways
- Cellular reprogramming techniques - methods to reset telomere length in specific cell types
- Biomarker development - tools to monitor telomere status and treatment efficacy
Natural Ways to Support Telomere Health
While awaiting advanced therapies, individuals can take proactive steps to support their telomere health. Research has identified several lifestyle factors that positively influence telomere length and telomerase activity:
Nutrition for Telomere Support
Certain dietary patterns show strong associations with longer telomeres. Mediterranean-style diets rich in antioxidants, omega-3 fatty acids, and polyphenols provide cellular protection. Specific nutrients linked to telomere health include vitamin D, folate, and magnesium. Minimizing processed foods and sugar intake also supports telomere maintenance.
Exercise and Telomere Length
Regular physical activity emerges as one of the most powerful telomere-supporting interventions. Both aerobic exercise and resistance training show benefits, with endurance athletes often displaying longer telomeres than sedentary individuals. The optimal exercise prescription appears to be moderate-intensity activity performed consistently over time.
Stress Management and Telomere Protection
Chronic stress accelerates telomere shortening through increased oxidative damage and inflammation. Mindfulness practices, meditation, and stress reduction techniques have demonstrated measurable effects on telomerase activity. Studies show that individuals practicing regular meditation can maintain longer telomeres compared to non-meditators.
The Future of Telomere Therapeutics
The successful demonstration of telomerase gene therapy in mice represents a pivotal moment in aging research. As scientists work toward safe human applications, several exciting developments are on the horizon:
Precision medicine approaches will likely tailor telomere interventions to individual genetic profiles and health status. Advanced delivery systems may enable targeted telomerase activation in specific tissues or organs. Combination therapies integrating telomere support with other longevity interventions could amplify benefits.
The pharmaceutical industry has taken notice, with multiple companies developing telomerase-targeting compounds. Clinical trials are underway testing various approaches to telomere maintenance in age-related diseases. These efforts bring the promise of telomere therapeutics closer to reality.
Ethical Considerations and Societal Impact
As telomere science advances toward human application, important ethical questions arise. Extended healthspan could transform society, affecting everything from retirement planning to healthcare systems. Ensuring equitable access to telomere therapeutics will be crucial for preventing disparities in longevity.
The potential to significantly extend human healthspan also raises philosophical questions about the nature of aging and mortality. However, the focus remains on adding healthy, productive years rather than simply extending lifespan. The goal is compression of morbidity - maintaining vitality until very late in life.
Conclusion: The Telomere Revolution
The demonstration that telomerase activation can extend lifespan by 24% while improving multiple health parameters marks a watershed moment in longevity science. This proof-of-principle study validates decades of telomere research and opens exciting possibilities for human health optimization.
While gene therapy applications await further development, the fundamental discovery that telomere maintenance can delay organismal aging provides hope for future interventions. Combined with our growing understanding of lifestyle factors that support telomere health, individuals can take proactive steps today while anticipating more powerful tools tomorrow.
The telomere revolution has begun. As research continues to unlock the secrets of these cellular timekeepers, we move closer to a future where extended healthspan becomes achievable for everyone. The journey from that first immortalized cell line to successful organism-wide life extension demonstrates the power of persistent scientific inquiry and the tremendous potential within our own biology.
Frequently Asked Questions About Telomeres
What exactly are telomeres and why do they matter for aging?
Telomeres are protective DNA-protein structures at the ends of chromosomes that act like biological clocks. They shorten with each cell division, and when they become critically short, cells stop dividing and enter senescence. This process is fundamental to aging. Maintaining telomere length through telomerase activation has been shown to extend lifespan by up to 24% in mammals while improving multiple health markers including insulin sensitivity, bone density, and neuromuscular function.
How can I naturally support my telomere health?
You can support telomere health through several evidence-based lifestyle strategies. Regular moderate exercise, particularly endurance activities, has strong associations with longer telomeres. A Mediterranean-style diet rich in antioxidants, omega-3 fatty acids, and whole foods provides cellular protection. Stress management through meditation or mindfulness practices can boost telomerase activity. Quality sleep, maintaining a healthy weight, and avoiding smoking also contribute to telomere maintenance. These interventions work synergistically to slow telomere shortening and may even promote lengthening in some cases.
Is telomerase activation safe, or does it increase cancer risk?
The landmark study by Maria Blasco's team provided crucial evidence that controlled telomerase activation does not increase cancer incidence. While cancer cells often show high telomerase activity, the regulated activation achieved through gene therapy appears safe. The key distinction lies in physiological versus pathological activation. Properly controlled telomerase expression provides anti-aging benefits without triggering malignant transformation. This finding has opened the door for developing safe telomerase-based therapies for human use.
When will telomere therapies be available for humans?
While direct gene therapy remains years away, several telomere-supporting interventions are already available or in development. Small molecule telomerase activators are being tested in clinical trials. Nutraceutical approaches using natural compounds show promise. Some researchers estimate that approved telomerase-targeting drugs could be available within 5-10 years for specific conditions. Meanwhile, lifestyle interventions proven to support telomere health can be implemented immediately. The future likely holds a combination of pharmaceutical and natural approaches to telomere maintenance.
Can telomere length be measured, and should I get tested?
Yes, telomere length can be measured through specialized laboratory tests using blood samples. These tests typically measure average telomere length in white blood cells. While interesting, current telomere testing has limitations. Results can vary between labs, and we're still learning how to interpret individual measurements. Rather than focusing on single tests, tracking changes over time may be more valuable. Consider telomere testing if you're monitoring the effects of specific interventions or have a family history of age-related diseases. Always discuss results with a healthcare provider familiar with telomere science.
What's the difference between telomeres and telomerase?
Telomeres are the actual protective structures at chromosome ends, consisting of repetitive DNA sequences and associated proteins. Telomerase is the enzyme responsible for adding new DNA to telomeres, counteracting their natural shortening. Think of telomeres as the protective caps and telomerase as the repair mechanism. Most adult cells have low telomerase activity, which is why telomeres shorten over time. Activating telomerase can maintain or even lengthen telomeres, as demonstrated in the mouse studies showing 24% life extension.
Do telomere supplements really work?
Several supplements show promise for supporting telomere health, though effects are generally modest compared to pharmaceutical interventions. Compounds like astragalus extracts, omega-3 fatty acids, and certain antioxidants have demonstrated telomerase-activating properties in studies. Quality and dosing matter significantly. The most compelling evidence supports comprehensive approaches combining multiple nutrients rather than single supplements. While supplements can be part of a telomere support strategy, they work best alongside healthy lifestyle practices. Always choose high-quality products and consult healthcare providers, especially if taking medications.
How do telomeres affect different organs and systems?
Telomere length impacts virtually every organ system, though effects vary by tissue type. In the cardiovascular system, shorter telomeres associate with increased heart disease risk. The immune system particularly depends on telomere maintenance, as immune cells must divide frequently. Brain health correlates with telomere length, affecting cognitive function and neurodegenerative disease risk. Metabolic organs like the liver and pancreas show improved function with longer telomeres, as seen in the improved insulin sensitivity in telomerase-treated mice. This system-wide impact explains the comprehensive health benefits observed in life extension studies.
What makes the recent telomere research breakthrough so significant?
The Blasco study represents a pivotal achievement because it successfully translated cellular findings to whole organisms, achieving 24% life extension in middle-aged mice through telomerase gene therapy. Previous studies showed promise in cells, but this research proved the concept works safely in living mammals without increasing cancer risk. The comprehensive health improvements, including better insulin sensitivity, stronger bones, and enhanced coordination, demonstrate that telomere intervention doesn't just extend lifespan but improves healthspan. This proof-of-principle brings us significantly closer to developing human therapies.
Are there any risks or downsides to having longer telomeres?
While longer telomeres generally correlate with better health outcomes, the relationship isn't always straightforward. Extremely long telomeres in certain contexts might theoretically allow damaged cells to persist longer than optimal. However, research consistently shows that physiologically longer telomeres within the normal range provide health benefits. The key is balanced telomerase activation that maintains healthy telomere length without allowing unlimited cell division. Current evidence strongly supports the benefits of telomere maintenance strategies, with the mouse studies showing no adverse effects from controlled telomerase activation.