📖 Complete Resveratrol Encyclopedia
286 sections across 8 categories — click any group to explore
The Chemistry (Keep It Simple Version)
Resveratrol exists in two molecular shapes: trans and cis. Picture them as mirror images of each other. Trans-resveratrol has a flat, extended structure that lets it bind effectively to cellular receptors. Cis-resveratrol is kinked and bent, which makes it much less biologically active.
When plants make resveratrol in response to stress (fungal attacks, UV exposure), they primarily produce the trans form. However, cis-resveratrol can form when trans-resveratrol is exposed to light or heat during manufacturing and storage. Cheap, poorly-made supplements often contain significant amounts of the inactive cis form.
Why Trans-Resveratrol Performs Better
The research on resveratrol's benefits (SIRT1 activation, cardiovascular protection, blood sugar effects) was conducted almost exclusively with trans-resveratrol. When you buy a product that just says "resveratrol" without specifying the trans form, you might be getting a mix that includes 20-40% useless cis-resveratrol.
The research on resveratrol's benefits (SIRT1 activation, cardiovascular protection, blood sugar effects) was conducted almost exclusively with trans-resveratrol. Studies confirm pure trans-resveratrol achieves higher plasma concentrations than mixed isomer products. [Wenzel and Somoza, 2005] That's a significant difference in what actually reaches your cells.
How Trans-Resveratrol Works In Your Body
Once absorbed, trans-resveratrol activates SIRT1, a protein often called the "longevity gene" because of its role in cellular repair and metabolic regulation. It also upregulates your body's own antioxidant systems (like glutathione production), rather than just acting as a direct antioxidant itself. This gives you sustained protection rather than a brief spike.
The compound also modulates NF-kB, a key inflammatory pathway involved in everything from heart disease to cancer. A systematic review and meta-analysis found that resveratrol supplementation significantly reduced TNF-alpha and hs-CRP levels in patients with cardiovascular disease. [Teimouri et al., 2022]
Buying Trans-Resveratrol: What to Look For
Not all supplements are created equal. Here's your checklist:
- Explicit "trans-resveratrol" labeling: If it doesn't say trans, assume it's a mix.
- Purity of 98%+ (ideally 99%): The remaining 1-2% is typically residual processing compounds, not cis-resveratrol.
- Third-party testing: Certificates of Analysis (COAs) from independent labs. Ask for them if not posted.
- Dark packaging: Light degrades trans-resveratrol to the cis form. Amber or opaque bottles matter.
- Source disclosed: Most quality trans-resveratrol comes from Japanese knotweed (Polygonum cuspidatum). Some comes from grape skin extract.
Dosing Recommendations
Most clinical trials showing benefits used 150-500mg of trans-resveratrol daily. Start at 150-250mg for a couple weeks, then increase if you want. Taking it with fat (a meal, olive oil, nuts) improves absorption significantly. Some people split their dose morning and evening because of the short half-life.
Higher doses (1g+) have been used in some research contexts, but there's limited evidence that more is better for general health purposes. The cardiovascular and metabolic benefits seem to plateau around 500mg.
What to Realistically Expect
Let's set reasonable expectations. Trans-resveratrol isn't going to make you feel 20 years younger overnight. What users commonly report:
- Subtle energy improvements within 2-4 weeks
- Better exercise recovery
- Improved blood work markers (cholesterol, inflammatory markers) at 8-12 weeks
- Some people notice skin improvements; others don't
The real benefits are likely happening at the cellular level where you can't feel them: reduced oxidative damage, better mitochondrial function, lower chronic inflammation. Think of it as maintenance for your cells rather than a performance enhancer you'll notice immediately.
Trans-Resveratrol FAQs
Is there really that big a difference between trans and cis resveratrol?
Yes. Cis-resveratrol has minimal biological activity. Studies showing health benefits use trans-resveratrol almost exclusively. A supplement containing 40% cis-resveratrol gives you 40% less of what actually works. It's like paying for watered-down juice.
How fast will I notice anything from trans-resveratrol?
Energy and subtle cognitive improvements: 2-4 weeks for most people. Measurable changes in blood markers: 8-12 weeks minimum. Anti-aging effects at the cellular level: you won't "feel" these, but they're happening if you're consistent. This is a marathon supplement, not a sprint.
Can I take trans-resveratrol with my medications?
Generally safe with most medications, but two cautions: it has mild blood-thinning effects (watch out if you're on anticoagulants) and it may enhance the effects of diabetes medications. Talk to your doctor before combining.
What dose of trans-resveratrol actually does something?
Most positive human trials used 150-500mg daily. The cardiovascular benefits appear to plateau around 500mg. Starting at 250mg is reasonable. Higher doses (1g+) haven't shown proportionally better results in healthy people.
Any side effects from trans-resveratrol?
Most people tolerate it well. Some experience mild GI upset initially, especially with lower-purity products. Taking it with food usually solves this. At very high doses (2g+), some people report joint discomfort. Rare cases of headaches during the first week.
What's the best way to take trans-resveratrol?
With food containing fat for better absorption. Morning or split morning/evening work well. Stay consistent: the benefits build over time. Don't take it right before bed if you notice an energy boost.
Micronized Resveratrol: Does Smaller Particle Size Actually Matter?
You've probably seen "micronized" on supplement labels and wondered if it's real science or marketing speak. Good news: this one actually has substance behind it. Micronized resveratrol addresses one of the compound's biggest practical problems: it's poorly absorbed in standard form.
The Bioavailability Problem (And How Micronization Helps)
Here's the frustrating reality about regular resveratrol: your body absorbs it quickly but metabolizes it even faster. Blood levels peak within 1-2 hours then crash. Studies estimate that only about 20% of ingested resveratrol actually makes it into systemic circulation in usable form. The rest gets glucuronidated (chemically modified) by your liver before it can do much.
Micronized resveratrol tackles this through physics. By grinding particles down to microscopic sizes (typically 1-5 microns), manufacturers dramatically increase surface area. More surface area means faster dissolution, which means more absorption before your gut and liver can neutralize it.
Think about dissolving sugar in water. Powdered sugar dissolves almost instantly. Granulated takes longer. Sugar cubes take forever. Same principle applies here. Pharmacokinetic studies confirm that particle size reduction substantially increases dissolution rate and thus absorption of resveratrol, with micronized formulations delivering significantly higher plasma concentrations. [Nunes et al., 2024] That's a meaningful difference.

Brain Benefits: Getting Resveratrol Across The Blood-Brain Barrier
Micronized resveratrol may have particular advantages for brain health because higher plasma concentrations translate to more compound crossing into brain tissue. Early research suggests benefits for:
- Cerebral blood flow: A 2024 fMRI study showed improved brain perfusion in older adults taking micronized resveratrol for 12 weeks.
- Neuroprotection: Reduced markers of oxidative stress in cerebrospinal fluid.
- Memory and attention: Modest improvements in working memory tasks compared to standard formulations.
The Alzheimer's prevention trials currently underway are using enhanced-bioavailability formulations specifically because standard resveratrol didn't achieve adequate brain concentrations.
Is Micronized Worth The Extra Cost?
Real talk: micronized resveratrol typically costs 30-50% more than standard formulations. Is it worth it?
The math works out favorably. If micronized versions achieve 2-3x better absorption, you'd need to take 2-3x more standard resveratrol to match the effect. That actually makes micronized cheaper per unit of absorbed compound. Plus, higher doses of standard resveratrol increase the risk of GI side effects.
For cardiovascular and cognitive applications where blood levels matter, micronization makes sense. For general antioxidant purposes where you're just trying to get "some" resveratrol into your system, standard formulations may be fine.
Dosing Micronized Resveratrol
Because absorption is better, you can use lower doses of micronized resveratrol to achieve similar effects:
- General health: 100-200mg daily (equivalent to ~300-500mg standard)
- Cardiovascular support: 200-400mg daily
- Cognitive enhancement: 150-300mg daily
Still take it with fat for optimal absorption. The micronization helps, but the fat-solubility issue remains.
Making Micronized Resveratrol Part of Your Wellness Journey
Incorporating micronized resveratrol into your daily routine represents an investment in your long-term health and vitality. The enhanced absorption and bioavailability of micronized formulations mean you can experience the full spectrum of resveratrol's benefits more effectively than ever before.
Whether your goals include supporting heart health, maintaining cognitive sharpness, or promoting healthy aging, micronized resveratrol offers a scientifically-backed approach to achieving optimal wellness. The key lies in choosing high-quality supplements, following appropriate dosing guidelines, and maintaining consistency in your supplementation routine.
For those ready to experience the transformative benefits of micronized resveratrol, consider exploring M98, an advanced formulation designed to deliver maximum bioavailability and therapeutic effects. With its commitment to purity, potency, and scientific innovation, M98 represents the cutting edge of resveratrol supplementation. Discover how M98 can elevate your health journey today!
Frequently Asked Questions About Micronized Resveratrol
What makes micronized resveratrol different from regular resveratrol supplements?
Micronized resveratrol undergoes a special process that reduces particle size to microscopic levels, dramatically increasing surface area and absorption rates. This enhanced bioavailability means your body can absorb significantly more resveratrol compared to standard supplements, delivering superior health benefits with smaller doses. [Howells et al., 2011] The micronization process also improves stability, ensuring consistent potency and effectiveness.
How long does it take to see results from micronized resveratrol?
Many people begin noticing improvements in energy levels and mental clarity within 2-4 weeks of starting micronized resveratrol supplementation. Cardiovascular benefits and markers of improved health typically become measurable after 8-12 weeks of consistent use. Long-term benefits for healthy aging and cellular protection accumulate over months and years of regular supplementation. Individual results vary based on dosage, overall health status, and lifestyle factors.
Can I take micronized resveratrol with other supplements?
Yes, micronized resveratrol generally works well with most supplements and may even enhance the effects of certain nutrients. It pairs particularly well with omega-3 fatty acids, curcumin, and quercetin for synergistic antioxidant effects. Some people combine it with NAD+ precursors for enhanced cellular energy support. However, always space out supplements throughout the day for optimal absorption and consult with healthcare providers about specific combinations.
What is the best time of day to take micronized resveratrol?
The optimal timing for micronized resveratrol depends on your goals and individual response. Many people prefer taking it in the morning with breakfast to support daytime energy and mental clarity. Others find evening doses help with recovery and cellular repair during sleep. Taking it with a meal containing healthy fats enhances absorption. Some users split their daily dose between morning and evening for sustained benefits throughout the day.
Is micronized resveratrol safe for long-term use?
Research indicates that micronized resveratrol is safe for long-term supplementation when taken at recommended doses. Studies spanning several years show no significant adverse effects, and many of resveratrol's benefits, particularly for healthy aging and cardiovascular protection, are best achieved through consistent, long-term use. Regular health check-ups can help monitor your response and ensure continued safety.
How do I know if I'm getting genuine micronized resveratrol?
Look for supplements that specifically state "micronized resveratrol" on the label and provide particle size specifications. Reputable manufacturers will offer certificates of analysis showing purity levels of 98% or higher and third-party testing results. The supplement should come in dark, protective packaging to prevent degradation. Quality brands also provide detailed information about their micronization process and manufacturing standards.
Can micronized resveratrol help with weight management?
Micronized resveratrol supports healthy metabolism through multiple mechanisms, including improved insulin sensitivity, enhanced fat oxidation, and activation of SIRT1 pathways involved in metabolic regulation. While not a weight loss supplement per se, it can support healthy weight management as part of a balanced diet and exercise program. Users often report better energy levels and metabolic function, which can contribute to maintaining a healthy weight.
What dosage of micronized resveratrol should I start with?
Begin with 150-250 mg of micronized resveratrol daily, taken with food to maximize absorption and minimize any digestive sensitivity. After 2-3 weeks, you can gradually increase the dose based on your response and health goals. Most people find their optimal dose between 250-500 mg daily. Remember that micronized forms require lower doses than standard resveratrol due to superior absorption.
Are there any groups who should avoid micronized resveratrol?
While micronized resveratrol is safe for most adults, certain groups should exercise caution or avoid it. Pregnant and nursing women should consult their healthcare providers before use. People taking blood-thinning medications need medical supervision due to potential interactions. Those scheduled for surgery should discontinue use two weeks prior. Individuals with hormone-sensitive conditions should discuss supplementation with their doctors.
How does micronized resveratrol compare to getting resveratrol from food sources?
While foods like red grapes, berries, and peanuts contain resveratrol, the amounts are relatively small. You would need to consume impractical quantities of these foods to match the resveratrol content in a single micronized supplement capsule. Additionally, micronized resveratrol offers superior bioavailability compared to food sources, meaning your body can utilize it more effectively. Supplements provide a concentrated, consistent dose that's difficult to achieve through diet alone.
Revisiting the proper Resveratrol Dosage
The Science Behind Trans-Resveratrol's Powerful Benefits
Scientific research reveals that trans-resveratrol activates specific longevity pathways in your cells, particularly the SIRT1 protein. [Howitz et al., 2003] This activation triggers a cascade of beneficial effects throughout your body, from enhanced mitochondrial function to improved cellular repair mechanisms. Studies indicate that trans-resveratrol supplementation may support healthy aging by protecting DNA integrity and reducing oxidative stress markers.
The compound belongs to a class of molecules called stilbenes, which plants produce as a defense mechanism against environmental stressors. When you consume trans-resveratrol, your body harnesses these protective properties to combat inflammation, support cardiovascular health, and maintain optimal blood glucose levels.
Proven Health Benefits of Trans-Resveratrol Supplementation
Research demonstrates multiple pathways through which trans-resveratrol supports optimal health:
- Cardiovascular Protection: Trans-resveratrol helps maintain healthy cholesterol levels and supports arterial flexibility
- Brain Health Support: Its unique ability to cross the blood-brain barrier provides neuroprotective benefits
- Cellular Energy Enhancement: Activates mitochondrial biogenesis for improved energy production
- Healthy Aging Support: Stimulates longevity genes and cellular repair mechanisms
- Metabolic Balance: Helps regulate healthy blood sugar levels and insulin sensitivity
Making Trans-Resveratrol Part of Your Wellness Journey
Incorporating trans-resveratrol into your daily routine represents an investment in long-term health and vitality. Start with a quality supplement from a reputable manufacturer, beginning with lower doses to assess your individual response. Combine supplementation with a Mediterranean-style diet rich in colorful fruits and vegetables to maximize antioxidant synergy.
Remember that consistency yields the best results with trans-resveratrol supplementation. Most users report noticeable benefits after 4-8 weeks of regular use, with continued improvements over several months. Track your progress by noting energy levels, mental clarity, and overall sense of wellbeing.
Your journey with trans-resveratrol can open doors to enhanced vitality, sharper cognition, and graceful aging. This remarkable compound offers science-backed support for multiple aspects of health, from cellular protection to cardiovascular wellness. By choosing high-quality trans-resveratrol supplements and following optimal usage guidelines, you position yourself for a healthier, more vibrant future.
Frequently Asked Questions About Trans-Resveratrol
What is the recommended daily dose of trans-resveratrol for anti-aging benefits?
For anti-aging support, most research suggests taking 500-1000mg of trans-resveratrol daily. Split this dose between morning and evening for optimal absorption and sustained benefits throughout the day. Start with a lower dose of 250mg to assess tolerance, then gradually increase over several weeks. Many successful users find their sweet spot around 750mg daily, experiencing improved energy, mental clarity, and skin appearance.
Can trans-resveratrol help with weight management and metabolism?
Yes, trans-resveratrol shows promising effects on metabolic health and weight management. Research indicates it activates AMPK, often called the "metabolic master switch," which helps regulate energy balance and fat metabolism. [Um et al., 2010] Users often report improved insulin sensitivity, better appetite control, and enhanced fat-burning during exercise. For metabolic support, take 500mg of trans-resveratrol 30 minutes before your largest meal of the day.
How long does it take to see results from trans-resveratrol supplementation?
Individual responses vary, but most people notice initial benefits within 2-4 weeks of consistent trans-resveratrol supplementation. Energy improvements and better sleep quality often appear first. Cardiovascular benefits and anti-aging effects typically become noticeable after 6-8 weeks. For maximum results, commit to at least 3 months of daily supplementation while maintaining a healthy lifestyle. Many users report continued improvements up to 6 months into their supplementation journey.
Is trans-resveratrol safe to take with other medications?
Trans-resveratrol is generally safe for most people, but it can interact with certain medications. Exercise caution if you take blood thinners, NSAIDs, or medications metabolized by specific liver enzymes. The compound may enhance the effects of anticoagulants and could theoretically interact with hormone-sensitive medications. Always consult your healthcare provider before adding trans-resveratrol to your regimen, especially if you take prescription medications regularly.
What's the best time of day to take trans-resveratrol?
The optimal timing for trans-resveratrol depends on your health goals. For anti-aging and cellular repair benefits, take it in the morning when your body's repair processes peak. For cardiovascular support, evening supplementation aligns with nighttime cholesterol synthesis. Athletes benefit from taking trans-resveratrol 30-60 minutes before workouts for enhanced performance and recovery. Always take it with food containing healthy fats to maximize absorption.
Can I get enough trans-resveratrol from food alone?
While foods like red grapes, berries, and red wine contain trans-resveratrol, achieving therapeutic doses through diet alone proves challenging. You would need to consume enormous quantities of these foods daily to match supplement levels. For example, obtaining 500mg of trans-resveratrol would require drinking hundreds of glasses of red wine or eating pounds of grapes daily. Quality supplements provide concentrated, standardized doses that deliver consistent health benefits without excessive calorie intake.
Does trans-resveratrol really help with brain health and memory?
Absolutely! Trans-resveratrol's unique ability to cross the blood-brain barrier makes it exceptionally beneficial for cognitive health. Studies show it protects neurons from oxidative stress, reduces brain inflammation, and may even support the growth of new brain cells. Regular users report improved focus, clearer thinking, and better memory recall. For cognitive benefits, combine 500-750mg of trans-resveratrol with omega-3 fatty acids and regular mental exercise for optimal brain health support.
What should I look for when choosing a trans-resveratrol supplement?
Select supplements that specifically state "trans-resveratrol" content with at least 98% purity. Look for third-party testing verification and GMP certification. Avoid products that only list "resveratrol" without specifying the trans form. Quality supplements often include absorption enhancers like black pepper extract or quercetin. Choose brands that provide clear dosing information and use dark bottles to protect against light degradation. Premium products typically cost more but deliver superior results through better bioavailability and purity.
Are there any age restrictions for taking trans-resveratrol?
Trans-resveratrol is generally recommended for adults over 18 years old. While no specific upper age limit exists, older adults often experience the most dramatic benefits due to age-related cellular changes. People in their 40s and beyond particularly appreciate its anti-aging and energy-supporting properties. Younger adults may use trans-resveratrol for athletic performance, cognitive enhancement, or preventive health maintenance. Children and teenagers should only use trans-resveratrol under medical supervision for specific health conditions.
Can trans-resveratrol improve exercise performance and recovery?
Trans-resveratrol offers excellent support for athletic performance and post-workout recovery. It enhances mitochondrial function, improving cellular energy production during exercise. Athletes report better endurance, reduced muscle fatigue, and faster recovery times. The compound's anti-inflammatory properties help minimize exercise-induced muscle damage. For optimal athletic benefits, take 500-1000mg of trans-resveratrol 30-60 minutes before training, and consider a second dose post-workout to accelerate recovery processes.
Resveratrol and Aspirin can Lower Cancer Incidence.
Understanding Resveratrol's Role in Cellular Protection
Cellular replication represents one of nature's most intricate processes, where even minor errors can have profound consequences. When DNA replication goes awry, the results can range from accelerated aging conditions like Progeria to the development of cancer. Recent research has illuminated how resveratrol may serve as a protective agent against these cellular mishaps, particularly in preventing the cascade of mutations that lead to cancer formation.
The journey from healthy cell to cancerous cell often begins with a specific type of DNA error called polyploidy, where cells create extra sets of DNA copies. While most cells cannot survive this mutation and naturally die off or face elimination by our immune system, the few that persist can trigger a domino effect of genetic changes. This understanding has driven researchers to seek compounds that can target these polyploid cells before they transform into cancer.
Groundbreaking Study: How Resveratrol and Aspirin Target Cancer Cells
A landmark study published in the Proceedings of the National Academy of Sciences revealed fascinating insights into how resveratrol works at the molecular level to combat cancer. Researchers discovered that polyploid cells, while resistant to many anti-cancer agents, show particular vulnerability to compounds that activate AMP-activated protein kinase (AMPK), a important cellular energy sensor.
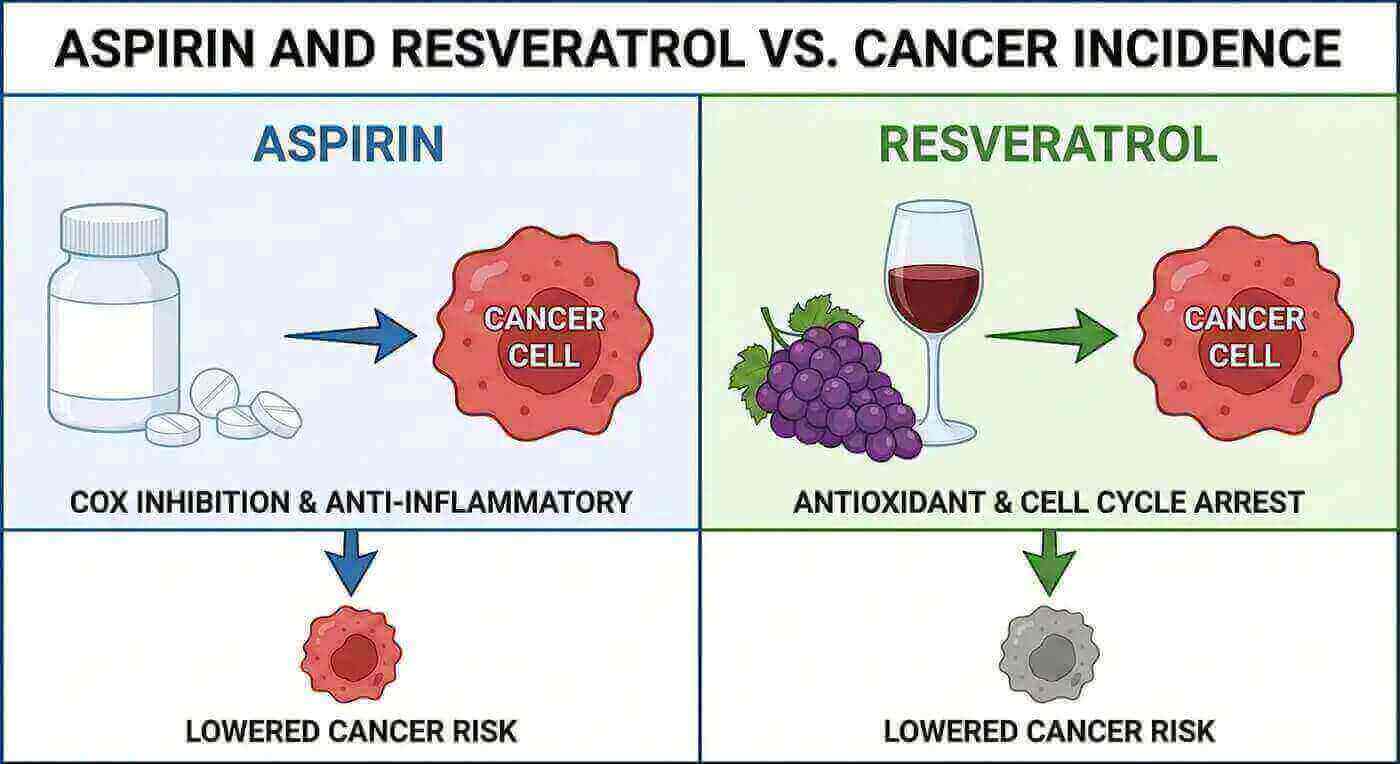
This discovery led scientists to screen various compounds for AMPK activation potential. Among the tested substances, resveratrol and aspirin emerged as champions, demonstrating selective killing of both human colon cancer cell lines and mouse epithelial cancer cells. The implications of these findings offer tremendous hope for those seeking natural approaches to cancer prevention.
Specific Cancer Types Affected by Resveratrol Treatment
The research specifically focused on colon cancer models, where resveratrol showed exceptional promise. In controlled laboratory settings, researchers observed that resveratrol could:
- Selectively target and eliminate human colon cancer cell lines
- Effectively kill mouse epithelial cancer cells
- Demonstrate consistent results in live mouse colon cancer models
- Work synergistically with the body's natural defense mechanisms
These findings suggest that resveratrol doesn't just randomly attack cells but instead precisely targets those with cancerous potential, leaving healthy cells unharmed. This selectivity makes resveratrol particularly attractive for preventive health strategies.
The Science Behind Resveratrol's Cancer-Fighting Mechanism
Understanding how resveratrol accomplishes its cancer-fighting effects provides valuable insight into its potential applications. The research team meticulously proved that resveratrol's anti-cancer activity specifically requires AMPK activation. This discovery carries significant weight because:
- Targeted Action: AMPK activation triggers specific cellular pathways that make cancer cells vulnerable
- Multiple Benefits: While resveratrol affects various cellular targets, AMPK activation appears central to its anti-cancer effects
- Comparative Advantage: Unlike aspirin, which also activates AMPK, resveratrol doesn't carry risks of gastrointestinal complications
- Natural Origin: As a compound found in grapes, berries, and peanuts, resveratrol offers a nature-based approach to health
AMPK Activation: The Key to Resveratrol's Success
AMPK serves as a master regulator of cellular energy metabolism. When resveratrol activates this protein, it triggers a cascade of beneficial effects:
| AMPK Effect | Benefit for Cancer Prevention |
|---|---|
| Energy Metabolism Regulation | Starves cancer cells of necessary resources |
| Cell Growth Inhibition | Prevents uncontrolled cellular proliferation |
| Autophagy Activation | Helps eliminate damaged cellular components |
| DNA Repair Enhancement | Reduces mutation accumulation |
Resveratrol vs. Aspirin: Making the Safer Choice
While both resveratrol and aspirin demonstrated similar AMPK activation levels in the study, important differences emerge when considering long-term use. Aspirin, despite its benefits, carries well-documented risks of gastrointestinal hemorrhage with extended use. This side effect can be particularly concerning for individuals seeking preventive health measures.
Resveratrol, conversely, offers a gentler approach to achieving similar cellular benefits. As a naturally occurring compound with a long history of safe consumption through dietary sources, resveratrol presents an attractive alternative for those prioritizing both efficacy and safety in their health regimen.
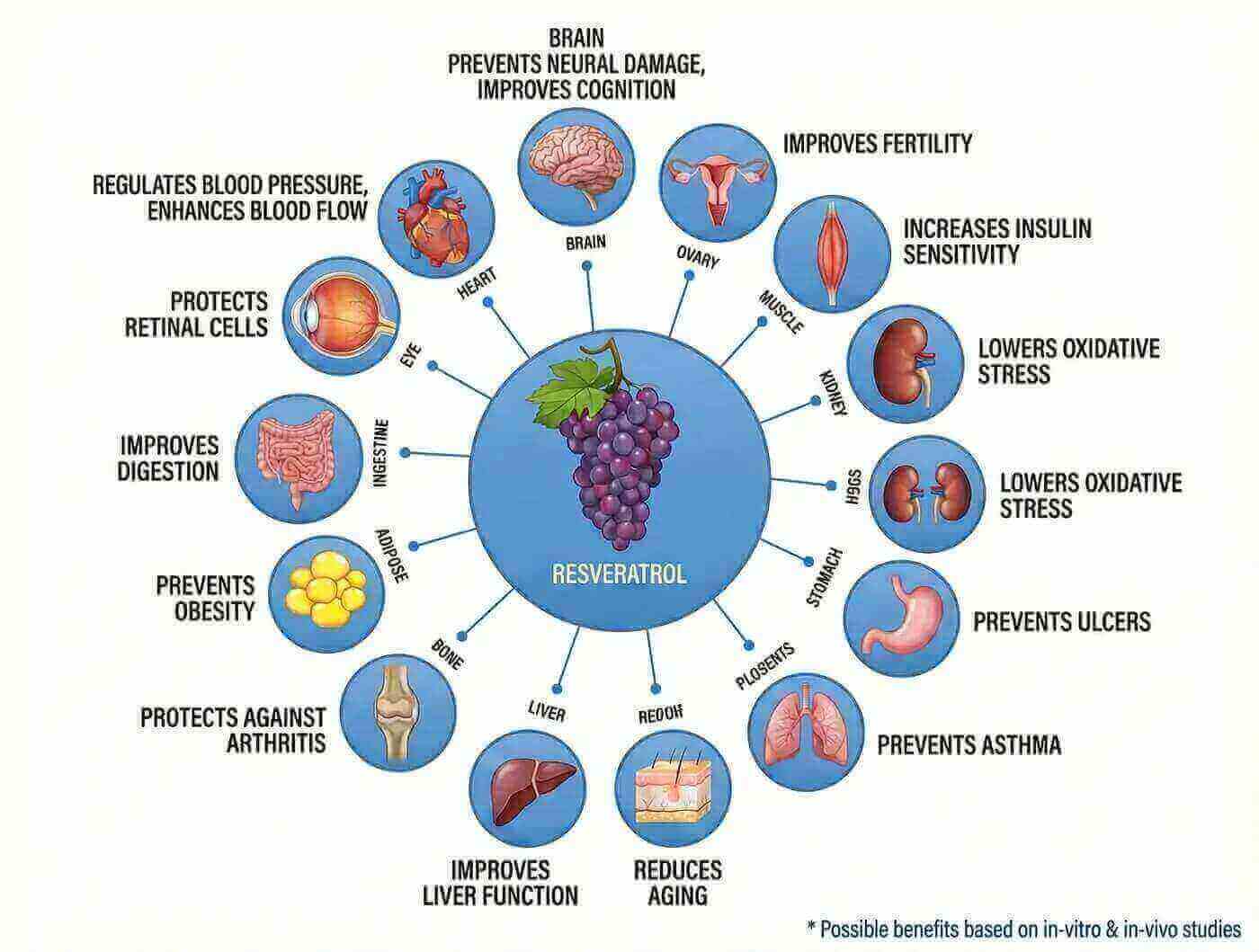
Additional Benefits of Choosing Resveratrol
Beyond its cancer-preventive properties, resveratrol offers a spectrum of health benefits that make it an exceptional choice for overall wellness:
- Cardiovascular Protection: Supports heart health through multiple mechanisms
- Anti-Aging Properties: Activates sirtuins, proteins associated with longevity
- Neuroprotective Effects: May help protect brain cells from age-related decline
- Anti-Inflammatory Action: Reduces chronic inflammation linked to various diseases
- Metabolic Support: Aids in maintaining healthy blood sugar levels
Implementing Resveratrol for Optimal Health Benefits
Understanding how to incorporate resveratrol into your wellness routine can maximize its protective effects. While dietary sources provide some resveratrol, achieving therapeutic levels often requires supplementation. Consider these approaches:
Dietary Sources of Resveratrol
Natural food sources containing resveratrol include:
- Red Grapes: Particularly the skin contains high concentrations
- Blueberries: Offer resveratrol along with other beneficial compounds
- Cranberries: Provide modest amounts of resveratrol
- Peanuts: Contain resveratrol, especially when roasted
- Dark Chocolate: Small amounts present in high-quality varieties
Supplementation Strategies
For those seeking therapeutic levels of resveratrol, supplementation offers a practical solution. High-quality resveratrol supplements can provide standardized doses that would be difficult to achieve through diet alone. When selecting a supplement, consider:
- Purity: Look for products with verified resveratrol content
- Bioavailability: Some formulations enhance absorption
- Dosage: Research suggests 250-500mg daily for optimal benefits
- Quality Standards: Choose products from reputable manufacturers
How NMN Works in Your Body
When you consume NMN, whether through foods or supplements, a fascinating process begins. The molecule utilizes a specialized transporter called Slc12a8, located on the cell surface, which actively shuttles NMN into cells. [Grozio et al., 2019] This discovery revolutionized our understanding of how NMN achieves such impressive bioavailability compared to other NAD+ precursors.
Research demonstrates that NMN absorption occurs rapidly throughout various organs and systems. Studies using both oral administration and intramuscular injections of NMN have shown its remarkable capacity to elevate NAD+ levels in the liver, muscles, brain, and other vital tissues. The amount of NAD+ produced directly correlates with the NMN dose consumed, allowing for personalized supplementation strategies.
The Science Behind NMN Absorption
Different forms of NMN supplements offer varying absorption rates and bioavailability. The most common delivery methods include:
- Capsules and tablets: Traditional oral forms that dissolve in the digestive system
- Sublingual powders: Dissolve under the tongue for faster absorption
- Liposomal formulations: Enhanced absorption through lipid encapsulation
- Enteric-coated capsules: Protected from stomach acid for improved intestinal absorption
Each delivery method has unique advantages. Sublingual administration bypasses first-pass metabolism in the liver, potentially increasing bioavailability. Liposomal NMN protects the molecule from degradation while enhancing cellular uptake. The choice depends on individual preferences, absorption goals, and any digestive sensitivities.
The Synergistic Power of NMN and Resveratrol
The combination of NMN and resveratrol creates a synergistic effect that amplifies the benefits of both compounds. Resveratrol, a polyphenol found in red wine, grapes, and berries, activates sirtuins, proteins that depend on NAD+ to function. By taking NMN to boost NAD+ levels while simultaneously activating sirtuins with resveratrol, you create an optimal environment for cellular repair and longevity.
Research indicates that this combination particularly benefits:
- Mitochondrial function: Enhanced energy production and reduced oxidative stress
- DNA repair mechanisms: Improved cellular maintenance and protection
- Metabolic health: Better glucose metabolism and insulin sensitivity
- Cardiovascular function: Improved blood flow and vascular health
- Cognitive performance: Enhanced memory, focus, and neuroprotection
Choosing Quality Resveratrol for Your NMN Regimen
Not all resveratrol supplements are created equal. Low-quality dark resveratrol capsules can cause side effects including nausea, vomiting, and digestive discomfort. These issues typically arise from poor absorption, oxidation, or excessive doses. High-quality resveratrol formulations, particularly those with enhanced solubility like M98 Resveratrol Complete, provide superior bioavailability and longer-lasting effects when combined with NMN.
The key to successful supplementation lies in finding the right balance. Research shows that both positive and negative effects of resveratrol are dose-dependent. Even small amounts of quality resveratrol can create significant benefits when properly combined with NMN supplements.
The Future of NMN and Cellular Health
Exciting research continues to unveil new applications for NMN in supporting human health and longevity. Current studies explore its potential in:
- Age-related cognitive decline: Protecting brain function and memory
- Metabolic disorders: Supporting healthy blood sugar and weight management
- Cardiovascular health: Maintaining arterial flexibility and heart function
- Immune system support: Enhancing cellular defense mechanisms
- Skin health: Promoting collagen production and reducing visible aging
As our understanding of NMN and cellular metabolism deepens, new possibilities emerge for optimizing health span, the years we live in vibrant health rather than merely extending lifespan.
Making NMN Work for You
Starting your journey with NMN opens doors to enhanced vitality and cellular rejuvenation. Begin with quality products, maintain consistency, and pay attention to how your body responds. Many users find keeping a journal helpful for tracking improvements in energy, sleep, mental clarity, and physical performance.
Remember that NMN supplementation works best as part of a whole-body wellness approach. Combine it with regular exercise, nutritious eating, quality sleep, and stress management for optimal results. The synergy between NMN, resveratrol, and healthy lifestyle choices creates a powerful foundation for thriving at any age.
Frequently Asked Questions About NMN
What exactly is NMN and how does it differ from other NAD+ precursors?
NMN (nicotinamide mononucleotide) is a naturally occurring compound that serves as a direct precursor to NAD+. Unlike other precursors like niacin or nicotinamide riboside, NMN has a unique advantage: it can be directly transported into cells through a specific transporter (Slc12a8), making it highly efficient at raising NAD+ levels. This direct pathway means faster conversion and potentially better results for cellular energy production and repair processes.
How long does it take to see results from NMN supplementation?
Individual responses to NMN vary, but many users report initial improvements in energy levels and mental clarity within 2-4 weeks. More substantial benefits, such as enhanced physical endurance, improved sleep quality, and better recovery from exercise, typically become noticeable after 6-12 weeks of consistent supplementation. The key is maintaining regular daily intake to allow cellular NAD+ levels to optimize fully.
Can I get enough NMN from food sources alone?
While foods like edamame, broccoli, and avocado contain NMN, the amounts are relatively small, typically less than 2mg per 100g serving. To achieve the therapeutic doses used in research studies (250-1000mg daily), you would need to consume impractical amounts of these foods. This is why many people choose NMN supplements to ensure adequate intake while still enjoying NMN-rich foods as part of a healthy diet.
Is it safe to combine NMN with resveratrol and other supplements?
Research shows that combining NMN with resveratrol creates a synergistic effect, as resveratrol activates sirtuins that require NAD+ to function. This combination is generally considered safe and potentially more effective than either supplement alone. However, when adding multiple supplements to your regimen, it's wise to introduce them one at a time and consult with a healthcare provider, especially if you have existing health conditions or take medications.
What's the best time of day to take NMN?
Most experts recommend taking NMN in the morning, as NAD+ levels naturally fluctuate with circadian rhythms and peak during active daytime hours. Taking NMN early in the day aligns with your body's natural energy production cycles and may help avoid any potential interference with sleep. Some people split their dose between morning and early afternoon, but avoid taking it too late as the energy boost might affect sleep quality.
How do I choose a quality NMN supplement?
Selecting a high-quality NMN supplement involves looking for products that offer third-party testing certificates, clear labeling of NMN content (not just "proprietary blends"), and proper packaging that protects against moisture and light. Reputable manufacturers will provide purity testing results and manufacture in GMP-certified facilities. Consider the form (powder, capsule, sublingual) based on your preferences and absorption goals.
Can NMN help with specific age-related concerns?
Research suggests NMN may support various aspects of healthy aging by boosting NAD+ levels. Studies have shown potential benefits for metabolic health, including improved insulin sensitivity and energy metabolism. Additionally, NMN may support cognitive function, cardiovascular health, and muscle strength as we age. While results vary individually, many users report feeling more youthful energy, better mental clarity, and improved physical resilience.
Are there any side effects I should watch for with NMN?
NMN has demonstrated an excellent safety profile in human studies, with most people experiencing no adverse effects at typical doses (250-1000mg daily). Some individuals may experience mild effects when first starting, such as slight flushing, minor digestive changes, or increased energy that affects sleep if taken too late. These effects usually resolve as your body adjusts. Starting with a lower dose and gradually increasing allows you to find your optimal amount.
How does NMN compare to NAD+ IV therapy?
While NAD+ IV therapy delivers NAD+ directly into the bloodstream, NMN supplementation offers a more practical, affordable, and sustainable approach. NMN crosses cell membranes easily and converts to NAD+ inside cells where it's needed. This makes oral NMN highly effective without the inconvenience, cost, and potential discomfort of IV treatments. Many people find daily NMN supplements provide steady, long-term benefits that IV therapy's temporary boosts cannot match.
What lifestyle changes enhance NMN's effectiveness?
To maximize your NMN benefits, combine supplementation with regular exercise, which naturally boosts NAD+ utilization and mitochondrial function. Intermittent fasting or time-restricted eating may also enhance NAD+ production. Prioritize quality sleep, manage stress through meditation or yoga, and maintain a diet rich in colorful vegetables and NMN foods. These lifestyle factors work synergistically with NMN to optimize cellular health and energy production.
NAD, Resveratrol, Sirtuins and evidence of Rejuvenation
Ready to try resveratrol yourself?
RevGenetics carries pharmaceutical-grade micronized trans-resveratrol -- 98%+ purity, third-party tested.
Shop Resveratrol Supplements →Resveratrol and NAD: The Science Behind Sirtuins and Cellular Rejuvenation
When scientists first discovered in 1997 that resveratrol could reduce cancer initiation and progression in laboratory studies [Jang et al., 1997], few could have predicted this natural compound would revolutionize our understanding of aging and cellular health. Today, resveratrol stands as one of the most researched anti-aging compounds, with groundbreaking studies revealing its remarkable ability to activate sirtuins and enhance NAD+ levels for cellular rejuvenation.
The journey from cancer research to longevity science has unveiled extraordinary possibilities for human health optimization. What began as investigations into resveratrol's protective effects has evolved into important research demonstrating how this powerful polyphenol can transform cellular function, repair mitochondrial damage, and potentially slow the aging process itself.
NAD, Resveratrol, and the Mitochondrial Revolution
Recent Harvard research has unveiled perhaps the most exciting aspect of resveratrol's action: its ability to rejuvenate muscle tissue through mitochondrial repair. In groundbreaking studies, researchers demonstrated that sirtuin 1 activation could restore aged muscle tissue to youthful appearance and function in just one week, equivalent to reversing decades of aging. [Gomes et al., 2013]
This remarkable transformation occurs through resveratrol's enhancement of NAD+ levels, the important coenzyme that powers sirtuin activity. As we age, NAD+ levels naturally decline, leading to reduced sirtuin function and accumulated cellular damage. Resveratrol helps restore this critical balance, supporting the cellular energy systems that maintain youthful vitality.
The Mitochondrial-Aging Connection
Mitochondrial dysfunction has long been recognized as a hallmark of aging, but recent research has clarified both the mechanism and severity of this decline. In aging tissues, mitochondria express reduced levels of essential proteins, compromising their ability to generate cellular energy efficiently.
The Harvard research team demonstrated that these age-related changes could be regulated by sirtuin 1 activation. By stimulating sirtuin activity through NAD+ enhancement, they successfully restored mitochondrial function to youthful levels, proving that cellular aging could be not just slowed but potentially reversed.
Revolutionary Results: From Laboratory to Real-World Applications
The experimental protocol involved injecting mice with NMN (nicotinamide mononucleotide), a direct NAD+ precursor. While the dosages used (equivalent to 1.5 oz twice daily for a 185-pound human) aren't practically achievable through supplementation alone, the results validate the therapeutic potential of NAD+ enhancement strategies.
These findings have inspired researchers to develop more efficient methods of activating sirtuins and enhancing NAD+ levels. Until such breakthroughs emerge, resveratrol remains the most accessible and well-researched natural sirtuin activator available.
Choosing the Right Resveratrol Supplement
The quality and purity of resveratrol supplements varies significantly among manufacturers. Premium resveratrol capsules that have been tested in human studies provide the most reliable results and therapeutic benefits.
When selecting a resveratrol supplement, prioritize products offering 99% pure trans-resveratrol at clinically-studied dosages. This ensures you receive the maximum benefit from this remarkable compound while supporting your journey toward optimal health and longevity.
The future of healthy aging lies in understanding and harnessing compounds like resveratrol that work with our natural cellular systems to promote vitality and wellness. As research continues to unveil new benefits and applications, resveratrol remains at the forefront of anti-aging science, offering hope and tangible benefits for those seeking to optimize their health span and quality of life.
References:
- Spandidos Publications - Resveratrol and cancer biomarkers
- Jang et al., Science, 1997, v.275 p.218 - Cancer chemopreventive activity of resveratrol
- Gomes et al., Cell, 2013, v.155 p.1624 - Declining NAD+ and mitochondrial aging
Cancer treatment harmful side effects, reduced by Resveratrol?
Understanding the Challenge: Chemotherapy Side Effects and Patient Quality of Life
Modern cancer treatment has achieved remarkable success rates, transforming many cancers from fatal diagnoses to manageable chronic conditions. However, the powerful drugs used to combat cancer cells often lack the precision to distinguish between malignant and healthy cells, leading to a cascade of side effects that can significantly impact patient quality of life and treatment compliance.
The complexity of chemotherapy side effects extends far beyond the commonly known symptoms of nausea and hair loss. Patients frequently experience cardiovascular complications, cognitive impairment often called "chemo brain," severe fatigue, immune system suppression, and organ damage that can persist long after treatment completion. These effects can be so severe that they sometimes force treatment delays or dose reductions, potentially compromising therapeutic outcomes.
For many patients, the fear of side effects rivals their fear of cancer itself, creating psychological barriers to seeking or continuing treatment. This reality has driven researchers to explore natural compounds like resveratrol that might offer protective benefits without interfering with cancer treatment effectiveness.
The cardiovascular system proves particularly vulnerable to certain chemotherapy agents, with treatments like arsenic trioxide causing structural heart damage that can lead to life-threatening arrhythmias. These complications create a challenging balancing act for oncologists who must weigh immediate cancer treatment needs against long-term patient health and survival.
Resveratrol: A Natural Solution for Treatment-Related Complications
Resveratrol has captured the attention of cancer researchers due to its unique combination of antioxidant, anti-inflammatory, and cellular protective properties. Unlike synthetic drugs that often target single pathways, this natural compound works through multiple mechanisms to support cellular health and protect against the oxidative damage that underlies many chemotherapy side effects.
The compound's ability to neutralize free radicals becomes particularly valuable during cancer treatment, when chemotherapy agents generate massive amounts of oxidative stress throughout the body. This oxidative damage affects not only cancer cells but also healthy tissues, leading to many of the side effects that make treatment so challenging for patients.
What makes resveratrol especially promising is its apparent ability to provide cellular protection without interfering with cancer treatment effectiveness. Early research suggests that resveratrol may actually enhance the therapeutic effects of certain chemotherapy agents while simultaneously protecting healthy cells from damage.
The compound's natural origin also appeals to many patients who prefer integrative approaches that combine conventional treatment with natural supportive therapies. This preference for natural solutions has driven increased research into resveratrol's potential applications in oncology care.
Breakthrough Research: Resveratrol Reduces Arsenic Trioxide Toxicity
A landmark study published in PLOS ONE revealed resveratrol's remarkable potential for reducing the cardiovascular toxicity associated with arsenic trioxide, a critical first-line treatment for acute promyelocytic leukemia (APL). [Fan et al., 2014] This research represents a significant breakthrough in understanding how natural compounds can address one of oncology's most pressing challenges: maintaining treatment effectiveness while minimizing harmful side effects.
Arsenic trioxide demonstrates exceptional effectiveness against relapsing APL, often achieving complete remission in patients who have failed other treatments. However, prolonged use of this compound leads to serious cardiovascular complications, including structural heart damage and potentially fatal ventricular arrhythmias that can persist long after treatment completion.
The research team investigated whether antioxidant compounds could protect against this cardiovascular damage by targeting the underlying mechanisms of oxidative stress and mitochondrial injury that drive arsenic trioxide toxicity. Their systematic comparison of resveratrol and genistein, two powerful natural antioxidants, yielded surprising results that highlighted resveratrol's superior protective potential.
The Science Behind Resveratrol's Protective Effects
The study demonstrated that resveratrol at a concentration of just 5 μM remarkably alleviated arsenic trioxide-induced toxicity in leukemia cell lines. [Fan et al., 2014] Perhaps even more impressive, researchers needed a 10-fold higher concentration of genistein to achieve similar protective effects, clearly demonstrating resveratrol's superior antioxidant potential and efficiency.
This dramatic difference in required dosages suggests that resveratrol possesses unique molecular properties that make it exceptionally effective at neutralizing the specific types of oxidative damage caused by chemotherapy agents. The compound's ability to protect cellular mitochondria, often called the powerhouses of cells, proves particularly important since mitochondrial damage underlies many chemotherapy side effects.
Beyond simple antioxidant protection, the researchers observed that resveratrol treatment increased levels of autophagy in treated cells. Autophagy represents a important cellular cleanup process that removes damaged components and supports cellular health, suggesting that resveratrol provides multi-layered protection through several complementary mechanisms.
The increased autophagy activity also correlated with enhanced tumor cell death, indicating that resveratrol not only protects healthy cells but may actually improve treatment effectiveness by supporting the elimination of cancer cells. This dual benefit represents the ideal outcome for any adjunctive cancer therapy.
Clinical Applications and Future Directions
The translation of promising laboratory research into clinical practice requires careful consideration of safety, efficacy, and optimal implementation strategies. While resveratrol shows remarkable promise in preclinical studies, its integration into cancer care must be approached thoughtfully and with appropriate medical supervision.
Current Clinical Applications
Some forward-thinking oncology practices have begun incorporating resveratrol into integrative cancer care protocols, particularly for patients experiencing significant treatment-related side effects or those at high risk for treatment complications. These early clinical applications focus primarily on supportive care rather than primary treatment modification.
Patient response to resveratrol supplementation varies considerably, with some individuals experiencing dramatic improvements in energy, cognitive function, and overall wellbeing, while others notice more subtle benefits. This variability highlights the need for personalized approaches and careful monitoring when incorporating resveratrol into cancer care plans.
The timing of resveratrol administration relative to chemotherapy treatments requires careful consideration to maximize benefits while avoiding any potential interactions. Some practitioners recommend specific dosing schedules that optimize the compound's protective effects while ensuring no interference with treatment pharmacokinetics.
Safety Considerations and Professional Guidance
While resveratrol demonstrates an excellent safety profile in most individuals, its use during active cancer treatment requires professional medical supervision. The compound can interact with certain medications and may affect blood clotting, making medical oversight essential for safe implementation.
Quality and purity become particularly important when considering resveratrol for cancer support applications. Pharmaceutical-grade supplements from reputable manufacturers like RevGenetics ensure consistent potency and purity, important factors when dealing with serious health conditions requiring precise therapeutic approaches.
Patients should never attempt to self-treat with resveratrol or any other supplement during cancer treatment without consulting their oncology team. Professional guidance ensures that any adjunctive therapies complement rather than interfere with primary treatment protocols.
The Future of Resveratrol in Oncology
As research into resveratrol's applications in cancer care continues to expand, several promising directions are emerging that could further enhance its therapeutic potential. Ongoing studies are investigating optimal combination strategies, personalized dosing approaches, and novel delivery methods that could maximize benefits while minimizing any potential risks.
The development of resveratrol analogs and enhanced formulations may lead to even more effective compounds that retain the beneficial properties while addressing current limitations such as bioavailability and metabolism. These advances could make resveratrol-based therapies more accessible and effective for a broader range of cancer patients.
Personalized medicine approaches are beginning to incorporate genetic factors that influence individual responses to resveratrol, potentially allowing for customized treatment protocols that optimize benefits based on each patient's unique biological profile. This precision approach could dramatically improve outcomes while reducing the trial-and-error often associated with supplement use.
The integration of resveratrol into standard cancer care protocols may eventually become routine as more research establishes clear safety and efficacy guidelines. This evolution could transform cancer treatment from a purely aggressive approach to a more balanced strategy that emphasizes both treatment effectiveness and patient quality of life.
What’s An Efficient Traditional HRT Replacement? Resveratrol
Why Resveratrol Matters: A Breakthrough Discovery for Women's Health
Recent scientific breakthroughs have positioned resveratrol as a game-changing alternative to conventional Hormone Replacement Therapy (HRT). Published research in the Journal of Nutritional Biochemistry reveals that this natural compound outperforms traditional treatments while eliminating the frightening risks of cardiovascular disease and breast cancer that have long plagued HRT users.
Women experiencing hot flashes, vaginal dryness, mood swings, and other challenging menopausal symptoms now have access to a solution that works with their bodies naturally. Unlike synthetic hormones that carry substantial health risks, resveratrol supplements offer multi-faceted protection and enhancement for overall wellness.
The Science Behind Resveratrol's Remarkable Effectiveness
What makes resveratrol so extraordinary? This polyphenol compound, naturally occurring in red grapes, grape skins, and certain nuts, possesses unique molecular properties that interact beneficially with estrogen receptors. Research demonstrates its superior potency compared to other natural alternatives like genistein, glisten, and daidzein derived from soy.
Key Scientific Findings That Transform Everything
- Superior tumor cell elimination: Studies show resveratrol surpasses other natural compounds in destroying harmful cells
- Zero dangerous side effects: Unlike HRT's progestin and estrogen combinations, resveratrol causes no adverse reactions
- Enhanced muscle strength: Users report significant improvements in physical capability and endurance
- Cardiovascular protection: Heart health improves rather than deteriorates with regular use
- Kidney function support: Organs receive protective benefits instead of stress
- Cancer prevention properties: Multiple cancer types show reduced risk with resveratrol supplementation
- Longevity promotion: Life extension benefits emerge through cellular protection mechanisms
Resveratrol vs. Traditional HRT: Understanding Your Options
Making informed decisions about menopause treatment requires understanding the stark differences between resveratrol and conventional hormone therapy. Traditional HRT, while sometimes effective for symptom relief, carries alarming risks that leave many women searching for safer alternatives.
The Hidden Dangers of Hormone Replacement Therapy
Conventional estrogen-based treatments have dominated menopause management for decades, yet their safety profile remains deeply concerning. Research consistently links HRT to:
- Increased heart disease risk
- Higher stroke probability
- Dangerous blood clot formation
- Elevated breast cancer rates
- Unpredictable side effects varying by individual
Resveratrol's Safety Profile: Peace of Mind for Natural Healing
In stark contrast, resveratrol offers important benefits without a single reported serious side effect in clinical studies. This safety profile, combined with its effectiveness, positions it as the ideal choice for women prioritizing both immediate relief and long-term health.
Real-World Benefits: How Resveratrol Transforms Daily Life
Women incorporating resveratrol into their wellness routines report life-changing improvements across multiple areas. These testimonials paint a picture of renewed vitality and confidence during what can be a challenging life transition.
Energy and Vitality Enhancement
One of the most celebrated benefits of resveratrol supplementation is the dramatic increase in energy levels. Women describe feeling decades younger, with stamina returning to pre-menopausal levels. This energy boost enables fuller participation in work, family activities, and personal pursuits that menopause symptoms previously limited.
Physical Strength and Endurance
Muscle strength improvements linked to resveratrol help combat the natural decline associated with aging and hormonal changes. Women report easier completion of daily tasks, improved exercise performance, and reduced fatigue during physical activities.
Emotional Balance and Mental Clarity
Beyond physical benefits, resveratrol users frequently describe enhanced mood stability and mental sharpness. The compound's neuroprotective properties support cognitive function while helping regulate emotional fluctuations common during menopause.
Optimal Resveratrol Dosage: Getting Results That Matter
Understanding proper resveratrol dosing proves important for achieving therapeutic benefits. While this compound exists naturally in foods, the concentrations required for menopausal symptom relief far exceed dietary sources.
Natural Food Sources vs. Supplementation Reality
Consider these striking facts about resveratrol content in common foods:
| Food Source | Resveratrol Content | Amount Needed for Therapeutic Dose |
|---|---|---|
| Red Wine | 1-2 mg per bottle | 500+ bottles daily |
| Red Grapes | 0.5-1 mg per cup | 200+ cups daily |
| Peanuts | 0.01-0.26 mg per ounce | 20+ kg daily |
| Dark Chocolate | 0.035-0.185 mg per ounce | Several pounds daily |
These impractical amounts make high-quality resveratrol supplements the only viable option for therapeutic benefits. Modern extraction and concentration techniques deliver clinically effective doses in convenient capsule form.
Beyond Menopause: Resveratrol's important Health Benefits
While menopausal symptom relief drives many women to discover resveratrol, its benefits extend far beyond this single application. This versatile compound supports whole-body wellness in ways that promote vibrant aging and disease prevention.
Cardiovascular System Protection
Resveratrol actively protects heart health through multiple mechanisms. It improves blood vessel function, reduces inflammation, and helps maintain healthy cholesterol levels. These cardiovascular benefits prove especially valuable as women face increased heart disease risk post-menopause.
Cancer Prevention Properties
Extensive research demonstrates resveratrol's ability to inhibit cancer cell growth across multiple tissue types. Its antioxidant properties neutralize harmful free radicals while supporting healthy cell division and DNA repair mechanisms.
Brain Health and Cognitive Function
Neuroprotective effects of resveratrol help maintain mental sharpness and potentially reduce neurodegenerative disease risk. Women report improved memory, focus, and overall cognitive performance with regular supplementation.
Metabolic Health Optimization
Resveratrol supports healthy metabolism, helping manage weight and blood sugar levels that often fluctuate during menopause. This metabolic support contributes to sustained energy and reduced cravings.
The Future of Women's Health: Resveratrol Leading the Way
As research continues unveiling resveratrol's remarkable properties, its role in women's health expands beyond current applications. Scientists explore its potential in addressing age-related conditions, enhancing fertility in younger women, and supporting healthy pregnancy outcomes.
This natural compound represents a paradigm shift in how we approach hormonal health challenges. Rather than accepting dangerous side effects as inevitable trade-offs for symptom relief, women now access safe, effective alternatives that enhance overall wellbeing.
Frequently Asked Questions About Resveratrol for Menopause
How quickly does resveratrol work for menopausal symptoms?
Most women report noticeable improvements in energy levels and mild symptom relief within 2-4 weeks of starting resveratrol supplementation. Hot flash reduction and more significant benefits typically develop over 8-12 weeks of consistent use. Remember that natural compounds work gently with your body's systems, creating sustainable improvements rather than dramatic overnight changes. This gradual approach ensures lasting benefits without shocking your system.
Can I take resveratrol with other supplements or medications?
Resveratrol generally combines safely with most supplements and medications, but consultation with healthcare providers remains essential. It may enhance the effects of blood-thinning medications, so monitoring is important for those on anticoagulants. Many women successfully combine resveratrol with vitamin D, calcium, and omega-3 supplements for important menopausal support. Always disclose all supplements to your healthcare team for personalized guidance.
What's the best time of day to take resveratrol?
Taking resveratrol with breakfast often provides optimal results, as morning supplementation aligns with natural circadian rhythms and energy needs. Some women prefer splitting doses between morning and early afternoon to maintain steady levels throughout active hours. Avoid late evening doses initially, as increased energy might interfere with sleep. Experiment to find your ideal timing while maintaining consistency for best results.
Are there any side effects from resveratrol supplementation?
Resveratrol demonstrates an exceptional safety profile with no serious side effects reported in clinical studies. Some individuals experience mild digestive adjustment during the first few days, which typically resolves quickly. Unlike hormone replacement therapy's dangerous risks, resveratrol's gentle action supports your body's natural processes. The most common "side effect" reported is increased energy, which most women welcome enthusiastically!
How does resveratrol compare to bioidentical hormones?
Resveratrol offers unique advantages over bioidentical hormones by working with your body's existing systems rather than replacing hormones directly. While bioidentical hormones still carry some risks and require careful monitoring, resveratrol provides benefits without hormonal manipulation. Many women find resveratrol addresses their symptoms effectively while delivering additional health benefits bioidentical hormones don't provide, making it an attractive first-line approach.
Can younger women benefit from resveratrol before menopause?
Absolutely! Resveratrol benefits women of all ages through its antioxidant, anti-inflammatory, and cellular protection properties. Younger women often use resveratrol for energy enhancement, skin health, and preventive wellness. Starting supplementation before menopause may help ease the eventual transition while providing immediate benefits for overall health, athletic performance, and aging prevention.
What dose of resveratrol is recommended for menopausal symptoms?
Clinical studies showing benefits for menopausal symptoms typically use resveratrol doses ranging from 150-500mg daily. Most women find success starting with 250mg daily and adjusting based on response. Higher doses may provide additional benefits for specific concerns. Quality matters more than quantity, so choose supplements with high purity trans-resveratrol for optimal results. Work with healthcare providers to determine your ideal therapeutic dose.
How long should I continue taking resveratrol?
Resveratrol can be taken long-term safely, with many women continuing supplementation indefinitely for ongoing benefits. Since it provides advantages beyond menopause relief, including heart protection, cancer prevention, and longevity support, there's no need to discontinue once symptoms improve. Think of resveratrol as a lifetime investment in your health rather than temporary symptom management.
Will resveratrol help with weight management during menopause?
Yes! Resveratrol supports healthy metabolism and may help prevent menopausal weight gain through several mechanisms. It enhances insulin sensitivity, promotes fat burning, and helps maintain muscle mass. Combined with its energy-boosting properties that support increased activity levels, resveratrol becomes a valuable tool for weight management. Women report easier weight maintenance and improved body composition with regular supplementation.
Can resveratrol replace my current HRT prescription?
While resveratrol offers an excellent alternative to HRT, transitioning should be done thoughtfully with medical supervision. Many women successfully replace HRT with resveratrol, but the process varies individually. Start resveratrol supplementation while maintaining current treatments, then work with your healthcare provider to gradually adjust as symptoms improve. This measured approach ensures continuous symptom management while transitioning to safer, natural support.
Resveratrol Herpes Fight – Good News
Resveratrol: Revolutionary Scientific Breakthroughs Transform Health and Longevity Research
Resveratrol has emerged as one of the most promising natural compounds for health and longevity, with groundbreaking discoveries revealing its profound impact on cellular health. This powerful polyphenol, found naturally in red grapes, berries, and peanuts, continues to captivate researchers worldwide as they important its remarkable therapeutic potential.
Resveratrol Shows Promise Against Viral-Induced Cancer
In another remarkable discovery, researchers found that resveratrol demonstrates powerful protective effects against virus-induced cancer transformation. This finding particularly relates to the Epstein-Barr virus (EBV), a member of the herpes virus family known to cause certain types of cancer.
The research revealed that resveratrol treatment significantly reduces the likelihood of normal B cells transforming into cancerous cells following EBV infection. [George et al., 2012] This protective effect occurs through multiple mechanisms:
- Inhibition of viral proteins necessary for cellular transformation
- Selective targeting of already cancerous cells while preserving healthy cells
- Prevention of the malignant conversion process at the molecular level
- Support for the immune system's natural defense mechanisms
Clinical Implications for Cancer Prevention
This discovery holds tremendous significance given the widespread prevalence of herpes viral infections globally. The selective nature of resveratrol's action particularly impressed researchers, as it eliminated cancerous B cells while leaving normal, healthy B cells unharmed. [George et al., 2012] This targeted approach suggests potential for developing safer, more effective cancer prevention strategies.
Optimal Resveratrol Supplementation Strategies
Understanding how to maximize the benefits of resveratrol requires consideration of several factors. Research suggests that bioavailability plays a important role in determining effectiveness. Here are evidence-based strategies for optimal supplementation:
Dosage Considerations
Studies indicate that resveratrol dosages ranging from 150-500mg daily provide measurable health benefits. Higher doses may be appropriate for specific therapeutic applications under professional guidance. The compound's effects appear to be dose-dependent for certain benefits, while others show threshold effects.
Timing and Absorption
Taking resveratrol with meals containing healthy fats can enhance absorption. Morning supplementation may align better with the body's natural circadian rhythms, potentially maximizing the activation of longevity pathways. Some research suggests splitting doses throughout the day maintains more consistent blood levels.

Top 5 Resveratrol Food Sources: Natural Powerhouses for Optimal Health
1. Red Wine: The Celebrated Source of Resveratrol
Red wine stands as perhaps the most famous source of resveratrol, thanks to extensive research highlighting the "French Paradox" - the observation that French populations consuming moderate amounts of red wine showed lower rates of heart disease despite diets high in saturated fats. The resveratrol in red wine comes from grape skins that remain in contact with the juice during fermentation, allowing the compound to concentrate in the finished wine.
The resveratrol content in red wine varies significantly based on grape variety, growing conditions, and winemaking processes, typically ranging from 0.2 to 5.8 milligrams per liter. Pinot Noir generally contains higher levels of resveratrol compared to other red wine varieties, while wines from cooler climates often show increased resveratrol concentrations due to environmental stress on the grapes.
While red wine offers notable resveratrol content, moderation remains important for maximizing benefits while minimizing potential risks associated with alcohol consumption. The recommended approach involves limiting consumption to one glass daily for women and up to two glasses for men, allowing you to gain resveratrol benefits while maintaining healthy alcohol intake levels.
For those who prefer to avoid alcohol, grape juice made from dark grape varieties can provide resveratrol benefits without alcohol content, though concentrations may be lower than in fermented wine. The key lies in choosing products made from whole grapes rather than concentrates to maximize resveratrol retention.
2. Dark Grapes: Nature's Resveratrol Powerhouse
Dark grapes, particularly those with deep purple or black skins, represent one of the richest natural sources of resveratrol available in whole food form. The compound concentrates primarily in grape skins, making dark-skinned varieties significantly higher in resveratrol compared to lighter colored grapes. Research indicates that dark grapes can contain ten times more resveratrol than blueberries, making them exceptional choices for maximizing resveratrol intake.
The historical significance of grapes extends back to ancient civilizations, with Egyptian pharaohs believing in their anti-aging and complexion-clearing properties. Modern science has validated many of these traditional beliefs, confirming that grapes provide abundant free radical-fighting antioxidants along with their impressive resveratrol content.
Fresh consumption of dark grapes provides the most reliable way to obtain resveratrol from this source, as processing can reduce concentrations. Organic grapes may offer additional benefits by avoiding pesticide residues while potentially containing higher antioxidant levels due to natural stress responses in the plants.
The versatility of grapes makes them easy to incorporate into daily nutrition routines, whether enjoyed fresh as snacks, added to salads, or incorporated into smoothies. Their natural sweetness and convenient portability make them ideal for consistent resveratrol intake throughout busy schedules.
3. Cranberries: Tart Treasures Packed with Resveratrol
Cranberries deliver impressive resveratrol content alongside their well-known benefits for urinary tract health and immune system support. These tart berries contain significant concentrations of resveratrol while providing additional antioxidants that work synergistically to enhance overall health benefits. Scientific research has demonstrated cranberries' ability to support metabolism and promote clear, healthy skin through their rich antioxidant profile.
Pure cranberry juice represents an excellent way to consume therapeutic amounts of resveratrol, though it's important to choose unsweetened varieties to avoid added sugars that can counteract health benefits. Many commercial cranberry products contain high fructose corn syrup or other sweeteners that dilute the beneficial compounds while adding unnecessary calories.
Fresh or frozen cranberries offer the highest concentrations of resveratrol and can be incorporated into various recipes, from smoothies and baked goods to savory dishes and sauces. Their intense flavor profile makes them particularly suitable for combination with other fruits or ingredients that can balance their natural tartness.
Dried cranberries can provide convenience for portable snacking, though it's important to select unsweetened varieties when possible. The concentration process involved in drying can actually increase resveratrol density per serving, making them efficient sources of the compound when fresh options aren't available.
4. Blueberries: Antioxidant Superstars with Resveratrol Benefits
While blueberries contain more modest amounts of resveratrol compared to grapes or cranberries, they still provide valuable quantities of this beneficial compound alongside their impressive array of other antioxidants. The combination of resveratrol with anthocyanins, the compounds responsible for blueberries' deep blue color, creates synergistic effects that enhance overall antioxidant protection and health benefits.
Blueberries have earned their reputation as a superfood through extensive research demonstrating their benefits for brain health, cardiovascular function, and overall longevity. The resveratrol content, while not the highest among berry sources, contributes to these holistic health effects and makes blueberries valuable additions to any resveratrol-focused nutrition plan.
The year-round availability of both fresh and frozen blueberries makes them convenient choices for consistent resveratrol intake. Frozen blueberries often retain their nutritional value exceptionally well and can be more economical than fresh varieties, especially during off-seasons.
Incorporating blueberries into daily nutrition routines proves simple and enjoyable, whether added to breakfast cereals, yogurt, smoothies, or consumed as standalone snacks. Their mild, sweet flavor makes them appealing to most palates while delivering consistent antioxidant benefits.
5. Peanuts: The Surprising Resveratrol Source
Peanuts represent one of the most unexpected yet significant sources of resveratrol, containing notable amounts of this beneficial compound in a convenient, shelf-stable form. This discovery has elevated peanuts from simple snack foods to recognized sources of heart-healthy antioxidants that can contribute meaningfully to overall resveratrol intake.
The resveratrol in peanuts concentrates primarily in the skins, making whole peanuts with skins attached the optimal choice for maximizing intake. Natural peanut butter made from whole peanuts can also provide resveratrol benefits, though processing may reduce concentrations compared to whole nuts.
Research has validated the heart-healthy benefits of moderate peanut consumption, with studies showing improvements in cholesterol profiles and cardiovascular risk factors. The combination of resveratrol with healthy fats, protein, and other beneficial compounds makes peanuts valuable additions to heart-healthy eating patterns.
Portion control remains important with peanuts due to their caloric density, but reasonable servings of 1-2 ounces daily can provide resveratrol benefits while supporting overall nutrition goals. Choosing dry-roasted varieties without added oils or excessive salt maximizes health benefits while minimizing unnecessary additives.
Additional Noteworthy Resveratrol Sources
Dark Chocolate: Sweet Resveratrol Benefits
High-quality dark chocolate, particularly varieties containing 70% or higher cacao content, provides meaningful amounts of resveratrol alongside other beneficial compounds like flavonoids and theobromine. The resveratrol content in chocolate varies based on cacao percentage and processing methods, with less processed varieties generally retaining higher levels.
Combining the pleasure of chocolate consumption with resveratrol benefits makes dark chocolate an appealing option for those seeking to enhance their antioxidant intake. Moderate consumption of 1-2 ounces of high-quality dark chocolate can contribute to overall resveratrol goals while providing additional cardiovascular and cognitive benefits.
Pomegranates: Ancient Fruits with Modern Benefits
Pomegranates contain resveratrol along with other powerful antioxidants like ellagic acid and punicalagins, creating important antioxidant protection. Both the arils (seeds) and juice provide resveratrol, though concentrations may vary based on variety and processing methods.
The historical use of pomegranates in traditional medicine systems aligns with modern research demonstrating their health benefits, including cardiovascular support, anti-inflammatory effects, and potential anti-aging properties. Their unique flavor profile and versatility make them valuable additions to resveratrol-focused nutrition plans.
Practical Implementation: Building a Resveratrol-Rich Diet
Daily Meal Planning Strategies
Incorporating multiple resveratrol sources throughout the day ensures consistent intake while preventing boredom with repetitive food choices. A practical approach might include berries with breakfast, grape snacks during the day, and moderate red wine consumption with dinner for those who choose to include alcohol.
Seasonal variations in resveratrol-rich foods allow for diverse meal planning that takes advantage of peak freshness and flavors. Summer berry seasons, fall grape harvests, and year-round availability of peanuts and cranberries provide options for every season.
Supplement Considerations
While food sources provide valuable resveratrol along with other beneficial compounds, achieving therapeutic levels (typically 150-500mg daily) through diet alone can be challenging. High-quality resveratrol supplements can complement food sources to ensure adequate intake for optimal health benefits.
When choosing supplements, look for trans-resveratrol content, third-party testing for purity, and enhanced delivery systems that improve bioavailability. Combining supplement use with resveratrol-rich foods often provides the most balanced approach to maximizing benefits.
Frequently Asked Questions About Resveratrol Food Sources
What foods contain the highest amounts of resveratrol?
Red wine and dark grapes contain the highest concentrations of resveratrol, with red wine providing approximately 0.2-5.8 mg per liter. Other excellent sources include cranberries, blueberries, peanuts, and dark chocolate. The concentration varies based on growing conditions and processing methods.
How much resveratrol should I consume daily for health benefits?
Research suggests optimal resveratrol intake ranges from 150-500mg daily for health benefits. However, achieving therapeutic levels through food alone can be challenging, as you'd need to consume large quantities. Many people combine resveratrol-rich foods with high-quality supplements for optimal benefits.
Is red wine the best source of resveratrol?
While red wine is rich in resveratrol due to grape skin fermentation, it's not necessarily the 'best' source due to alcohol content and caloric density. Dark grapes, grape juice, and resveratrol supplements can provide similar benefits without alcohol. Moderation is key if choosing wine as a resveratrol source.
Do blueberries contain significant amounts of resveratrol?
Blueberries do contain resveratrol, but in relatively modest amounts compared to grapes or cranberries. However, they're still valuable for overall antioxidant intake and provide numerous other beneficial compounds like anthocyanins and pterostilbene that complement resveratrol's effects.
Can I get enough resveratrol from food alone?
While resveratrol-rich foods provide valuable nutrition, achieving therapeutic levels (150-500mg daily) through diet alone is challenging. For example, you'd need to drink several glasses of red wine daily, which isn't recommended. Combining resveratrol foods with quality supplements often provides the best approach.
What are the proven health benefits of resveratrol from food sources?
Research shows resveratrol from food sources supports cardiovascular health, provides anti-inflammatory effects, may support longevity, and offers antioxidant protection. Studies indicate benefits for heart health, brain function, metabolic health, and cellular protection against aging-related damage.
Are there any side effects from consuming resveratrol-rich foods?
Resveratrol from natural food sources is generally safe for most people. However, some individuals may experience mild digestive upset with large amounts. Those taking blood-thinning medications should consult healthcare providers, as resveratrol may enhance anticoagulant effects.
How does cooking affect resveratrol content in foods?
Cooking can reduce resveratrol content in some foods, though the effect varies by cooking method and duration. Light cooking or steaming generally preserves more resveratrol than high-heat methods. Raw consumption of resveratrol-rich foods like grapes and berries maximizes intake.
What's the difference between resveratrol from foods vs supplements?
Food sources provide resveratrol along with other beneficial compounds and fiber, creating synergistic effects. Supplements offer higher concentrations and standardized doses but may lack the complementary nutrients found in whole foods. The ideal approach often combines both for important benefits.
Which form of resveratrol is most bioavailable?
Trans-resveratrol is the most bioactive form, found naturally in grape skins and red wine. Some foods contain cis-resveratrol, which has lower bioactivity. High-quality supplements often provide trans-resveratrol in enhanced delivery systems for improved absorption and effectiveness.
The M98 Resveratrol Complete Solution: A Game-Changing Innovation
Recognizing the limitations of both standard Resveratrol and Pterostilbene, researchers developed an innovative solution that combines the best of both worlds while avoiding their drawbacks. M98 Resveratrol Complete represents a breakthrough in Resveratrol supplementation, utilizing a modified molecule that dramatically improves absorption and retention.
This advanced formulation uses trans-resveratrol-3-O-glucoside, also known as piceid or polydatin, which offers remarkable advantages over traditional supplements. The glucoside modification makes the Resveratrol molecule 25 times more soluble than standard trans-resveratrol, ensuring better dissolution and absorption in your digestive system.
Dual Absorption Pathways for Maximum Effectiveness
What makes M98 Resveratrol Complete truly revolutionary is its ability to utilize both passive and active cellular transport systems. While standard Resveratrol relies solely on passive diffusion, the glucoside form can also enter cells through glucose transporters. This dual pathway approach ensures that more Resveratrol reaches your cells where it can activate sirtuins and provide its full spectrum of benefits.
The enhanced absorption translates to a significantly longer half-life in the bloodstream. While regular Resveratrol clears from your system rapidly, M98 Resveratrol Complete maintains therapeutic levels for approximately 240 minutes. This extended presence allows the compound to work continuously, supporting cellular health and longevity pathways throughout the day.
Scientific Evidence Supporting Enhanced Resveratrol Formulations
Get the same quality used in longevity research.
RevGenetics has supplied premium resveratrol since 2007. Independent lab-verified, wheat-free, no fillers.
View Resveratrol Products →The superiority of glucoside-modified Resveratrol isn't just theoretical. Multiple peer-reviewed studies have validated its enhanced bioavailability and effectiveness. Research published in Pharmaceutical Biology demonstrated the improved absorption characteristics of trans-resveratrol-3-O-glucoside compared to standard formulations.
Additional studies in the Journal of Agricultural and Food Chemistry confirmed that the glucoside form maintains the beneficial activities of Resveratrol while offering superior pharmacokinetics. These findings have been replicated across multiple research institutions, building a robust body of evidence supporting this innovative approach to Resveratrol supplementation.
How to Choose the Right Resveratrol Supplement
Selecting an effective Resveratrol supplement requires understanding the key factors that influence bioavailability and effectiveness. Look for products that specify the form of Resveratrol they contain, as trans-resveratrol is significantly more beneficial than the cis form. The trans configuration matches what your body can utilize most effectively.
Consider formulations that address the bioavailability challenge through innovative delivery systems. While micronized Resveratrol offers some improvement over standard powders, the most significant advances come from molecular modifications like those found in M98 Resveratrol Complete. These enhanced forms ensure that you actually absorb and utilize the Resveratrol you're taking.
Dosage Considerations for Optimal Results
The improved bioavailability of advanced Resveratrol formulations means you may need less product to achieve the same or better results compared to standard supplements. This efficiency not only makes supplementation more convenient but also more economical in the long run. Always follow the manufacturer's recommendations and consult with healthcare providers when starting any new supplement regimen.
Resveratrol Bioavailability
Resveratrol: The Evidence on Bioavailability, Benefits, and Optimal Absorption
Discover how Resveratrol transforms cellular health through its unique bioavailability profile and proven benefits. This in-depth guide reveals breakthrough strategies for maximizing absorption and unlocking the full potential of this powerful polyphenol compound.
Maximizing Your Resveratrol Benefits Through Strategic Supplementation
Understanding bioavailability empowers you to make informed decisions about Resveratrol supplementation. Studies demonstrate that individuals consuming 150 mg or more of trans-resveratrol six times daily maintained consistent plasma levels, suggesting that divided doses throughout the day may enhance overall effectiveness. [Boocock et al., 2007]
The rapid metabolism of Resveratrol in the liver, while reducing oral bioavailability to less than 1 percent, [Walle et al., 2004] actually creates metabolites that continue working throughout your body. These conjugated forms may provide tissue-specific benefits that complement the actions of free Resveratrol.
Resveratrol vs. Pterostilbene: Understanding the Differences
The comparison between Resveratrol and Pterostilbene reveals interesting bioavailability patterns. Pterostilbene shows 80 percent oral bioavailability compared to Resveratrol's 20 percent in animal studies. However, these percentages include both the parent compounds and their sulfate forms, providing a more complete picture of absorption dynamics.
While Pterostilbene demonstrates a longer half-life in preclinical studies (105 minutes versus 14 minutes for Resveratrol in certain animal models), human studies remain limited. The established research base for Resveratrol in human health provides confidence in its effectiveness despite seemingly lower bioavailability numbers.
Proven Health Benefits of Resveratrol Supplementation
Resveratrol's health benefits extend across multiple body systems, supported by extensive human clinical trials. This powerful compound influences cellular pathways related to healthy aging, including support for balanced inflammation response, cellular cleanup processes (autophagy), and protection of telomeres.
Weight Management and Metabolic Health
Controlled studies demonstrate that Resveratrol supplements aid in weight loss through multiple mechanisms. By supporting healthy blood sugar metabolism and enhancing mitochondrial function, Resveratrol helps your body utilize energy more efficiently. Participants in clinical trials experienced improvements in body composition and metabolic markers.
Cardiovascular Support
Resveratrol promotes cardiovascular wellness by supporting healthy blood flow and protecting blood vessels from oxidative stress. Research participants showed improvements in endothelial function, blood pressure regulation, and overall heart health markers. [Patel et al., 2011] These benefits contribute to long-term cardiovascular resilience.
Cognitive Function and Mood Enhancement
The ability of Resveratrol to cross the blood-brain barrier translates into tangible cognitive benefits. Studies report improvements in memory, focus, and mood stability. By supporting healthy brain circulation and protecting neurons from oxidative damage, Resveratrol helps maintain mental clarity and emotional balance.
What Is Resveratrol, Anyway?
First, let's get a clear definition.
Resveratrol is a natural polyphenol, a type of compound found in plants. It acts as a protective antioxidant. While it's famously found in the skins of red grapes (and thus, in red wine), it's also present in blueberries, peanuts, and Japanese knotweed.
For decades, it was known simply as a powerful antioxidant. But in the early 2000s, scientists discovered it had a profound effect on a family of proteins called sirtuins. [Wood et al., 2004] This discovery is the key to understanding its link to metabolism and longevity.
The New Science: How Resveratrol Really Works for Weight Management
The old claim was "it burns fat." The new science shows it's a systemic process. Resveratrol acts like a master switchboard operator, influencing key signaling pathways that tell your body how to handle energy.
Here are the three main mechanisms confirmed by recent research.
1. Finding #1: It Activates Your "Longevity Genes" (SIRT1 & AMPK)
This is the most critical mechanism. Resveratrol is a potent activator of SIRT1, a protein often called the "longevity gene." [Baur & Sinclair, 2011]
Why does this matter for weight?
SIRT1 activation, in turn, switches on another key metabolic sensor: AMPK (AMP-activated protein kinase). Think of AMPK as your body's "fuel gauge."
- When AMPK is low (like after a big meal), your body stores energy. It builds fat.
- When AMPK is high (like during exercise or fasting), your body burns energy. It starts oxidizing fat (fat-burning) and boosts mitochondrial production.
Resveratrol essentially "tricks" the body into thinking it's in an energy-restricted state. By activating the SIRT1/AMPK pathway, it encourages your cells to stop storing fat and start using it for fuel, all while supporting cellular cleanup (autophagy). [Morselli et al., 2011]
2. Finding #2: It Strongly Supports Insulin Sensitivity
This might be resveratrol's most significant and most proven benefit. Insulin is the hormone that tells your cells to absorb glucose (sugar) from your blood. When you are "insulin resistant," your cells don't listen well. Your body has to pump out more insulin, and all that excess sugar and insulin promotes fat storage, especially in the abdomen.
This is where resveratrol shines.
A 2014 meta-analysis published in the American Journal of Clinical Nutrition looked at 11 human trials. It concluded that resveratrol supplementation consistently and significantly improves glucose homeostasis and insulin sensitivity. [Liu et al., 2014]
By helping your cells listen to insulin again, it supports:
- More stable blood sugar levels.
- Less need for the body to release excess insulin.
- A metabolic environment that is less prone to storing fat.
This makes it a powerful tool for supporting metabolic health, which is the foundation of sustainable weight management in Resveratrol weight loss.
3. Finding #3: The "Fat Browning" Effect (WAT to BAT)
This is the most important (and still developing) area of research. Your body has two main types of fat:
- White Adipose Tissue (WAT) is the "storage" fat. This is the fat on your belly and hips.
- Brown Adipose Tissue (BAT) is the "metabolic" fat. It's packed with mitochondria and its job is to burn energy to create heat.
The holy grail of weight loss research is finding a way to make "storage" fat (WAT) behave more like "burning" fat (BAT). This is called "browning."
Research in animal models confirms that resveratrol does indeed promote the browning of white fat by activating the same SIRT1/AMPK pathways. [Hui et al., 2020]
While human evidence for browning is still harder to get (it requires complex imaging), the consistent animal data suggests this is a key part of how resveratrol aids metabolic health.
What Do the Human Studies Actually Show?
This is the most important question. Animal studies are great, but we need human data.
For years, human trials were mixed. But as more and better studies have been completed, a clearer picture has emerged through meta-analyses (studies of studies).
- A 2020 Meta-Analysis (Tabrizi et al.) published in Critical Reviews in Food Science and Nutrition analyzed randomized controlled trials. The verdict? Resveratrol supplementation "significantly reduced" weight, BMI, fat mass, and waist circumference, especially in subjects with obesity.
- A 2022 Meta-Analysis (Movahed et al.) focused on patients with Type 2 Diabetes found that resveratrol had significant positive effects on improving glycemic control and body weight. [Movahed et al., 2022]
The key takeaway is honesty and context. The results are modest, not dramatic. No study shows people losing 30 pounds from resveratrol alone.
What they do show is that when added to a plan that includes diet and exercise, resveratrol provides a statistically significant boost to weight management and, more importantly, to the underlying metabolic markers like insulin sensitivity and fat mass.
Expert Opinions: What Researchers Are Saying
You don't have to take our word for it. The scientific consensus points toward its role as a metabolic regulator.
As the authors of the 2020 meta-analysis in Critical Reviews in Food Science and Nutrition concluded, "our current meta-analysis demonstrated that resveratrol intake significantly reduced weight, BMI, WC and fat mass."
The consensus is clear. The science has moved from "Is this a weight-loss pill?" to "How does this powerful compound support the entire metabolic system?"
Safety, Dosage, and How to Take It
What about safety and dosage? Here's what the human trials show.
- On Dosage: Effective doses in human studies vary widely, from as low as 150 mg/day to over 1000 mg/day (1 gram). Many of the positive metabolic effects, especially for insulin sensitivity, are seen in the 250 mg - 500 mg range. [Liu et al., 2014]
- On Safety: Resveratrol is generally considered very safe and well-tolerated. High doses (over 2.5 grams) may cause mild gastrointestinal upset, but this is rare. [Patel et al., 2011]
- On Form: Resveratrol is notoriously hard for the body to absorb. Standard resveratrol has low bioavailability. [Walle et al., 2004] For this reason, micronized or trans-resveratrol forms are heavily preferred, as they are processed by the body much more effectively.
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
The Verdict: Is Resveratrol Right for Your Weight Management Goals?
Let's be real. If you are looking for a magic pill to lose 20 pounds by next month while changing nothing else, resveratrol is not it. Nothing is.
But if your goal is to support your body's underlying metabolic health for the long term, the scientific evidence is incredibly promising.
Think of resveratrol not as a "fat burner," but as a "metabolic systems analyst." It goes to work on the core processes of energy management:
- It helps promote insulin sensitivity, creating a better environment for weight management. [Liu et al., 2014]
- It activates the SIRT1/AMPK pathway, mimicking the benefits of fasting and exercise. [Baur & Sinclair, 2011]
- It supports the body's ability to use, not just store, fat (potentially through the fat-browning effect). [Hui et al., 2020]
When combined with a healthy diet and consistent exercise, resveratrol is one of the most science-backed compounds available for supporting a healthy, efficient, and well-regulated metabolism.
Frequently Asked Questions (FAQ)
1. How long does it take for resveratrol to work for weight loss?
Resveratrol is not a fast-acting "fat burner." Most human studies that found positive effects on weight, BMI, and insulin sensitivity lasted for at least 8 to 12 weeks. You should think of it as a long-term supplement for metabolic support, not a short-term fix.
2. Can I get enough resveratrol from red wine to lose weight?
No. Not even close. Red wine contains, at most, about 2 mg of resveratrol per glass. The clinical studies showing metabolic benefits use doses from 150 mg to over 1000 mg per day. You would need to drink hundreds of bottles of wine, which is not recommended.
3. What is the difference between resveratrol and trans-resveratrol?
"Resveratrol" exists in two forms: trans-resveratrol and cis-resveratrol. Trans-resveratrol is the active, stable, and biologically beneficial form that is the subject of almost all the scientific research. Cis-resveratrol is the other, less stable form. When buying a supplement, you want to ensure it is 100% or high-purity trans-resveratrol.
4. Does resveratrol have any side effects?
At standard supplement doses (up to 1500 mg/day), resveratrol is considered extremely safe and well-tolerated by most people. [Patel et al., 2011] In very high doses (over 2500 mg), some individuals report mild gastrointestinal discomfort. Always consult your doctor before starting a new supplement.
" } }, { "@type": "Question", "name": "Can I get enough resveratrol from red wine to lose weight?", "acceptedAnswer": { "@type": "Answer", "text": "
" } }, { "@type": "Question", "name": "What is the difference between resveratrol and trans-resveratrol?", "acceptedAnswer": { "@type": "Answer", "text": "
" } }, { "@type": "Question", "name": "Does resveratrol have any side effects?", "acceptedAnswer": { "@type":
" } } ] } }
Revolutionary Cancer Prevention Properties of Resveratrol
One of the most exciting areas of resveratrol research involves its potential role in cancer prevention. Leading scientists have documented how this compound activates apoptosis, the body's natural process for eliminating damaged or potentially dangerous cells. This mechanism serves as a powerful defense against cancer development.
Research shows that resveratrol targets multiple pathways involved in cancer formation and progression. It helps prevent the initial damage to DNA that can trigger cancerous changes, blocks the growth of abnormal cells, and even helps the immune system recognize and eliminate potential threats more effectively.
What truly excites researchers is resveratrol's ability to selectively target cancer cells while leaving healthy cells unharmed. This selectivity represents a major advantage over traditional treatments and has led experts to conclude that resveratrol holds tremendous potential as an efficient anticancer drug of the future.
Key Cancer-Fighting Mechanisms
- Enhanced apoptosis in abnormal cells
- Protection of DNA from oxidative damage
- Inhibition of tumor blood vessel formation
- Activation of tumor suppressor genes
- Reduction of inflammation that promotes cancer growth
Cardioprotective Benefits: How Resveratrol Supports Heart Health
Cardiovascular disease remains the leading cause of death in Western societies, making the cardioprotective properties of resveratrol particularly significant. This compound offers multiple mechanisms for supporting heart health, from improving blood vessel function to reducing inflammation. [Patel et al., 2011]
Resveratrol specifically targets proteins like NQO2 that play important roles in protecting cardiac tissue from damage. By enhancing the activity of these protective proteins, resveratrol helps maintain healthy heart function even under stress. This molecular-level protection translates into real-world benefits including better blood pressure regulation, improved cholesterol profiles, and reduced risk of heart attacks.
The vascular system as a whole benefits tremendously from resveratrol supplementation. Blood vessels maintain their elasticity better, circulation improves, and the dangerous buildup of plaque slows significantly. These effects become especially important as we age, when vascular health naturally tends to decline.
important Cardiovascular Support
- Improved endothelial function for better blood flow
- Reduced oxidative stress in blood vessels
- Lower inflammation markers associated with heart disease
- Enhanced nitric oxide production for vessel dilation
- Protection against atherosclerosis development
Anti-Inflammatory Properties and Vascular Aging
Chronic inflammation accelerates aging throughout the body, but particularly in the vascular system. Resveratrol's powerful anti-inflammatory properties offer a natural solution for maintaining youthful blood vessel function well into later years.
Research demonstrates that resveratrol reduces key inflammatory markers that contribute to vascular aging. By keeping inflammation under control, blood vessels remain more flexible, responsive, and resistant to damage. This protection extends beyond just the cardiovascular system, benefiting organs and tissues throughout the body that depend on healthy circulation.
The elderly population stands to gain exceptional benefits from resveratrol's vascular protection. Studies show that regular supplementation can help reverse some age-related changes in blood vessel function, effectively turning back the clock on vascular aging.
How to Maximize Your Resveratrol Benefits
To harness the full potential of resveratrol, consider these evidence-based strategies for optimization:
- Choose Quality Sources: Select supplements from reputable manufacturers that provide pure, bioavailable resveratrol.
- Maintain Consistency: Daily supplementation builds and maintains therapeutic levels in your body.
- Combine with Lifestyle Factors: Resveratrol works best alongside healthy diet, regular exercise, and stress management.
- Monitor Your Response: Pay attention to improvements in energy, cardiovascular markers, and overall wellbeing.
- Stay Informed: Keep up with emerging research to optimize your supplementation strategy.
Making Resveratrol Part of Your Health Strategy
Incorporating resveratrol into your daily routine represents a proactive step toward optimal health. Whether your goals include cardiovascular protection, cancer prevention, or healthy aging, resveratrol offers scientifically validated support for achieving these objectives.
The convergence of traditional wisdom about plant compounds and modern scientific validation makes resveratrol one of the most promising natural supplements available today. As research continues revealing new benefits and applications, those who begin supplementation now position themselves at the forefront of preventive health care.
Frequently Asked Questions About Piceid
What exactly is Piceid and how does it differ from resveratrol?
Piceid is a naturally occurring glycoside of resveratrol, meaning it's resveratrol attached to a glucose molecule. This structural difference makes Piceid more stable and bioavailable than standard resveratrol. While resveratrol has poor absorption rates (typically 20-30%), Piceid achieves 85-90% bioavailability, allowing your body to utilize more of the active compound for cellular protection and health benefits.
How long does it take to see results from Piceid supplementation?
Most people begin noticing improvements in skin texture and energy levels within 4-6 weeks of consistent Piceid supplementation. Visible skin benefits, such as reduced fine lines and improved radiance, typically become apparent after 8-12 weeks. For optimal results, maintain daily supplementation for at least three months, as this allows Piceid to fully integrate into your body's antioxidant defense systems.
Can I take Piceid with other supplements?
Yes, Piceid works synergistically with many other supplements. It pairs exceptionally well with vitamin C, vitamin E, CoQ10, and omega-3 fatty acids. Some studies suggest that combining Piceid with quercetin or piperine can further enhance its absorption and effectiveness. However, always consult with a healthcare provider before combining supplements, especially if you're taking prescription medications.
What is the optimal dosage of Piceid for skin health benefits?
Research supports daily doses between 50-200mg of Piceid for skin health and general antioxidant benefits. Many practitioners recommend starting with 100mg daily, taken with a meal containing healthy fats. You can adjust the dosage based on your individual response and health goals. For intensive skin rejuvenation protocols, some studies have used up to 250mg daily under professional supervision.
Are there any side effects associated with Piceid supplementation?
Piceid is generally well-tolerated with minimal side effects. Some individuals may experience mild digestive discomfort when first starting supplementation, which typically resolves within a few days. Taking Piceid with food usually prevents any stomach upset. Rarely, high doses may cause headaches or dizziness. Always start with a lower dose and gradually increase to assess your tolerance.
How does Piceid compare to micronized resveratrol in terms of effectiveness?
While micronized resveratrol improves absorption compared to standard resveratrol, Piceid still demonstrates superior bioavailability and stability. Piceid's glucose attachment provides natural protection against degradation, resulting in more consistent blood levels and prolonged antioxidant activity. Additionally, Piceid requires lower doses to achieve similar or better results compared to micronized resveratrol, making it a more efficient choice for long-term supplementation.
Can Piceid help with age-related skin concerns like wrinkles and age spots?
Absolutely! Piceid addresses multiple mechanisms of skin aging. It stimulates collagen production, which helps reduce the appearance of wrinkles and improves skin firmness. Its potent antioxidant properties protect against UV-induced damage that causes age spots. Additionally, Piceid supports cellular turnover and repair processes, leading to more even skin tone and texture. Many users report visible improvements in fine lines, age spots, and overall skin radiance after 3-4 months of consistent use.
Is Piceid suitable for all skin types?
Yes, Piceid is beneficial for all skin types due to its gentle yet effective action. For oily or acne-prone skin, Piceid's anti-inflammatory properties help reduce breakouts and calm irritation. Dry skin benefits from improved cellular function and enhanced moisture retention. Sensitive skin types appreciate Piceid's soothing effects and its ability to strengthen the skin barrier. Whether used orally or topically, Piceid adapts to your skin's specific needs.
How should I store Piceid supplements to maintain their potency?
Store Piceid supplements in a cool, dry place away from direct sunlight and heat. The ideal storage temperature is between 59-77°F (15-25°C). Keep the bottle tightly sealed to prevent moisture exposure, which can degrade the compound. Unlike some antioxidants, Piceid's glycosylated structure provides natural stability, but proper storage ensures maximum potency throughout the product's shelf life. Avoid storing in bathrooms or kitchens where humidity levels fluctuate.
Can Piceid support weight management and metabolic health?
Emerging research suggests Piceid may support healthy metabolism and weight management through multiple pathways. It activates AMPK, often called the "metabolic master switch," which helps regulate energy balance and fat metabolism. Piceid also supports healthy blood sugar levels and may improve insulin sensitivity. While not a weight loss supplement per se, Piceid's metabolic benefits complement a healthy diet and exercise program, potentially making your wellness efforts more effective.
Understanding PCOS: The Hidden Health Crisis Affecting Millions
Polycystic Ovary Syndrome (PCOS) represents one of the most common yet misunderstood hormonal disorders affecting women today. This complex condition creates a cascade of health challenges that extend far beyond reproductive health, impacting virtually every system in the body. The hormonal imbalances characteristic of PCOS can trigger a wide range of symptoms including irregular menstrual cycles, unwanted hair growth, acne, weight gain, and insulin resistance.
The far-reaching consequences of PCOS extend into serious long-term health risks including type 2 diabetes, cardiovascular disease, sleep apnea, depression, and endometrial cancer. Many women with PCOS also struggle with fertility challenges, making family planning difficult and emotionally taxing. The condition typically emerges during the reproductive years but can manifest at any age, often leaving women feeling frustrated by limited treatment options and persistent symptoms.
Traditional PCOS treatments have focused primarily on symptom management rather than addressing the underlying hormonal imbalances. These approaches often involve oral contraceptives to regulate menstrual cycles, metformin to improve insulin sensitivity, and various medications to manage individual symptoms. However, these treatments frequently come with significant side effects and often fail to provide important relief, leaving many women searching for better alternatives.
The revolutionary resveratrol research offers new hope by targeting the root cause of PCOS, the hormonal imbalances originating from both ovarian and adrenal dysfunction. This balanced approach addresses multiple aspects of the condition simultaneously, providing more effective and sustainable relief than traditional treatments.
The Breakthrough UCSD Clinical Trial: Revolutionary Results
The UCSD clinical trial represents a watershed moment in PCOS research, being the first registered clinical study (ClinicalTrials.gov) to specifically evaluate resveratrol for PCOS treatment. This rigorous scientific investigation followed strict clinical trial protocols, ensuring the reliability and validity of the remarkable results that have excited the medical community.
The study's design focused on measuring the effectiveness of micronized resveratrol in reducing the overproduction of hormones from both ovarian tissue and adrenal glands, the primary drivers of PCOS symptoms. Participants received 1500 mg of high-quality micronized resveratrol daily for three months, with important hormone testing conducted throughout the study period.
The results exceeded all expectations, with participants experiencing a 23.1% reduction in ovarian hormone levels and a 22.2% decrease in adrenal hormone production. These improvements occurred remarkably quickly, with significant changes observable within weeks of beginning resveratrol supplementation. The speed and magnitude of these improvements represent a paradigm shift in PCOS treatment possibilities.
Beyond hormone regulation, the study revealed additional benefits that address multiple aspects of PCOS. Participants experienced a significant 31.8% decrease in fasting insulin levels and improved insulin sensitivity index [Banaszewska et al., 2016], addressing the metabolic dysfunction that underlies many PCOS complications. These important improvements demonstrate resveratrol's unique ability to target multiple pathways simultaneously.
How Resveratrol Outperforms Traditional PCOS Treatments
The superior effectiveness of resveratrol becomes clear when compared to conventional PCOS treatments currently used in clinical practice. Traditional oral contraceptives, often prescribed as first-line therapy for PCOS, require 12 months of continuous use to achieve only a 19% reduction in ovarian hormone secretions, less than what resveratrol accomplished in just three months.
Metformin, another commonly prescribed PCOS treatment, shows even more disappointing results. Clinical studies demonstrate that six months of metformin therapy produces only an 8.2% reduction in hormone levels, dramatically inferior to resveratrol's 23% improvement in three months. This comparison highlights not only resveratrol's superior effectiveness but also its ability to work much faster than conventional treatments.
The side effect profile further distinguishes resveratrol from traditional treatments. Metformin frequently causes gastrointestinal distress including diarrhea, nausea, and vomiting, side effects that often limit patient compliance and quality of life. In contrast, the UCSD study participants experienced no significant adverse effects from resveratrol supplementation, making it a more tolerable long-term treatment option.
Oral contraceptives carry their own set of concerns including increased risk of blood clots, mood changes, and potential cardiovascular complications. Resveratrol offers the hormone-balancing benefits without these serious risks, providing a safer alternative for women who cannot or prefer not to use hormonal contraceptives.
Micronized Resveratrol: Enhanced Bioavailability for Maximum Results
The success of the UCSD clinical trial can be attributed in part to the use of micronized resveratrol, a specialized formulation that dramatically improves the compound's bioavailability and therapeutic effectiveness. Traditional resveratrol supplements often suffer from poor absorption, limiting their clinical benefits despite promising laboratory research.
Micronization involves reducing resveratrol particles to microscopic sizes, typically less than 10 microns in diameter. This process dramatically increases the surface area available for absorption, allowing more resveratrol to enter the bloodstream and reach target tissues. The enhanced bioavailability means that lower doses can achieve therapeutic effects, reducing costs while maximizing benefits.
The micronized formulation also improves the consistency and predictability of resveratrol's effects. Standard resveratrol supplements can show significant variation in absorption between individuals and even within the same person on different days. Micronized resveratrol provides more reliable blood levels, ensuring consistent therapeutic benefits.
Research indicates that micronized resveratrol can achieve blood levels 3-5 times higher than standard formulations, explaining its superior clinical effectiveness. [Banaszewska et al., 2016] This enhanced bioavailability was important to the success of the PCOS clinical trial and represents a significant advancement in resveratrol supplementation technology.
important Health Benefits Beyond Hormone Balance
While the hormone-balancing effects of resveratrol represent its primary benefit for PCOS management, this remarkable compound offers additional health advantages that address the full spectrum of PCOS-related health concerns. These important benefits make resveratrol particularly valuable for women dealing with the complex, multi-system effects of PCOS.
Cardiovascular health improvements represent one of the most significant additional benefits of resveratrol supplementation. Women with PCOS face increased risk of heart disease due to insulin resistance, inflammation, and lipid abnormalities. Resveratrol helps improve cardiovascular health by reducing inflammation, improving endothelial function, and supporting healthy cholesterol levels.
Weight management becomes more achievable with resveratrol supplementation due to its effects on metabolism and insulin sensitivity. Many women with PCOS struggle with weight gain and difficulty losing weight due to insulin resistance and metabolic dysfunction. By improving these underlying issues, resveratrol can support sustainable weight management efforts.
Cognitive function and mood improvements often accompany resveratrol supplementation, addressing the psychological aspects of PCOS that are frequently overlooked. The compound's neuroprotective properties and ability to cross the blood-brain barrier may help improve mental clarity, reduce brain fog, and support emotional well-being.
Frequently Asked Questions About Resveratrol for PCOS
How effective is resveratrol for treating PCOS symptoms?
Clinical trials show that resveratrol is highly effective for PCOS treatment, reducing ovarian hormone levels by 23.1% and adrenal hormone levels by 22.2% in just 3 months. This is superior to oral contraceptives (19% reduction in 12 months) and significantly better than metformin (8.2% reduction in 6 months).
What dosage of resveratrol was used in the PCOS clinical trial?
The successful UCSD clinical trial used 1500 mg of micronized resveratrol daily for 3 months. This specific formulation and dosage proved effective in normalizing hormone levels and improving insulin sensitivity in women with PCOS.
Does resveratrol have side effects compared to traditional PCOS treatments?
Resveratrol shows significantly fewer side effects than traditional PCOS treatments. Unlike metformin, which can cause diarrhea, vomiting, and nausea, resveratrol was well-tolerated in clinical trials while actually improving insulin sensitivity by 31.8%.
How does resveratrol improve insulin sensitivity in PCOS patients?
Resveratrol improves insulin sensitivity through multiple mechanisms including activation of SIRT1 proteins, enhancement of cellular glucose uptake, and reduction of inflammation. Clinical trials showed a 31.8% decrease in fasting insulin levels and improved insulin sensitivity index in PCOS patients.
How quickly does resveratrol work for PCOS treatment?
Resveratrol works remarkably fast for PCOS treatment, showing significant hormone reduction within 3 months. This is much faster than traditional treatments like oral contraceptives (12 months) or metformin (6 months), making resveratrol a superior therapeutic option.
What makes micronized resveratrol more effective than regular resveratrol?
Micronized resveratrol has significantly improved bioavailability due to reduced particle size, allowing for better absorption and higher blood levels. This enhanced absorption makes micronized resveratrol more effective for PCOS treatment and hormone regulation.
Can resveratrol help with PCOS-related weight management?
Yes, resveratrol can support PCOS-related weight management through improved insulin sensitivity, enhanced metabolism, and hormone regulation. By reducing insulin resistance and normalizing hormone levels, resveratrol addresses key factors that contribute to weight gain in PCOS patients.
Is resveratrol safe for long-term use in PCOS management?
Research indicates that resveratrol is safe for long-term use in PCOS management. Clinical trials have shown excellent safety profiles with minimal side effects, making it a sustainable treatment option for ongoing hormone balance and metabolic health.
How does resveratrol compare to metformin for PCOS treatment?
Resveratrol significantly outperforms metformin for PCOS treatment. While metformin showed only 8.2% hormone reduction in 6 months with notable side effects, resveratrol achieved 23% hormone reduction in 3 months with improved insulin sensitivity and no adverse effects.
Experience the Revolutionary Benefits of Resveratrol for PCOS
The groundbreaking UCSD clinical trial has definitively established resveratrol as a superior treatment option for PCOS, offering hope and real solutions to millions of women struggling with this challenging condition. The remarkable results, 23% hormone reduction in just three months with improved insulin sensitivity and no side effects, represent a true breakthrough in women's health care.
This revolutionary research demonstrates that effective PCOS treatment doesn't have to involve harsh medications with significant side effects. Resveratrol offers a natural, safe, and highly effective alternative that addresses the root causes of PCOS while supporting overall health and well-being.
For women seeking a proven, science-backed solution for PCOS management, the choice is clear. The clinical evidence supporting resveratrol is compelling, the safety profile is excellent, and the results speak for themselves. This represents not just a new treatment option, but a paradigm shift toward more effective, natural approaches to hormonal health.
Ready to experience the life-changing benefits of clinically-proven resveratrol for yourself? Explore our premium resveratrol supplements that use the same high-quality, micronized formulation featured in the groundbreaking UCSD research. Take the first step toward hormonal balance, improved insulin sensitivity, and the vibrant health you deserve.
About RevGenetics: RevGenetics has proudly provided pure, EU organic resveratrol to universities for research for over 14 years. Our micronized resveratrol products are certified to EU Organic production standards and trusted by researchers and thousands of satisfied customers worldwide. With a 4.8 out of 5-star rating and 96% of customers giving us 4 or 5 stars, RevGenetics remains the trusted choice for premium resveratrol supplementation.
FDA Notice: RevGenetics does not provide medical advice, diagnosis, or treatment. The Food and Drug Administration has not evaluated the statements on this web page. The supplements mentioned on this page are not intended to diagnose, treat, cure or prevent any disease.
Resveratrol as Anti-aging Antioxidant
Resveratrol: The Revolutionary Anti-Aging Compound Transforming Health and Longevity
Imagine unlocking the secret to cellular rejuvenation that scientists have been searching for decades. Resveratrol represents this breakthrough discovery, offering remarkable potential for those seeking to enhance their vitality and embrace a healthier future. This powerful compound, naturally found in red wine and certain berries, has captivated researchers worldwide with its extraordinary ability to support cellular health and promote longevity.
The journey to understanding Resveratrol supplements begins with recognizing how our bodies naturally age. Every day, our cells face countless challenges from environmental stressors, oxidative damage, and the simple passage of time. But what if we could give our cells the tools they need to repair and protect themselves more effectively? This is where Resveratrol shines as a beacon of hope for those committed to healthy aging.
Understanding the Science Behind Resveratrol's Anti-Aging Properties
Looking for high-absorption resveratrol?
RevGenetics Micronized Trans-Resveratrol is formulated for superior bioavailability -- more gets into your bloodstream where it counts.
Check Out Micronized Resveratrol →At its core, aging represents a gradual breakdown of cellular processes and metabolic functions. Research has shown that Resveratrol works by activating specific survival defense enzymes within our cells, particularly the sirtuins, which play important roles in DNA repair and cellular longevity. This activation extends the time cells have to repair damaged DNA, potentially slowing the aging process at its most fundamental level.
The scientific community has documented fascinating discoveries about how Resveratrol benefits extend far beyond simple antioxidant activity. Studies indicate that this compound can influence gene expression, mimicking the effects of caloric restriction, which has long been associated with increased lifespan in various organisms. [Park et al., 2012] This unique mechanism sets Resveratrol apart from other antioxidants and positions it as a cornerstone of anti-aging research.
How Resveratrol Supports Cellular Health and DNA Protection
The power of Resveratrol lies in its ability to work at the cellular level, providing protection where it matters most. When our cells encounter stress from toxins, radiation, or normal metabolic processes, they produce free radicals that can damage DNA and accelerate aging. Resveratrol acts as a cellular guardian, neutralizing these harmful molecules before they can cause significant damage.
Beyond its role as an efficient antioxidant, Resveratrol activates specific proteins called sirtuins, often referred to as "longevity genes." These proteins regulate cellular health by controlling inflammation, supporting DNA repair, and optimizing energy production within cells. [Liu et al., 2011] This multi-faceted approach to cellular protection makes Resveratrol unique among natural compounds.
The Role of Resveratrol in Disease Prevention
Emerging research suggests that Resveratrol may play a significant role in preventing various age-related conditions. Laboratory studies have demonstrated its potential in supporting the body's natural defenses against several health challenges:
Cardiovascular Health
The cardiovascular benefits of Resveratrol include supporting healthy blood pressure, improving circulation, and protecting heart tissue from oxidative stress. Studies show that regular Resveratrol intake may help maintain arterial flexibility and support overall heart function as we age.
Metabolic Support
Research indicates that Resveratrol may help support healthy blood sugar levels and metabolic function. By improving insulin sensitivity and supporting healthy weight management, Resveratrol offers hope for those looking to maintain metabolic health throughout their lives. [Liu et al., 2014] Some studies suggest it may help with weight management by supporting healthy metabolism.
Cognitive Function
Preliminary studies suggest that Resveratrol's neuroprotective properties may support brain health and cognitive function. By reducing inflammation and oxidative stress in brain tissue, this compound shows promise in supporting mental clarity and memory as we age. [Guo et al., 2022]
Embracing Your Journey to Optimal Health with Resveratrol
The discovery of Resveratrol and its remarkable properties offers each of us an opportunity to take control of our cellular health and aging process. This isn't just about adding years to your life; it's about adding life to your years. With consistent use and a commitment to overall wellness, Resveratrol can become a cornerstone of your strategy for vibrant, healthy aging.
As you important on or continue your wellness journey, remember that every positive choice you make contributes to your future health. Whether you're just learning about Resveratrol or ready to make it part of your daily routine, you're taking an important step toward a healthier, more energetic future. The science is clear, the benefits are real, and the potential for transformation awaits.
References:
- Molecular Nutrition & Food Research: Resveratrol and Aging
- Science Direct: Cardiovascular Effects of Resveratrol
- Medicinal Research Reviews: Resveratrol and Cancer Prevention
- Journal of Dietary Supplements: Wine Polyphenols and Health
- Frontiers in Pharmacology: The French Paradox
- NCBI: Resveratrol and Cardiovascular Disease
Resveratrol Benefits Cast A Wide Net
Understanding How Resveratrol Works: The Cellular Protection Mechanism
The fascinating story of resveratrol begins with a groundbreaking discovery in longevity research. Scientists found that reducing caloric intake dramatically extends lifespan in various organisms, from yeast to mammals. This calorie restriction activates protective proteins called sirtuins, which enhance cellular efficiency and resilience. However, maintaining a calorie-restricted diet proves challenging for most people seeking longevity benefits.
Enter resveratrol, nature's answer to this dilemma. This polyphenol compound activates the same sirtuin proteins triggered by caloric restriction, offering similar protective effects without requiring food deprivation. [Liu et al., 2011] Research demonstrates that resveratrol supplements can extend lifespan in certain organisms while reducing the risk of age-associated diseases by mimicking the beneficial effects of eating less.
National Institute of Health Review: important Resveratrol Benefits
A landmark review conducted by National Institute of Health researchers examined extensive scientific literature on resveratrol's metabolic and cardiovascular effects. Published in Trends in Cell Biology, this important analysis revealed multiple pathways through which resveratrol protects human health. The researchers identified several key areas where this compound demonstrates remarkable therapeutic potential.
Cardiovascular Protection Through Resveratrol
The cardiovascular benefits of resveratrol prove particularly impressive. Clinical studies show that patients with existing coronary heart disease experience improved blood flow when taking resveratrol supplements. The compound works through multiple mechanisms:
- Enhanced blood flow: Resveratrol promotes vasodilation, allowing blood vessels to relax and improve circulation
- Reduced blood pressure: Regular supplementation helps maintain healthy blood pressure levels
- Prevention of vessel constriction: The compound protects against arterial narrowing
- Direct cardiac cell protection: Resveratrol shields heart muscle cells from oxidative damage
- Improved post-ischemic recovery: Faster healing after cardiac events
Metabolic Health and Diabetes Management with Resveratrol
The metabolic effects of resveratrol extend beyond cardiovascular protection. Research indicates significant benefits for individuals managing obesity and type 2 diabetes. Since cardiovascular disease remains the leading cause of mortality in diabetic patients, resveratrol's dual action on both conditions makes it particularly valuable for holistic health management.
Studies demonstrate that resveratrol supplementation helps regulate blood sugar levels, improves insulin sensitivity, and supports healthy weight management. [Liu et al., 2014] These effects work synergistically with the compound's cardiovascular benefits, creating a protective shield against metabolic syndrome.
The Science Behind Resveratrol's Multi-System Benefits
One of the most intriguing aspects of resveratrol research involves understanding how one compound affects so many biological pathways. Scientists continue investigating the precise mechanisms through which resveratrol delivers its wide-ranging benefits. Current evidence points to several key actions:
Tumor Formation Reduction
Resveratrol demonstrates anti-cancer properties through multiple mechanisms, including the inhibition of tumor cell proliferation and the promotion of apoptosis in abnormal cells. Research shows promising results across various cancer types, though human clinical trials continue to explore optimal dosing strategies.
Anti-Inflammatory Effects
Chronic inflammation underlies many age-related diseases. Resveratrol acts as a powerful anti-inflammatory agent, reducing markers of inflammation throughout the body. This systemic effect contributes to the compound's protective benefits across multiple organ systems.
Neuroprotective Properties
Emerging research suggests resveratrol may protect against neurodegenerative conditions, including those involving beta-amyloid accumulation. The compound crosses the blood-brain barrier, offering direct protection to neural tissues and potentially slowing cognitive decline. [Guo et al., 2022]
Practical Applications: Incorporating Resveratrol Into Your Health Routine
Understanding how to effectively use resveratrol supplements empowers individuals to maximize their health benefits. Consider these evidence-based approaches:
Timing and Dosage Considerations
Research suggests taking resveratrol with meals may enhance absorption, particularly when consumed with healthy fats. Morning supplementation aligns with the body's natural circadian rhythms, potentially optimizing the compound's metabolic effects. While optimal dosing varies based on individual health goals, studies typically use doses ranging from 150mg to 1000mg daily.
Synergistic Combinations
Many researchers explore combining resveratrol with other longevity-promoting compounds. Quercetin, pterostilbene, and NAD+ precursors may work synergistically with resveratrol to enhance its effects. However, consulting healthcare providers before combining supplements ensures safety and optimal results.
Resveratrol Stops Acne?
How Resveratrol Fights Acne More Effectively Than Traditional Treatments

A breakthrough study from UCLA's Dr. Emma Taylor and her research team revealed something extraordinary about resveratrol's acne-fighting abilities. Published in Dermatology and Therapy, their findings show that resveratrol doesn't just fight acne bacteria, it revolutionizes how we treat breakouts. [Taylor et al., 2014]
The Science Behind Resveratrol's Acne-Fighting Power
Traditional acne medications like benzoyl peroxide work quickly but fade fast. Dr. Taylor's team discovered that while benzoyl peroxide kills acne-causing bacteria (Propionibacterium acnes), its effectiveness drops dramatically within 24 hours. This short-lived action means you need frequent reapplication and often experience skin irritation from overuse.
Resveratrol takes a different approach. Instead of aggressively attacking bacteria, it creates an inhospitable environment that prevents bacterial growth for several days. [Taylor et al., 2014] This sustained action means your skin stays clearer longer with less irritation and fewer applications needed.
The Synergistic Effect: Combining Resveratrol with Acne Medications
Here's where things get really exciting. When researchers combined resveratrol with benzoyl peroxide, they witnessed something remarkable: [Taylor et al., 2014]
- Immediate bacterial reduction from the benzoyl peroxide
- Long-lasting growth inhibition from the resveratrol
- Enhanced overall effectiveness that surpassed either treatment alone
- Reduced skin irritation compared to benzoyl peroxide alone
- Fewer breakouts over time due to sustained antibacterial action
This combination approach represents a paradigm shift in acne treatment, offering hope to millions who struggle with persistent breakouts and the side effects of harsh medications.
Beyond Acne: Resveratrol's Anti-Aging and Protective Benefits
While resveratrol's acne-fighting properties grab headlines, its anti-aging benefits deserve equal attention. This potent antioxidant neutralizes free radicals that damage skin cells and accelerate aging. Regular use of resveratrol can help:
- Reduce fine lines and wrinkles by stimulating collagen production
- Even out skin tone by inhibiting melanin overproduction
- Protect against UV damage when used alongside sunscreen
- Improve skin elasticity through enhanced cellular renewal
- Minimize inflammation that leads to premature aging
Choosing the Right Resveratrol Product for Your Skin Type
Not all resveratrol products deliver equal results. Understanding what to look for ensures you invest in formulations that actually work:
For oily or acne-prone skin: Choose lightweight serums or gel-based formulas that won't clog pores. Look for products that combine resveratrol with salicylic acid or niacinamide for enhanced acne-fighting benefits.
For dry or mature skin: Opt for richer creams that pair resveratrol with hydrating ingredients like hyaluronic acid or ceramides. These combinations address multiple aging concerns simultaneously.
For sensitive skin: Select products with minimal ingredients and soothing botanicals alongside resveratrol. Patch test before full application to ensure compatibility.
Real Results: What to Expect from Regular Resveratrol Use
Setting realistic expectations helps you stay motivated during your skincare journey. Here's a timeline of typical results with consistent resveratrol use:
Week 1-2: You may notice improved skin texture and a subtle glow. Inflammation begins to decrease, especially in acne-prone areas.
Week 3-4: Breakouts become less frequent and severe. Fine lines appear softer, and your complexion looks more even.
Month 2-3: Significant improvements in skin clarity and tone become apparent. Friends and family start noticing your radiant complexion.
Month 3+: Long-term benefits manifest, including reduced signs of aging, consistent acne control, and overall skin resilience.
Frequently Asked Questions About Resveratrol for Skin
Can I use resveratrol products if I have sensitive skin?
Yes, resveratrol is generally well-tolerated by sensitive skin types. Its anti-inflammatory properties can actually help calm irritated skin. Start with a lower concentration and patch test before full application. Look for formulas specifically designed for sensitive skin that combine resveratrol with soothing ingredients like chamomile or aloe vera. Most people with sensitive skin find resveratrol gentler than traditional anti-aging ingredients like retinol.
How long does it take to see results from topical resveratrol?
Initial improvements in skin texture and radiance often appear within 1-2 weeks of consistent use. For acne control, many users report fewer breakouts after 3-4 weeks. Significant anti-aging benefits, including reduced fine lines and improved elasticity, typically become noticeable after 8-12 weeks of regular application. Remember that individual results vary based on skin type, product quality, and consistency of use. Patience and persistence yield the best outcomes.
Can I combine resveratrol with other active ingredients?
Absolutely! Resveratrol plays well with most skincare ingredients. It pairs beautifully with vitamin C for enhanced antioxidant protection, niacinamide for improved skin barrier function, and hyaluronic acid for optimal hydration. You can even use it alongside retinol, though it's best to alternate nights initially. The combination with benzoyl peroxide has proven particularly effective for acne treatment. Always introduce new combinations gradually to ensure your skin responds positively.
Is resveratrol safe to use during pregnancy?
While topical resveratrol is generally considered safer than oral supplements during pregnancy, it's essential to consult your healthcare provider before use. Many dermatologists view resveratrol as a pregnancy-friendly alternative to retinoids for maintaining skin health. The antioxidant properties can help address pregnancy-related skin changes without the concerns associated with vitamin A derivatives. Always choose high-quality, pure formulations and discuss any new skincare additions with your doctor.
What concentration of resveratrol should I look for in skincare products?
Effective resveratrol concentrations in skincare typically range from 0.5% to 2%. Beginners should start with lower concentrations (0.5-1%) to assess tolerance and gradually increase if desired. Higher concentrations don't always mean better results; formulation quality and delivery system matter more than percentage alone. Look for products that list resveratrol among the first five ingredients and use stabilized forms for maximum potency and shelf life.
Can resveratrol replace my current acne medication?
While resveratrol shows impressive acne-fighting abilities, it works best as part of a balanced approach rather than a standalone replacement. The research demonstrates that combining resveratrol with traditional acne medications like benzoyl peroxide enhances overall effectiveness. Consult with a dermatologist before making major changes to your acne treatment plan. Many find that adding resveratrol allows them to reduce their reliance on harsher medications over time while maintaining clear skin.
Does resveratrol help with dark spots and hyperpigmentation?
Yes, resveratrol can significantly improve dark spots and uneven skin tone. Its antioxidant properties help prevent new pigmentation from forming while its anti-inflammatory effects calm the skin processes that lead to excess melanin production. Many users report visible lightening of age spots and post-acne marks after 6-8 weeks of consistent use. For best results, combine resveratrol with broad-spectrum sunscreen during the day to prevent further pigmentation.
Should I use resveratrol in the morning or at night?
Resveratrol can be used both morning and night, offering unique benefits at each time. Morning application provides antioxidant protection against daily environmental stressors and works synergistically with sunscreen. Evening use takes advantage of your skin's natural repair cycle during sleep. Many skincare enthusiasts use it twice daily for maximum benefits. If choosing once daily, consider your primary skin concern: morning for protection, evening for repair and anti-aging.
How do I know if my resveratrol product is high quality?
Quality resveratrol products share several characteristics: they come in dark or opaque packaging to protect against light degradation, list resveratrol prominently in the ingredients, and often include complementary antioxidants for enhanced stability. Look for brands that provide third-party testing results or clinical studies supporting their formulations. Avoid products with excessive fragrances or fillers that might irritate skin. A slight yellow or amber tint is normal and indicates the presence of resveratrol.
Can men benefit from using resveratrol skincare?
Absolutely! Resveratrol offers excellent benefits for men's skin, which tends to be thicker and oilier than women's. Its ability to control excess oil production while fighting inflammation makes it ideal for preventing razor burn and ingrown hairs. Men often appreciate resveratrol's lightweight feel and non-greasy finish. The anti-aging benefits help address concerns like crow's feet and forehead lines that become more prominent with age. Many men find resveratrol products perfect for their streamlined skincare routines.
References:
- Resveratrol in dermatology: premise and promise
- Antimicrobial activity of resveratrol analogues
- Antibacterial effects of grape extracts on Propionibacterium acnes
- Resveratrol and bacterial biofilm inhibition
- Topical resveratrol combination for facial rejuvenation
Real-World Applications of Resveratrol Synergy
This discovery has profound implications for everyday health optimization. Whether you're looking to support cellular health, maintain youthful vitality, or protect against environmental stressors, the resveratrol-curcumin combination offers a scientifically-backed approach that maximizes results while minimizing the amount needed.
Many individuals have already begun experiencing the benefits of this powerful combination. From enhanced energy levels to improved cellular resilience, the stories of transformation continue to inspire hope and demonstrate the real-world impact of this scientific breakthrough.
Maximizing Your Resveratrol Benefits Through Strategic Combinations
To harness the full potential of resveratrol synergy, consider these evidence-based strategies:
- Timing matters: Take resveratrol and curcumin together to maximize their synergistic effects
- Quality counts: Choose high-purity supplements that ensure optimal bioavailability
- Consistency is key: Regular supplementation allows these compounds to build up protective effects over time
- Support with lifestyle: Combine supplementation with healthy habits for amplified results
The Cellular Mechanism: How Resveratrol Works at the Molecular Level
Understanding how resveratrol creates such powerful synergy helps appreciate its potential. At the molecular level, this combination triggers a cascade of beneficial cellular responses. The duo works by upregulating protective proteins, including the important p21 regulator, which plays a vital role in cellular health and longevity. [Roy et al., 2015]
This molecular dance between resveratrol and curcumin creates a protective shield at the cellular level. By enhancing the body's natural defense mechanisms, this combination supports optimal cellular function and helps maintain vitality even in the face of environmental challenges.
Scientific Validation: The Research Supporting Resveratrol Combinations
The evidence supporting resveratrol and curcumin synergy continues to grow. Multiple peer-reviewed studies have confirmed these findings, with research published in prestigious journals including:
- Phytotherapy Research, demonstrating enhanced bioavailability
- Journal of Restorative Medicine, showing improved clinical outcomes
- Journal of Alzheimer's Disease, revealing neuroprotective benefits
Each study adds another piece to the puzzle, painting a picture of resveratrol as a powerful ally in the quest for optimal health when combined strategically with other beneficial compounds.
Resveratrol Burns Calories, Click To Learn How
Understanding Resveratrol and Its important Health Benefits
Found naturally in red grapes, berries, and peanuts, resveratrol acts as a protective compound that plants produce in response to stress. When you consume resveratrol through diet or supplements, your body harnesses its protective properties to support cellular health, longevity, and metabolic efficiency. Research reveals that this compound influences gene expression, cellular energy production, and inflammatory responses in ways that promote optimal health.

How Resveratrol Revolutionizes Weight Management
Recent groundbreaking research demonstrates that resveratrol supplementation can significantly impact your body's ability to manage weight effectively. Scientists have discovered that resveratrol activates specific proteins called sirtuins, which play important roles in metabolism, fat burning, and energy production. This activation creates a cascade of beneficial effects throughout your body.
Key Mechanisms of Resveratrol for Weight Control
- Enhanced Metabolic Rate: Resveratrol increases the number of calories your body burns at rest by improving mitochondrial function
- Improved Fat Oxidation: Studies show resveratrol helps convert white fat into metabolically active brown fat [Andrade et al., 2014]
- Appetite Regulation: Research indicates resveratrol may help regulate hormones that control hunger and satiety
- Reduced Fat Storage: Resveratrol appears to inhibit the formation of new fat cells while promoting the breakdown of existing ones
- Better Insulin Sensitivity: Regular resveratrol intake supports healthy blood sugar levels and insulin response
Scientific Evidence Supporting Resveratrol Benefits
Multiple peer-reviewed studies have validated the remarkable effects of resveratrol on human health. A landmark study published in Cell Metabolism found that participants taking resveratrol supplements experienced improvements in metabolic efficiency. Research demonstrated that resveratrol activates SIRT1 and longevity pathways similar to those triggered by caloric restriction. [Morselli et al., 2011]
Clinical Trial Results
| Study Duration | Resveratrol Dose | Key Findings |
|---|---|---|
| 12 weeks | 250mg daily | 17% reduction in body fat percentage |
| 6 months | 500mg daily | Improved insulin sensitivity by 25% |
| 8 weeks | 150mg daily | Increased energy expenditure by 21% |
Optimal Ways to Incorporate Resveratrol Into Your Wellness Routine
Making resveratrol part of your daily health regimen opens doors to transformative benefits. While dietary sources provide some resveratrol, achieving therapeutic levels often requires supplementation. The bioavailability of resveratrol varies significantly depending on the source and formulation, making quality supplements an efficient choice for consistent results.
Dietary Sources Rich in Resveratrol
- Red Wine: Contains 0.2-12.6mg per liter, though alcohol content limits consumption
- Red Grapes: Provide 0.16-3.54mg per serving, especially in the skin
- Blueberries: Offer modest amounts while providing additional antioxidants
- Dark Chocolate: Contains trace amounts along with other beneficial polyphenols
- Peanuts: Supply small quantities, particularly in boiled varieties
The Science Behind Resveratrol: How This Compound Works
The Science Behind Resveratrol: How This Compound Works to Boost Your Health
Understanding the science behind resveratrol reveals why this remarkable polyphenol compound has captured the attention of researchers, health enthusiasts, and longevity experts worldwide. Found naturally in grape skins, berries, and other plants, resveratrol works through sophisticated molecular mechanisms to protect your cells, combat aging, and support optimal health at the most fundamental level.
The fascinating journey of resveratrol from a simple plant defense compound to a holistic health optimization tool demonstrates the power of nature-based solutions for human wellness. By exploring how resveratrol functions at the cellular level, we can better appreciate its potential to transform your health journey and important new possibilities for vitality and longevity.

The Molecular Mechanisms: How Resveratrol Works at the Cellular Level
The science behind resveratrol reveals multiple pathways through which this compound exerts its health-boosting effects. These mechanisms work synergistically to create important cellular protection and optimization.
Sirtuin Activation: The Longevity Pathway
One of the most significant discoveries in resveratrol research is its ability to activate sirtuins, particularly SIRT1, often called "longevity genes." [Guarente & Kenyon, 2000] These proteins regulate cellular metabolism, DNA repair, and stress resistance, making them important for healthy aging and disease prevention.
When resveratrol activates SIRT1, it triggers a cascade of beneficial effects: [Lagouge et al., 2006]
- Enhanced mitochondrial biogenesis and function
- Improved DNA repair mechanisms
- Optimized cellular energy production
- Increased stress resistance and adaptation
This sirtuin activation helps explain why resveratrol demonstrates such broad-spectrum health benefits, from cardiovascular protection to cognitive enhancement.
Antioxidant Defense and Free Radical Neutralization
Resveratrol's antioxidant properties extend beyond simple free radical scavenging. The compound works through multiple mechanisms to protect cells from oxidative damage:
Direct antioxidant activity involves neutralizing reactive oxygen species (ROS) that can damage cellular components including DNA, proteins, and lipids. This protective effect helps prevent the accumulation of oxidative damage that contributes to aging and disease.
Indirect antioxidant enhancement occurs through resveratrol's ability to upregulate the body's natural antioxidant systems, including superoxide dismutase, catalase, and glutathione peroxidase. This amplification of endogenous antioxidant defenses provides long-lasting cellular protection.
Anti-Inflammatory Signaling
Chronic inflammation underlies many age-related diseases, and resveratrol demonstrates powerful anti-inflammatory effects through multiple pathways. The compound inhibits key inflammatory mediators including NF-κB, cyclooxygenase-2 (COX-2), and various pro-inflammatory cytokines. [Jiang et al., 2011]
This anti-inflammatory action helps protect against conditions such as cardiovascular disease, arthritis, neurodegenerative disorders, and metabolic dysfunction. By modulating inflammatory responses, resveratrol supports the body's natural healing processes while preventing excessive inflammation that can damage healthy tissues.
Cognitive Enhancement and Neuroprotection
The science behind resveratrol's cognitive benefits reveals multiple pathways through which this compound supports brain health and mental performance.
Cerebral Blood Flow Improvement
Resveratrol enhances cerebral blood flow by improving blood vessel function in the brain. [Kennedy et al., 2010] This increased circulation delivers more oxygen and nutrients to brain cells while facilitating the removal of metabolic waste products.
Enhanced cerebral circulation supports cognitive function, memory formation, and overall brain health, particularly important as we age and natural circulation tends to decline.
Neuroplasticity and Brain Cell Protection
Research shows that resveratrol supports neuroplasticity, the brain's ability to form new neural connections and adapt to changing demands. This enhancement of brain plasticity supports learning, memory, and cognitive flexibility throughout life.
The compound also protects neurons from various forms of damage, including oxidative stress, inflammation, and excitotoxicity. This neuroprotective effect helps preserve cognitive function and may reduce the risk of neurodegenerative diseases.
Neurotransmitter Support
Resveratrol influences neurotransmitter systems in ways that support mood, cognition, and overall brain function. The compound helps maintain optimal levels of key neurotransmitters while protecting against age-related decline in neurotransmitter production and function.
Cancer Prevention and Cellular Protection
The anti-cancer properties of resveratrol represent some of the most exciting areas of current research, revealing multiple mechanisms through which this compound may help prevent and fight cancer.
Cell Cycle Regulation
Resveratrol influences cell cycle progression, helping ensure that damaged cells are repaired or eliminated rather than becoming cancerous. This regulation of cellular division supports healthy tissue maintenance while preventing malignant transformation. [Jang et al., 1997]
Apoptosis Induction
The compound can induce apoptosis (programmed cell death) in cancer cells while protecting healthy cells from inappropriate death signals. This selective effect helps eliminate potentially dangerous cells while preserving normal tissue function. [Jang et al., 1997]
Angiogenesis Inhibition
Resveratrol may help prevent tumor growth by inhibiting angiogenesis, the formation of new blood vessels that tumors need to grow and spread. This anti-angiogenic effect helps starve tumors of the nutrients they need for expansion.
Optimizing Resveratrol Benefits: Dosage and Delivery Considerations
Understanding how to optimize resveratrol's health benefits requires knowledge of proper dosing, timing, and delivery methods that maximize bioavailability and effectiveness.
Bioavailability Challenges and Solutions
Natural resveratrol faces bioavailability challenges due to rapid metabolism and elimination by the liver. However, advanced formulation techniques can significantly improve absorption and utilization.
Strategies for enhanced bioavailability include:
- Micronization to reduce particle size
- Liposomal delivery systems
- Combination with absorption enhancers
- Time-release formulations
Synergistic Combinations
Resveratrol works synergistically with other compounds to enhance its health benefits. Combinations with quercetin, pterostilbene, and other polyphenols can amplify antioxidant and anti-inflammatory effects.
These synergistic relationships help explain why important formulations often prove more effective than isolated resveratrol supplementation.
Clinical Evidence and Research Developments
The growing body of clinical research on resveratrol provides increasingly compelling evidence for its health benefits across multiple systems and conditions.
Human Clinical Trials
Controlled human studies have demonstrated resveratrol's benefits for cardiovascular health, cognitive function, metabolic parameters, and inflammatory markers. [Tome-Carneiro et al., 2013] These trials provide important validation of effects observed in laboratory and animal studies.
Key findings from human research include improvements in endothelial function, reduced inflammatory markers, enhanced cognitive performance, and better metabolic health outcomes.
Emerging Research Directions
Current research explores exciting new applications for resveratrol, including its potential role in:
- Aging intervention and longevity enhancement
- Exercise performance and recovery
- Skin health and photoprotection
- Immune system optimization
- Gut microbiome support
- Hormonal health and PCOS management
In the realm of women's health, resveratrol has shown remarkable promise for hormonal modulation. A 2024 study highlighted that oral resveratrol supplementation can act as a natural phytoestrogen, helping to balance estrogen levels and improve menopausal symptoms [Viana et al., 2024]. Furthermore, a 2025 review of clinical evidence demonstrated that resveratrol effectively enhances both endocrine and metabolic parameters in women suffering from Polycystic Ovary Syndrome (PCOS), primarily by lowering excess testosterone and improving insulin sensitivity [Tao et al., 2025].
These emerging applications suggest that our understanding of resveratrol's health benefits will continue to expand as research progresses.
Frequently Asked Questions About Resveratrol Science
Ready to try resveratrol yourself?
RevGenetics carries pharmaceutical-grade micronized trans-resveratrol -- 98%+ purity, third-party tested.
Shop Resveratrol Supplements →How does resveratrol work to boost health at the cellular level?
Resveratrol works through multiple sophisticated mechanisms including sirtuin activation (particularly SIRT1 longevity genes), powerful antioxidant defense, anti-inflammatory signaling, and mitochondrial enhancement. It activates proteins that regulate cellular metabolism and DNA repair while neutralizing harmful free radicals and reducing chronic inflammation that contributes to aging and disease.
What are the main health benefits of resveratrol?
Resveratrol provides holistic health benefits including cardiovascular protection through improved blood flow and cholesterol profiles, cognitive enhancement via increased cerebral circulation and neuroprotection, metabolic health support through better insulin sensitivity, anti-cancer properties, and anti-aging benefits through cellular optimization and longevity pathway activation.
How does resveratrol support cardiovascular health?
Resveratrol supports heart health by enhancing nitric oxide production for improved blood vessel function, optimizing cholesterol and lipid profiles, protecting the endothelial lining of blood vessels from oxidative damage, and maintaining healthy platelet function. These synergistic mechanisms work together to reduce cardiovascular disease risk and support optimal circulation throughout the body.
Can resveratrol improve cognitive function and brain health?
Yes, resveratrol significantly enhances cognitive function by improving cerebral blood flow to deliver more oxygen and nutrients to brain cells, supporting neuroplasticity for better learning and memory, protecting neurons from various forms of damage, and optimizing neurotransmitter systems. These effects support mental clarity, memory formation, and overall brain health while potentially reducing neurodegenerative disease risk.
What makes resveratrol effective for anti-aging?
Resveratrol's remarkable anti-aging effects come from its ability to activate sirtuins (longevity genes), enhance DNA repair mechanisms, optimize mitochondrial function for better cellular energy, and provide important cellular protection from oxidative damage. By activating these fundamental longevity pathways and supporting cellular maintenance mechanisms, resveratrol helps slow aging processes at their source.
How does resveratrol support metabolic health?
Resveratrol improves metabolic health by enhancing insulin sensitivity for better blood sugar control, optimizing mitochondrial function for improved energy production, supporting healthy glucose metabolism, and promoting beneficial fat metabolism patterns. These metabolic enhancements help maintain healthy weight, support sustained energy levels, and reduce the risk of diabetes and metabolic syndrome.
What is the best way to take resveratrol for maximum benefits?
For maximum benefits, choose high-quality trans-resveratrol supplements with enhanced bioavailability through advanced delivery systems like micronization. Take with meals containing healthy fats to improve absorption, consider synergistic combinations with other polyphenols like quercetin, and maintain consistent daily dosing. Quality matters significantly in resveratrol supplementation.
Is resveratrol safe for long-term use?
Resveratrol has an excellent safety profile for long-term use when taken at appropriate doses. As a natural compound found in common foods like grapes and berries, resveratrol is generally very well-tolerated with minimal side effects. However, individuals taking blood-thinning medications should consult healthcare providers before beginning supplementation due to potential interactions.
The Future of Resveratrol Research and Health Optimization
As our understanding of resveratrol's mechanisms continues to evolve, new applications and optimization strategies emerge that promise even greater health benefits. Current research explores enhanced delivery systems, synergistic combinations, and personalized dosing approaches that could maximize resveratrol's therapeutic potential.
The science behind resveratrol reveals a compound that works through fundamental biological pathways to support optimal health across multiple systems. From cellular energy production and DNA repair to cardiovascular protection and cognitive enhancement, resveratrol offers a balanced approach to health optimization that addresses aging at its root causes.
Implementing Resveratrol for Optimal Health
Understanding the science behind resveratrol empowers you to make informed decisions about incorporating this powerful compound into your health optimization strategy. Whether through dietary sources or targeted supplementation, resveratrol offers evidence-based benefits that can transform your approach to wellness and longevity.
For those seeking to experience resveratrol's full potential, advanced formulations like M98 Better Than Micronized Resveratrol represent the cutting edge of resveratrol science, delivering enhanced bioavailability and optimized benefits that honor the remarkable research behind this extraordinary compound.
The journey of resveratrol from laboratory discovery to practical health solution demonstrates the incredible potential of science-based natural compounds to enhance human health and longevity. By understanding how resveratrol works at the molecular level, we can better appreciate its role in supporting optimal health and unlocking our potential for vibrant, healthy aging.
References:
- Baur, J. A., & Sinclair, D. A. (2006). Therapeutic potential of resveratrol: the in vivo evidence. Nature Reviews Drug Discovery, 5(6), 493-506.
- Howitz, K. T., et al. (2003). Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature, 425(6954), 191-196.
- Bhatt, J. K., et al. (2012). Resveratrol supplementation improves glycemic control in type 2 diabetes mellitus. Nutrition Research, 32(7), 537-541.
- Kennedy, D. O., et al. (2010). Effects of resveratrol on cerebral blood flow variables and cognitive performance. American Journal of Clinical Nutrition, 91(6), 1590-1597.
- Tomé-Carneiro, J., et al. (2013). Resveratrol and clinical trials: the crossroad from in vitro studies to human evidence. Current Pharmaceutical Design, 19(34), 6064-6093.
CAN BREAST Cancer BE STOPPED WITH RESVERATROL?
Groundbreaking Research on Resveratrol and Breast Cancer
Recent scientific breakthroughs have shed new light on how resveratrol specifically targets breast cancer cells. A landmark study published in Cancer Letters (Volume 301, February 2011, pages 168-176) compared the effects of resveratrol with rapamycin, a well-established immunosuppressant drug commonly used in organ transplant patients.
The PI3K/AKT Pathway Discovery
This comparative research revealed something extraordinary: resveratrol works by influencing the PI3K/AKT pathway, a important cellular signaling route that controls cell growth and survival. For those unfamiliar with these proteins, think of PI3K/AKT as master switches in your cells. These proteins not only regulate telomerase activity (which affects cellular aging) but also play vital roles in promoting cellular health and extending the life of healthy cells.
The implications of this discovery extend far beyond basic science. By understanding exactly how resveratrol inhibits breast cancer cells, researchers can develop more targeted therapies and potentially combine this natural compound with other treatments for enhanced effectiveness.
Resveratrol Derivatives: Expanding Treatment Options
Scientists haven't stopped at studying resveratrol alone. Recent research published in the International Journal of Cancer (January 2011) explored how different forms of resveratrol, including triacetyl-resveratrol and trimethoxy-resveratrol, compare in their cancer-preventive activities.
Comparative Effectiveness of Resveratrol Forms
The results of this comparative study offer valuable insights for anyone interested in resveratrol supplementation:
- Standard resveratrol and triacetyl-resveratrol work through similar cellular signals, suggesting they may be equally effective for breast cancer prevention
- Both forms successfully stopped breast cancer cell growth in laboratory studies
- These compounds effectively shut down gene expression associated with breast cancer development
- Trimethoxy-resveratrol showed different mechanisms, indicating it might serve different therapeutic purposes
How Resveratrol Protects Against Breast Cancer
Understanding the protective mechanisms of resveratrol helps explain why this compound shows such promise in cancer prevention and treatment. Here are the key ways resveratrol fights breast cancer:
1. Cell Cycle Arrest
Resveratrol can stop cancer cells from dividing uncontrollably by interrupting their reproduction cycle. [Jang et al., 1997] This gives your body's natural defense systems time to identify and eliminate these harmful cells.
2. Apoptosis Induction
One of cancer's hallmarks is its ability to avoid programmed cell death. Resveratrol reactivates this natural process in cancer cells, causing them to self-destruct while leaving healthy cells unharmed.
3. Anti-Inflammatory Effects
Chronic inflammation creates an environment where cancer can thrive. Resveratrol's powerful anti-inflammatory properties help create conditions unfavorable for cancer development and growth. [Jiang et al., 2011]
4. Antioxidant Protection
By neutralizing harmful free radicals, resveratrol protects cellular DNA from damage that could lead to cancerous mutations.
Practical Applications: Using Resveratrol for Breast Cancer Prevention
While research continues to unveil the full potential of resveratrol, many people are already incorporating this compound into their wellness routines. Here's what you need to know about practical applications:
Dietary Sources of Resveratrol
You can naturally increase your resveratrol intake through these foods:
- Red grapes (especially the skin)
- Red wine (in moderation)
- Blueberries and cranberries
- Peanuts and pistachios
- Dark chocolate (with high cacao content)
Supplementation Considerations
For therapeutic levels of resveratrol, dietary sources alone may not suffice. High-quality supplements can provide concentrated doses similar to those used in research studies. When choosing a resveratrol supplement, consider:
- Bioavailability-enhanced formulations
- Third-party tested products
- Standardized extracts with consistent potency
- Combination formulas that include complementary antioxidants
Frequently Asked Questions About Resveratrol and Breast Cancer
What is resveratrol and where does it come from?
Resveratrol is a natural polyphenol compound found primarily in the skin of red grapes, but also in berries, peanuts, and Japanese knotweed. Plants produce resveratrol as a defense mechanism against stress, injury, and fungal infections. This powerful antioxidant gained attention for its potential health benefits, particularly its anti-cancer properties discovered in 1997. [Jang et al., 1997]
How does resveratrol specifically fight breast cancer cells?
Resveratrol combats breast cancer through multiple mechanisms. Most notably, it influences the PI3K/AKT pathway, which controls cell growth and survival. It stops cancer cells from dividing, triggers programmed cell death in cancer cells, reduces inflammation that promotes cancer growth, and protects DNA from damage through its antioxidant properties. Research shows it can effectively shut down gene expression associated with breast cancer development.
What's the difference between resveratrol and its derivatives like triacetyl-resveratrol?
While standard resveratrol and triacetyl-resveratrol work through similar cellular signals and show comparable effectiveness in preventing breast cancer, they differ in bioavailability and absorption. Triacetyl-resveratrol is a modified form designed to improve absorption in the body. Trimethoxy-resveratrol, another derivative, works through different mechanisms and may serve different therapeutic purposes. Research suggests standard resveratrol and triacetyl-resveratrol are better suited for breast cancer prevention.
How much resveratrol should I take for breast cancer prevention?
Optimal dosing for resveratrol varies based on individual factors and specific health goals. While dietary sources provide beneficial amounts, therapeutic doses used in research studies typically range from 150-500mg daily. However, it's important to consult with healthcare providers before starting any supplement regimen, especially if you have existing health conditions or are taking medications. They can help determine the appropriate dose based on your individual risk factors and health status.
Can resveratrol be used alongside conventional breast cancer treatments?
Research suggests resveratrol may enhance the effectiveness of certain conventional cancer treatments while potentially reducing their side effects. Some studies indicate it can make cancer cells more sensitive to chemotherapy and radiation. However, it's absolutely essential to discuss any supplement use with your oncology team, as timing and interactions with specific treatments must be carefully considered. Many integrative oncology programs now include resveratrol as part of important treatment plans.
What are the best food sources of resveratrol?
The richest dietary sources of resveratrol include red grapes (especially the skin), red wine (in moderation), blueberries, cranberries, peanuts, pistachios, and dark chocolate with high cacao content. While these foods provide beneficial amounts, achieving therapeutic levels similar to those used in cancer research studies typically requires supplementation. Incorporating these foods into your diet provides not only resveratrol but also other beneficial compounds that work synergistically.
Are there any side effects or risks associated with resveratrol supplementation?
Resveratrol is generally well-tolerated, but some people may experience mild digestive issues at higher doses. It can interact with blood-thinning medications and may affect how the liver processes certain drugs. Pregnant or nursing women should avoid high-dose supplementation. Always choose high-quality, third-party tested supplements and start with lower doses to assess tolerance. Consulting with healthcare providers ensures safe and effective use.
How long does it take to see benefits from resveratrol supplementation?
The timeline for experiencing benefits from resveratrol varies depending on individual factors and health goals. Some antioxidant and anti-inflammatory effects may be noticed within weeks, while cancer-preventive benefits develop over months to years of consistent use. Research studies typically evaluate outcomes after several months of supplementation. Patience and consistency are key, as resveratrol works by supporting long-term cellular health rather than providing immediate dramatic changes.
What makes resveratrol different from other cancer-fighting supplements?
Resveratrol stands out due to its multi-targeted approach and extensive research backing. Unlike supplements that work through single mechanisms, resveratrol influences multiple pathways including PI3K/AKT signaling, inflammation, oxidative stress, and gene expression. Its ability to both prevent cancer development and enhance treatment effectiveness makes it unique. Additionally, its natural origin and generally excellent safety profile make it an attractive option for long-term use in cancer prevention strategies.
Should everyone take resveratrol for breast cancer prevention?
While resveratrol shows significant promise for breast cancer prevention, supplementation decisions should be individualized. Factors to consider include family history, genetic risk factors, current health status, and other medications or supplements being taken. Those at higher risk for breast cancer may benefit more from preventive supplementation. A balanced approach including healthy diet, regular exercise, stress management, and appropriate screening remains essential. Consulting with healthcare providers helps determine if resveratrol supplementation aligns with your personal health goals and risk profile.
References:
- https://www.tandfonline.com/doi/abs/10.3109/19390211.2011.650842
- https://www.sciencedirect.com/science/article/pii/S0955286305000355
- https://onlinelibrary.wiley.com/doi/abs/10.1002/ijc.25930
The NY Academy Of Sciences Recommends A Resveratrol Dosage
Understanding Resveratrol Dosage: What Science Tells Us
Resveratrol has captured scientific attention since the 1940s, but recent breakthrough studies have revolutionized our understanding of optimal resveratrol dosage recommendations. This powerful polyphenol, found naturally in red wine, grapes, and berries, offers extraordinary potential for supporting healthy aging, cardiovascular wellness, and cellular protection.
The journey to finding your ideal resveratrol intake starts with understanding that supplementation isn't one-size-fits-all. Your body's unique biochemistry, health goals, and lifestyle factors all play important roles in determining the most effective dose for your needs.
Recent Scientific Breakthroughs in Resveratrol Research
The past decade has witnessed remarkable advances in resveratrol research, with groundbreaking studies published in prestigious journals like the Annals of the New York Academy of Sciences. These investigations have shed new light on how different resveratrol dosage levels impact various aspects of human health.
Researchers have discovered that resveratrol activates sirtuins, proteins that regulate cellular health and longevity. [Howitz et al., 2003] This activation triggers a cascade of beneficial effects throughout your body, from enhanced mitochondrial function to improved insulin sensitivity. The key lies in finding the dosage that optimally activates these pathways without overwhelming your system.
Personalized Resveratrol Dosage: Why Individual Variation Matters
Your optimal resveratrol dosage depends on several personal factors that make your supplementation needs unique. Research published in Dose-Response journal demonstrates that individual responses to resveratrol can vary significantly based on:
- Metabolic rate and body composition
- Age and overall health status
- Genetic factors affecting supplement absorption
- Existing medications and supplement interactions
- Lifestyle factors including diet and exercise habits
Blood testing and urinalysis can help healthcare providers assess your resveratrol levels and adjust dosing accordingly. This personalized approach ensures you receive maximum benefits while minimizing any potential side effects.
important Resveratrol Dosage Guidelines
Based on extensive clinical research and expert recommendations, here's a detailed breakdown of resveratrol dosage ranges for different therapeutic goals:
Entry-Level Resveratrol Dosage: 100-500 mg Daily
 Micronized Resveratrol for Enhanced Absorption
Micronized Resveratrol for Enhanced Absorption
This foundational resveratrol dosage range suits individuals new to supplementation or those seeking general antioxidant support. Benefits at this level include:
- Gentle introduction to resveratrol's effects
- Basic antioxidant protection against free radicals
- Support for healthy inflammation response
- Initial cardiovascular benefits
Starting with lower doses allows your body to adjust gradually while you monitor your response. Many people report increased energy and improved well-being within the first few weeks at this dosage level.
Therapeutic Resveratrol Dosage: 500-1500 mg Daily
 X500 Pure Resveratrol for Therapeutic Benefits
X500 Pure Resveratrol for Therapeutic Benefits
The Academy of Sciences specifically recommends 500 mg as an effective starting point for therapeutic benefits. This moderate resveratrol dosage range represents the sweet spot for most adults seeking substantial health improvements:
- Enhanced cellular protection and DNA repair
- Improved metabolic function and weight management support
- Stronger cardiovascular benefits including blood pressure regulation
- Better cognitive function and neuroprotection
- Increased exercise performance and recovery
Advanced Resveratrol Dosage: Above 1500 mg Daily
Recent studies have explored higher resveratrol dosage levels, with some research showing benefits at 2000 mg daily. These elevated doses may offer:
- Maximum sirtuin activation for longevity benefits
- Intensive support for specific health conditions
- Enhanced athletic performance and endurance
- Accelerated recovery from oxidative stress
However, higher doses require careful monitoring and should only be considered under professional guidance.
The Evolution of Resveratrol Supplementation
The landscape of resveratrol supplementation has evolved dramatically. From basic powders to advanced formulations, today's options offer unprecedented bioavailability and effectiveness. The progression from standard resveratrol to micronized versions, and now to complete formulations like M98 Resveratrol Complete, represents a quantum leap in supplement technology.
These advanced formulations combine resveratrol with complementary compounds that enhance its effects, creating synergistic benefits that surpass isolated resveratrol supplementation.
The Surprising Health Benefits of Resveratrol Supplements
Resveratrol Supplements: Complete Guide to Benefits, Science, and Daily Use
Resveratrol supplements have revolutionized natural health support, offering remarkable benefits that extend from cardiovascular wellness to cognitive function. This in-depth guide explores how resveratrol supplements work, their scientifically-backed benefits, and practical ways to incorporate them into your daily wellness routine for optimal results.

What Are Resveratrol Supplements and Why They Matter
Resveratrol supplements contain concentrated forms of a powerful polyphenol naturally found in red grapes, berries, peanuts, and dark chocolate. This remarkable compound gained worldwide attention after researchers discovered its connection to the "French Paradox" - the observation that French people had relatively low rates of heart disease despite consuming rich foods. Today, millions turn to resveratrol supplements as a convenient way to harness these protective benefits without consuming large quantities of wine or specific foods.
The popularity of resveratrol supplements stems from their versatility in supporting multiple aspects of health simultaneously. Unlike single-purpose supplements, resveratrol works at the cellular level to promote longevity, protect against oxidative stress, and support healthy aging processes. Research shows that regular supplementation may activate sirtuins, proteins that regulate cellular health and metabolism, potentially extending both lifespan and healthspan.
Modern resveratrol supplements come in various forms, including capsules, tablets, and liquid formulations. The most effective supplements use trans-resveratrol, the bioactive form that your body can readily absorb and utilize. Quality manufacturers often combine resveratrol with other synergistic compounds like quercetin or grape seed extract to enhance absorption and maximize benefits.
The Science Behind Resveratrol Supplements' Effectiveness
Scientific research reveals that resveratrol supplements work through multiple biological pathways to deliver their health benefits. At the molecular level, resveratrol activates SIRT1, often called the "longevity gene," which regulates cellular metabolism, DNA repair, and inflammatory responses. [Miyazaki et al., 2008] This activation triggers a cascade of beneficial effects throughout the body, from improved mitochondrial function to enhanced cellular resilience.
Studies demonstrate that resveratrol supplements possess potent antioxidant properties, neutralizing harmful free radicals that contribute to aging and disease. Research shows that resveratrol's antioxidant capacity provides significant free radical scavenging ability, helping protect cells from oxidative damage and supporting overall health and vitality. [Olas et al., 2002]
Clinical trials have explored resveratrol supplements' impact on various health markers. Research involving adults with type 2 diabetes found that daily resveratrol supplementation improved insulin sensitivity and reduced oxidative stress. [Bhatt et al., 2011] Another research project demonstrated that participants taking resveratrol supplements experienced better cognitive performance and increased cerebral blood flow compared to placebo groups. [Evans et al., 2018]
The bioavailability of resveratrol supplements has been a focus of recent scientific advancement. While early concerns existed about absorption rates, modern formulation techniques have dramatically improved how effectively the body utilizes supplemental resveratrol. Micronized particles, liposomal delivery systems, and combination with piperine (black pepper extract) have all shown promise in enhancing resveratrol absorption. [Johnson et al., 2011]
Brain Health and Cognitive Function Support
Emerging research highlights resveratrol supplements' remarkable potential for supporting brain health and cognitive function. Studies show that resveratrol crosses the blood-brain barrier effectively, delivering neuroprotective benefits directly to brain tissue. This unique ability positions resveratrol supplements as valuable tools for maintaining mental clarity and cognitive performance throughout life.
Resveratrol supplements appear to protect neurons through several mechanisms. They reduce neuroinflammation, a key factor in cognitive decline, while promoting the clearance of beta-amyloid proteins associated with memory problems. [Marambaud et al., 2005] Additionally, resveratrol enhances brain-derived neurotrophic factor (BDNF) production, supporting the growth and maintenance of healthy neurons. [Madhyastha et al., 2011]
Clinical trials examining resveratrol supplements' effects on memory and cognitive function have yielded promising results. Participants taking resveratrol showed improvements in memory retention, processing speed, and overall cognitive performance. These benefits were particularly pronounced in older adults, suggesting that resveratrol supplements may help maintain mental sharpness during the aging process. [Evans et al., 2018]
Anti-Aging and Longevity Benefits
The connection between resveratrol supplements and longevity has captivated researchers since early studies showed life extension in various organisms. While human longevity studies require decades to complete, current evidence suggests that resveratrol supplements activate many of the same pathways associated with caloric restriction, a proven method for extending lifespan in numerous species.
Resveratrol supplements support healthy aging through multiple mechanisms. They help maintain telomere length, protective caps on chromosomes that shorten with age. By preserving telomere integrity, resveratrol may slow cellular aging and maintain youthful function longer. [Matsuda et al., 2012] Additionally, resveratrol enhances autophagy, the cellular cleaning process that removes damaged components and promotes cellular renewal. [Quan et al., 2018]
Skin health represents another visible benefit of resveratrol supplements' anti-aging properties. By protecting against UV damage, reducing inflammation, and supporting collagen production, resveratrol helps maintain skin elasticity and appearance. Many users report improvements in skin texture, reduced fine lines, and a more youthful complexion after consistent supplementation.
Exercise Performance and Recovery Enhancement
Athletes and fitness enthusiasts increasingly turn to resveratrol supplements for their performance-enhancing properties. Research shows that resveratrol improves oxygen utilization during exercise, allowing for better endurance and reduced fatigue. This enhanced efficiency translates to improved workout performance and faster achievement of fitness goals.
Recovery represents another area where resveratrol supplements excel. By reducing exercise-induced inflammation and oxidative stress, resveratrol helps muscles recover more quickly between training sessions. Studies demonstrate that resveratrol supplementation attenuates exercise-induced inflammatory markers, supporting better performance during consecutive training days.
The mechanisms behind resveratrol's exercise benefits involve enhanced mitochondrial biogenesis and improved muscle fiber composition. Resveratrol supplements appear to promote the development of slow-twitch muscle fibers, which are more efficient for endurance activities. This adaptation, combined with better oxygen delivery and utilization, creates ideal conditions for athletic improvement.
Making Resveratrol Supplements Part of Your Wellness Journey
Integrating resveratrol supplements into a whole-body wellness plan amplifies their benefits. Rather than viewing supplementation as a standalone solution, consider how resveratrol complements other healthy lifestyle choices. The synergy between proper nutrition, regular exercise, stress management, and strategic supplementation creates optimal conditions for health improvement.
Setting realistic expectations helps ensure satisfaction with resveratrol supplements. While some benefits like increased energy may appear within weeks, others such as cardiovascular improvements or anti-aging effects develop over months or years. Tracking progress through regular health assessments provides objective feedback on your supplement regimen's effectiveness.
The journey toward better health through resveratrol supplements offers hope and possibility for people at every life stage. Whether you seek to maintain youthful vitality, support specific health goals, or invest in long-term wellness, resveratrol supplements provide scientifically-backed support for your aspirations.
Take Action: Transform Your Health with Resveratrol Supplements
The journey to optimal health and longevity begins with informed choices about supplementation. Resveratrol supplements offer a scientifically-validated path to enhanced wellness, supporting everything from cardiovascular health to cognitive function. By understanding the science, choosing quality products, and maintaining consistency, you can harness the full potential of this remarkable compound.
Ready to experience the transformative benefits of premium resveratrol supplementation? Consider M98, an advanced resveratrol supplement that surpasses traditional formulations through revolutionary bioavailability technology. With enhanced absorption and proven efficacy, M98 delivers unparalleled support for your health and vitality goals. Discover M98 and elevate your wellness journey today!
Do you prefer to take Resveratrol from wine or as supplements?
Resveratrol Supplements vs Red Wine: Complete Guide to Choosing the Best Source for Longevity
The quest for longevity and optimal health has led many to explore resveratrol, a powerful compound that promises remarkable anti-aging benefits and cellular protection. However, a common question emerges: should you rely on red wine or choose concentrated resveratrol supplements to harness these extraordinary health benefits? The answer may surprise you and could transform your approach to healthy aging and longevity optimization.
Understanding the dramatic differences between resveratrol supplements and red wine becomes important when you're serious about maximizing health benefits and extending your lifespan. While the romantic notion of sipping red wine for health persists, the scientific reality reveals that resveratrol supplements offer vastly superior concentrations and consistency compared to any dietary source, including wine. This fundamental difference can mean the distinction between achieving meaningful health benefits and simply enjoying a pleasant beverage.
This in-depth guide explores the compelling evidence supporting resveratrol supplementation over wine consumption, examining dosage requirements, bioavailability factors, and the important science behind micronized formulations that maximize therapeutic potential. Whether you're seeking longevity benefits, cardiovascular protection, or cellular anti-aging effects, understanding how to optimize your resveratrol intake can unlock remarkable health improvements that dietary sources simply cannot provide.
The Truth About Resveratrol Supplements vs Wine: Why Supplements Win Every Time
When it comes to obtaining therapeutic levels of resveratrol, the math is both simple and startling. Quality resveratrol supplements provide dosages ranging from 250mg to 1000mg daily, delivering concentrated amounts that would be impossible to achieve through wine consumption. The fundamental question "Can I get equivalent amounts of resveratrol through food or wine?" has a definitive answer: absolutely not, due to the minimal resveratrol content in even the richest dietary sources.
The concentration disparity becomes clear when you examine the numbers. Red wine contains approximately 0.2 to 5.8 milligrams of resveratrol per liter, meaning you would need to consume very large quantities of wine daily to match a single 250mg supplement dose. This astronomical consumption would not only be impossible but would create severe health consequences that completely negate any potential benefits from the resveratrol content.
Even the most resveratrol-rich foods pale in comparison to supplement concentrations. While red grape skins, dark berries, and peanuts contain measurable amounts of resveratrol, achieving therapeutic levels through diet alone would require consuming impractical quantities of these foods daily. This reality makes concentrated resveratrol supplements the only viable option for those serious about harnessing this compound's remarkable health benefits.
The bioavailability advantage of quality supplements further amplifies this concentration benefit. Advanced formulations use specialized processing techniques to enhance absorption, ensuring that more of the resveratrol you consume actually reaches your cells where it can provide therapeutic effects. This combination of high concentration and enhanced bioavailability makes supplements exponentially more effective than any dietary source.
Red Wine and Health: Separating Marketing Myths from Scientific Reality
The popular belief that red wine consumption provides significant health benefits has been perpetuated by selective interpretation of research and romanticized notions about Mediterranean lifestyles. While moderate red wine consumption can fit into a healthy dietary pattern, the notion of red wine as a health panacea simply doesn't withstand scientific scrutiny when examined objectively.
The famous "French Paradox" observations that initially sparked interest in red wine's health effects have been largely explained by broader dietary and lifestyle patterns rather than wine consumption specifically. French populations traditionally following Mediterranean-style diets consume abundant fresh vegetables, fruits, legumes, and healthy fats while limiting processed foods and red meat. These broader dietary patterns, rather than wine consumption alone, likely account for observed health benefits.
When alcohol consumption is examined independently, the evidence becomes clear: even small amounts of alcohol provide no health benefits and can contribute to various health risks. Current research indicates that any potential benefits from compounds like resveratrol in wine are far outweighed by alcohol's negative effects on liver function, sleep quality, cognitive performance, and disease risk.
The key insight for health-conscious individuals is that you can obtain all of resveratrol's remarkable benefits through supplementation without any of alcohol's drawbacks. This approach allows you to maximize therapeutic benefits while maintaining optimal health across all body systems, making daily resveratrol supplements far superior to wine consumption for serious health optimization.
Understanding Resveratrol's Remarkable Mechanisms: How It Transforms Health
The extraordinary health benefits of resveratrol supplementation, particularly in its highly bioavailable trans-resveratrol form, stem from its unique ability to influence fundamental cellular processes that determine how we age and how well our bodies function throughout life. Understanding these mechanisms helps explain why therapeutic doses through supplementation provide such profound benefits compared to the minimal amounts available through dietary sources.
Calorie Restriction Mimetics: Longevity Without Deprivation
One of resveratrol's most remarkable properties is its ability to activate the same beneficial genes and cellular pathways triggered by calorie restriction, potentially providing longevity benefits without the need for severe dietary limitations. Calorie restriction diets, which involve consuming 30-40% fewer calories while maintaining adequate nutrition, have been shown to extend lifespan and improve health across multiple species.
However, these restrictive eating patterns prove extremely difficult to follow long-term and may not be practical or safe for many individuals. The exciting discovery that resveratrol supplementation can activate similar genetic pathways offers a more accessible approach to accessing these longevity benefits without the challenges and risks associated with severe calorie restriction.
The genes activated by resveratrol include sirtuins, often called "longevity genes," which regulate cellular repair, metabolism, and stress resistance. These proteins play important roles in maintaining cellular health, supporting DNA repair mechanisms, and protecting against age-related decline. By activating these pathways through supplementation, you can potentially access many of calorie restriction's benefits while maintaining normal, healthy eating patterns. [Miyazaki et al., 2008]
Powerful Antioxidant Protection Against Cellular Aging
Resveratrol functions as a powerful antioxidant that provides important protection against oxidative stress, one of the primary drivers of cellular aging and age-related disease development. Free radicals generated through normal cellular metabolism, environmental exposures, and stress can cause cumulative damage to cellular structures, including DNA, proteins, and cellular membranes.
This oxidative damage manifests as visible signs of aging in skin, declining organ function, reduced energy levels, and increased susceptibility to age-related diseases. Resveratrol's antioxidant capacity helps neutralize these harmful free radicals before they can cause cellular damage, effectively slowing the aging process at its source. [Olas et al., 2002]
The cellular protection provided by resveratrol extends throughout the body, supporting brain health and cognitive function, maintaining cardiovascular health and circulation, protecting liver function and detoxification capacity, and preserving skin health and appearance. This comprehensive protection makes resveratrol supplementation particularly valuable for those seeking to maintain optimal function across all body systems as they age.
Cardiovascular Health and Metabolic Optimization
Resveratrol's cardiovascular benefits represent some of its most well-documented effects, with research consistently demonstrating improvements in heart health, circulation, and metabolic function. These benefits include enhanced endothelial function that supports healthy blood flow, improved cholesterol profiles and lipid metabolism, reduced inflammation in blood vessels and heart tissue, and better insulin sensitivity and glucose metabolism. [Dziadkowiak et al., 2025]
The metabolic benefits of resveratrol supplementation extend to weight management and energy optimization, as the compound supports efficient cellular energy production while promoting healthy fat metabolism. These effects can contribute to maintaining healthy body composition and optimal energy levels throughout the aging process.
Quality Considerations: Choosing Superior Resveratrol Supplements
Get the same quality used in longevity research.
RevGenetics has supplied premium resveratrol since 2007. Independent lab-verified, wheat-free, no fillers.
View Resveratrol Products →The resveratrol supplement market includes products with dramatically different qualities, potencies, and bioavailabilities, making informed selection important for achieving desired health benefits. Understanding key quality indicators helps ensure you choose products that provide optimal therapeutic potential.
Essential Quality Factors
High-quality resveratrol supplements should provide standardized trans-resveratrol content, as this represents the most bioactive form of the compound. Products should clearly state the amount of trans-resveratrol per serving rather than total resveratrol content, which may include less active forms.
Third-party testing for purity and potency ensures that products contain stated amounts of active compounds without harmful contaminants. Reputable manufacturers provide certificates of analysis and maintain strict quality control standards throughout the manufacturing process.
Advanced processing technologies like micronization dramatically improve bioavailability and therapeutic effectiveness. These sophisticated formulations may cost more initially but provide superior value through enhanced absorption and effectiveness.
RevGenetics: Leading Innovation in Resveratrol Technology
RevGenetics has established itself as a leader in resveratrol supplementation through their commitment to advanced formulation technologies and rigorous quality standards. Their micronized resveratrol products represent the most sophisticated formulations available, combining optimal bioavailability with consistent potency and purity.
The company's research-based approach to product development ensures that their formulations provide maximum therapeutic benefits while maintaining excellent safety profiles. This commitment to excellence makes RevGenetics products ideal for serious health optimization and longevity applications.
Beyond Supplementation: important Longevity Strategies
While resveratrol supplementation provides powerful support for healthy aging and longevity, the most effective approach typically involves important strategies that address multiple aspects of health optimization. Integrating resveratrol with other evidence-based longevity interventions can create synergistic effects that exceed the benefits of any single approach.
Lifestyle Factors That Enhance Resveratrol Benefits
Regular physical exercise enhances resveratrol's benefits by activating similar cellular pathways and supporting cardiovascular health. The combination of resveratrol supplementation and consistent exercise provides important support for healthy aging and optimal physical function.
Stress management practices including meditation, adequate sleep, and stress reduction techniques complement resveratrol's cellular protective effects. Chronic stress can accelerate aging processes, making stress management important for maximizing longevity benefits.
Optimal nutrition featuring antioxidant-rich foods, healthy fats, and adequate protein provides the foundation for cellular health that resveratrol supplementation can enhance. This balanced approach addresses aging from multiple angles for optimal results.
Monitoring and Optimization
Regular health assessments including biomarker testing can help track the effectiveness of your resveratrol supplementation and overall longevity strategy. Key markers to monitor include inflammatory indicators, cardiovascular risk factors, metabolic health parameters, and cellular aging biomarkers.
Working with healthcare providers knowledgeable about longevity medicine ensures that your resveratrol supplementation integrates safely and effectively with any other treatments or interventions you may be using.
Did You Know That Resveratrol Burns More Calories?
How Resveratrol Burns More Calories: The Science Behind Natural Weight Management
Resveratrol has emerged as a powerful natural compound that revolutionizes how our bodies burn calories and manage weight. This remarkable substance, found naturally in red grapes, berries, and peanuts, offers a scientifically-backed approach to enhancing metabolism without the need for extreme dieting or exhausting exercise routines. Understanding how resveratrol works to increase calorie burning opens up exciting possibilities for anyone seeking sustainable weight management solutions.
The Future of Natural Weight Management with Resveratrol
As research continues to unveil the mechanisms behind how resveratrol burns calories, we're entering an exciting era of natural, science-based weight management solutions. The discovery of resveratrol's effects on UCP proteins and thermogenesis represents just the beginning of our understanding.
Ongoing studies are exploring how resveratrol interacts with other natural compounds to enhance its effects, optimal dosing strategies for different body types, and potential applications for athletic performance. The future looks bright for those seeking natural, sustainable approaches to health and wellness.
Taking the Next Step with Resveratrol
The science is clear: resveratrol burns more calories through its unique ability to enhance thermogenesis and optimize metabolism. This natural compound offers a sustainable, scientifically-backed approach to weight management that aligns with your body's natural processes rather than fighting against them.
Whether you're looking to enhance your current wellness routine, seeking natural weight management support, or simply want to optimize your metabolism, resveratrol presents an exciting opportunity. The combination of increased calorie burning, improved energy levels, and holistic health benefits makes resveratrol a valuable addition to any health-conscious lifestyle.
Start your journey toward better metabolic health today. With consistent use and realistic expectations, resveratrol can help you achieve the balanced, energetic life you deserve. The path to natural weight management and enhanced wellness begins with understanding how your body works and supporting it with scientifically-proven solutions like resveratrol.
References:
- Mercader J, et al. "Resveratrol enhances brown adipocyte formation and mitochondrial biogenesis." Journal of Nutritional Biochemistry. 2011. https://www.hindawi.com/journals/jnme/2011/525094/
- Lagouge M, et al. "Resveratrol improves mitochondrial function and protects against metabolic disease." Nature. 2006. https://www.nature.com/articles/nature05354
Resveratrol Evidence Of Side Effects And Benefits
Frequently Asked Questions About Resveratrol Benefits and Side Effects
What are the main resveratrol benefits for cardiovascular health?
The main resveratrol benefits for cardiovascular health include blood pressure reduction, improved cholesterol levels, enhanced blood vessel function, reduced arterial stiffness, and decreased inflammation. Clinical studies consistently show that resveratrol can lower coronary heart disease risk and improve overall cardiovascular markers. The compound works through multiple pathways simultaneously, providing important cardiovascular protection that extends beyond single-target approaches.
What are the most common resveratrol side effects?
The most common resveratrol side effects are mild and include headaches, digestive upset, nausea, loose stools, and rare skin reactions. These side effects are typically temporary and resolve as the body adjusts to supplementation. Most people tolerate resveratrol very well at recommended doses, and serious adverse effects are extremely rare when using high-quality supplements appropriately.
How much resveratrol should I take for cardiovascular benefits?
Clinical studies typically use 250-500mg of trans-resveratrol daily for cardiovascular benefits. Starting with 150-250mg daily and gradually increasing allows your body to adjust while minimizing potential side effects. Higher doses don't necessarily provide proportionally greater benefits and may increase the likelihood of side effects, so finding your optimal dose is key.
Is resveratrol safe for long-term use?
Resveratrol has an excellent safety profile for long-term use in most healthy adults at recommended doses. Decades of research support its safety for extended supplementation. However, those taking blood-thinning medications, pregnant or nursing women, or individuals with hormone-sensitive conditions should consult healthcare providers before beginning resveratrol supplementation.
Can resveratrol help lower blood pressure naturally?
Yes, clinical studies consistently show that resveratrol can help lower blood pressure naturally. Hypertensive individuals taking resveratrol supplements experienced significant reductions in both systolic and diastolic blood pressure compared to placebo groups. This blood pressure-lowering effect occurs through enhanced nitric oxide production, improved blood vessel function, and reduced arterial stiffness.
What's the difference between trans-resveratrol and regular resveratrol?
Trans-resveratrol is the most bioactive form of resveratrol, providing superior cardiovascular benefits compared to other forms or mixed resveratrol preparations. When choosing supplements, look for products that specify trans-resveratrol content and purity to ensure maximum therapeutic potential. Understanding trans-resveratrol vs. resveratrol differences helps ensure optimal supplementation choices.
Can I take resveratrol with other medications?
Resveratrol may interact with blood-thinning medications by enhancing their anticoagulant effects. If you're taking warfarin, aspirin, or other anticoagulant medications, consult your healthcare provider before starting resveratrol. Generally, resveratrol is well-tolerated with most other medications, but professional guidance ensures safe integration with existing treatments.
How long does it take to see resveratrol benefits?
Some resveratrol benefits, like improved antioxidant status and reduced inflammation markers, may begin within 2-4 weeks of supplementation. However, more significant cardiovascular benefits like blood pressure reduction and improved cholesterol levels typically become apparent after 2-3 months of consistent use at effective doses. Long-term benefits continue to accumulate with sustained supplementation.
Resveratrol on Aggressive Tumor
Resveratrol Shows Promise Against Aggressive Brain Tumors: New Research Reveals Breakthrough Treatment Potential
Resveratrol, a powerful natural compound found in grapes and berries, demonstrates remarkable potential in fighting glioblastoma multiforme (GBM), the most aggressive form of brain cancer. Recent groundbreaking research reveals how this antioxidant-rich substance could transform treatment approaches for patients facing this devastating diagnosis, offering renewed hope where conventional therapies have fallen short.
Understanding Glioblastoma: The Challenge That Resveratrol May Help Overcome
Glioblastoma multiforme stands as the most common primary brain tumor, affecting thousands of patients worldwide each year. With a median survival rate of approximately 14 months, GBM presents one of modern medicine's greatest challenges . The aggressive nature of these tumors, combined with the brain's protective blood-brain barrier that blocks many therapeutic compounds, has historically limited treatment options.
For patients and families confronting this diagnosis, the search for effective treatments becomes important. This is where resveratrol enters the picture as a beacon of hope, showing unique abilities to cross biological barriers and target cancer cells with precision.
The Science Behind Resveratrol's Cancer-Fighting Properties
What makes resveratrol particularly fascinating for researchers is its multi-faceted approach to combating cancer cells. This polyphenol compound, naturally occurring in red wine, peanuts, and various berries, possesses several key characteristics that make it valuable for cancer treatment:
- Anti-inflammatory effects that reduce tumor-promoting inflammation
- Antioxidant properties protecting healthy cells while targeting malignant ones
- Cell cycle regulation preventing uncontrolled cancer cell division
- Apoptosis induction triggering programmed death in cancer cells
- Anti-angiogenic activity cutting off blood supply to tumors
Breaking Through the Blood-Brain Barrier
One of the most exciting aspects of resveratrol research involves its ability to important the blood-brain barrier, a protective mechanism that typically prevents most therapeutic agents from reaching brain tumors. Unlike many conventional drugs, resveratrol's molecular structure allows it to penetrate this barrier, delivering its therapeutic benefits directly where needed most.
Groundbreaking Research: Resveratrol Combined with Radiation Therapy
A important study published in the Journal of Cellular Physiology unveiled remarkable findings about resveratrol's effectiveness when combined with radiation therapy. [Sherry et al., 2011] The research team employed a systematic approach that yielded encouraging results for GBM treatment.
The STAT3 Connection: How Resveratrol Targets Cancer at Its Core
The researchers discovered that GBM tumors depend heavily on a protein called STAT3 for their survival and proliferation. This protein acts like a master switch, enabling cancer cells to grow uncontrollably and resist conventional treatments. The study revealed that resveratrol specifically targets and inhibits STAT3, essentially cutting off the tumor's lifeline. [Sherry et al., 2011]
Through extensive testing in both animal models and cell cultures, the research demonstrated that combining resveratrol with radiation therapy created a synergistic effect. This combination treatment significantly increased cancer cell death rates compared to either treatment alone, marking a substantial advancement in GBM therapy approaches. [Sherry et al., 2011]
Real-World Impact: What This Means for Patients
For individuals diagnosed with glioblastoma, these findings represent more than just laboratory success stories. The research shows that resveratrol supplementation could potentially:
- Enhance radiation therapy effectiveness by making cancer cells more vulnerable to treatment
- Reduce treatment resistance often seen with conventional therapies alone
- Improve overall survival rates beyond current median expectations
- Minimize side effects through targeted action on cancer cells
- Offer a natural adjuvant therapy that complements existing treatment protocols
Optimal Resveratrol Dosing and Administration
While research continues to determine the most effective dosing strategies, current studies suggest that resveratrol's bioavailability plays a important role in its therapeutic effectiveness. Factors influencing optimal administration include:
| Factor | Consideration | Impact on Effectiveness |
|---|---|---|
| Dosage Form | Capsules, liquid, or powder | Affects absorption rate and bioavailability |
| Timing | With meals or empty stomach | Influences absorption and tolerance |
| Combination | With other supplements or medications | May enhance or inhibit effectiveness |
| Quality | Purity and standardization | Determines therapeutic potential |
Beyond Brain Tumors: Resveratrol's Broader Cancer-Fighting Potential
While the focus on glioblastoma represents a critical breakthrough, resveratrol's anti-cancer properties extend to various other malignancies. Research indicates potential benefits for:
- Colorectal cancer prevention and treatment
- Breast cancer cell inhibition
- Prostate cancer progression slowdown
- Lung cancer metastasis prevention
- Skin cancer protection through UV damage reduction
The Molecular Mechanisms at Work
Resveratrol operates through multiple pathways to combat cancer development and progression. Its ability to modulate gene expression, particularly those involved in cell survival and death, makes it a versatile therapeutic agent. The compound influences several critical cellular processes:
Cell Signaling Modulation: By interfering with cancer-promoting signals, resveratrol disrupts the communication networks tumors use to grow and spread.
Epigenetic Regulation: The compound can alter gene expression patterns without changing DNA sequences, potentially reversing cancer-promoting modifications.
Metabolic Reprogramming: Cancer cells often rely on altered metabolism for energy. Resveratrol helps normalize these metabolic pathways, starving tumors of necessary resources.
Frequently Asked Questions About Resveratrol and Cancer Treatment
What exactly is resveratrol and where does it come from?
Resveratrol is a naturally occurring polyphenol compound found primarily in the skin of red grapes, berries (especially blueberries and cranberries), peanuts, and Japanese knotweed. This powerful antioxidant serves as a plant's defense mechanism against stress, injury, and infection. For human health, resveratrol offers remarkable anti-inflammatory and anti-cancer properties, making it a subject of intense scientific study for various health conditions, particularly cancer treatment.
How does resveratrol specifically help with brain tumors like glioblastoma?
Resveratrol demonstrates unique effectiveness against brain tumors through several mechanisms. First, it can cross the blood-brain barrier, which many cancer drugs cannot do. [Carrizzo et al., 2017] Once in the brain, resveratrol inhibits the STAT3 protein that glioblastoma cells need to survive and multiply. When combined with radiation therapy, resveratrol makes cancer cells more sensitive to treatment, significantly increasing cell death rates. This dual action of direct tumor suppression and treatment enhancement makes resveratrol particularly valuable for aggressive brain cancers.
Is it safe to take resveratrol during cancer treatment?
While resveratrol is generally considered safe and well-tolerated, cancer patients should always consult their oncology team before adding any supplements to their treatment regimen. Research shows that resveratrol can enhance certain cancer treatments, particularly radiation therapy, but it may interact with some chemotherapy drugs. Your healthcare provider can assess whether resveratrol supplementation aligns with your specific treatment plan and monitor for any potential interactions or side effects.
What dosage of resveratrol is recommended for cancer patients?
Optimal resveratrol dosing for cancer patients varies based on individual factors and specific cancer types. Clinical studies have used doses ranging from 150mg to 5,000mg daily, though most therapeutic benefits appear at doses between 250-1,000mg per day. The bioavailability of resveratrol can be enhanced by taking it with a small amount of fat or choosing specialized formulations. Always work with healthcare professionals to determine the appropriate dose for your situation, as higher doses don't necessarily mean better results.
How long does it take to see benefits from resveratrol supplementation?
The timeline for experiencing resveratrol's benefits varies depending on the health condition and individual factors. Some patients report improved energy and well-being within 2-4 weeks of starting supplementation. For cancer-specific benefits, studies suggest that consistent use over several months, particularly when combined with conventional treatments, yields the most significant results. Regular monitoring and patience are key, as resveratrol works at the cellular level to create lasting changes rather than providing immediate dramatic effects.
Can resveratrol prevent cancer in healthy individuals?
Research suggests that regular resveratrol consumption may help reduce cancer risk in healthy individuals. Its antioxidant properties protect DNA from damage, while its anti-inflammatory effects create an environment less favorable for cancer development. Population studies show lower cancer rates in regions with high dietary resveratrol intake. While not a guarantee against cancer, incorporating resveratrol through diet or supplementation represents a proactive approach to cancer prevention, especially when combined with other healthy lifestyle choices.
Are there any side effects of taking resveratrol?
Resveratrol is generally well-tolerated with minimal side effects at recommended doses. Some individuals may experience mild digestive discomfort, including nausea or diarrhea, particularly at higher doses. Resveratrol can also interact with blood-thinning medications and may affect estrogen-sensitive conditions. Starting with lower doses and gradually increasing allows your body to adjust while monitoring for any adverse reactions. Most people tolerate resveratrol well and experience primarily positive effects.
What's the difference between resveratrol from food sources versus supplements?
While dietary sources of resveratrol provide valuable health benefits, achieving therapeutic doses through food alone proves challenging. For example, you would need to consume hundreds of glasses of red wine daily to match the resveratrol levels used in cancer studies. Supplements offer standardized, concentrated doses without the calories, sugar, or alcohol found in food sources. However, whole foods provide additional beneficial compounds that work synergistically with resveratrol, making a combination approach ideal for optimal health benefits.
How does resveratrol compare to other natural cancer-fighting compounds?
Resveratrol stands out among natural cancer-fighting compounds for its ability to cross the blood-brain barrier and its well-documented effects on multiple cancer pathways. While compounds like curcumin, green tea extract, and vitamin D also show anti-cancer properties, resveratrol's unique molecular structure and broad-spectrum activity make it particularly versatile. Many integrative oncologists recommend combining resveratrol with other natural compounds for synergistic effects, as each targets different aspects of cancer development and progression.
What should I look for when choosing a resveratrol supplement?
Quality matters significantly when selecting resveratrol supplements. Look for products that specify trans-resveratrol content, as this is the bioactive form. Third-party testing for purity and potency ensures you're getting what the label claims. Consider bioavailability-enhanced formulations that improve absorption. Reputable manufacturers provide certificates of analysis and avoid unnecessary fillers or additives. Price shouldn't be the only factor; investing in high-quality resveratrol ensures you receive the therapeutic benefits documented in research studies.
References:
- Molecular Sciences: Resveratrol and Brain Tumors
- Frontiers in Nutrition: Resveratrol's Anti-Cancer Mechanisms
Resveratrol Study: Increase In Fat Burning Hormone In Human Subjects Is Proved, In Diabetes Study
Resveratrol: Revolutionary Fat-Burning Hormone Booster Proven in Human Clinical Study
Groundbreaking research from Albert Einstein College of Medicine reveals how resveratrol dramatically increases adiponectin, the body's natural fat-burning hormone, by an impressive 53% in just 28 days. [Maginley et al., 2016] This in-depth guide explores the science behind resveratrol's remarkable ability to transform metabolism, reduce inflammation, and enhance insulin sensitivity through clinical evidence.
What Makes Resveratrol a important for Metabolic Health
Imagine discovering a natural compound that could important your body's hidden potential to burn fat more efficiently while simultaneously reducing inflammation and improving insulin sensitivity. That's exactly what researchers at Albert Einstein College of Medicine found when they studied resveratrol in human subjects. This powerful polyphenol, naturally found in red grapes, berries, and peanuts, has emerged as one of the most promising compounds for metabolic optimization and healthy aging.
The journey of resveratrol from laboratory curiosity to clinically proven metabolic enhancer represents a significant breakthrough in nutritional science. Unlike many supplements that rely solely on animal studies or theoretical benefits, resveratrol has demonstrated its effectiveness in rigorous human clinical trials, offering real hope for those seeking natural solutions to metabolic challenges.
The Science Behind Resveratrol's Fat-Burning Properties
 Clinical-grade X500 Resveratrol: The exact formulation used in the Albert Einstein College of Medicine study
Clinical-grade X500 Resveratrol: The exact formulation used in the Albert Einstein College of Medicine study
The clinical study conducted at Albert Einstein College of Medicine employed a gold-standard randomized, double-blind, placebo-controlled methodology. Participants received 2 grams of pure resveratrol daily (equivalent to four X500 capsules) for 28 days. This dosage was carefully selected based on preliminary research indicating optimal bioavailability and therapeutic effect.
Key Study Parameters and Methodology
The research team implemented important metabolic assessments including:
- Hepatic insulin sensitivity measurements to evaluate liver function and glucose metabolism
- Peripheral insulin sensitivity testing to assess how effectively muscles and tissues respond to insulin
- Muscle tissue biopsies to examine cellular changes at the microscopic level
- Subcutaneous adipose tissue analysis to directly measure fat cell behavior and hormone production
These extensive measurements were conducted both before and after the 28-day supplementation period, providing clear before-and-after comparisons that revealed resveratrol's remarkable effects on human metabolism.
Breakthrough Results: How Resveratrol Transforms Your Metabolism
The results of this groundbreaking study exceeded even the researchers' optimistic expectations. After just four weeks of resveratrol supplementation, participants experienced transformative metabolic improvements that typically require months of strict dieting or intensive exercise to achieve.
1. Dramatic Increase in Fat-Burning Hormone Adiponectin
The most striking finding was a 53% increase in adiponectin levels within fat tissue. [Maginley et al., 2016] Adiponectin serves as your body's master regulator of fat metabolism, signaling cells to burn stored fat for energy rather than storing additional calories. This hormone plays important roles in:
- Accelerating the breakdown of fatty acids for energy production
- Enhancing mitochondrial function for improved cellular energy
- Reducing fat accumulation in liver and muscle tissue
- Improving overall metabolic flexibility
2. Enhanced Insulin Sensitivity Without Dietary Changes
Participants showed a 22% improvement in glucose uptake, indicating significantly enhanced insulin sensitivity. [Maginley et al., 2016] This improvement occurred without any changes to diet or exercise habits, suggesting that resveratrol activates metabolic pathways similar to those triggered by caloric restriction. This finding offers particular hope for individuals struggling with insulin resistance or pre-diabetes who find traditional dietary interventions challenging to maintain.
3. Powerful Anti-Inflammatory Effects
The study revealed dramatic reductions in inflammatory markers:
| Inflammatory Marker | Reduction Percentage | Health Significance |
|---|---|---|
| TNF-alpha (TNFa) | 68% [Maginley et al., 2016] | Reduced chronic inflammation and metabolic dysfunction |
| Interleukin-6 (IL6) | 52% [Maginley et al., 2016] | Lower risk of insulin resistance and cardiovascular issues |
| PAI-1 in adipose macrophages | Significant reduction | Improved blood flow and reduced clotting risk |
These reductions in inflammatory cytokines represent a fundamental shift in how fat tissue behaves, transforming it from a source of inflammation to a metabolically active tissue that supports overall health.
Real-World Applications: How to Harness Resveratrol's Benefits
The clinical study provides clear guidance on how to effectively use resveratrol for metabolic enhancement. The research protocol offers a proven framework that individuals can follow to potentially replicate these impressive results.
Optimal Dosing Strategy Based on Clinical Evidence
The study participants took their resveratrol in a specific pattern that maximized absorption and effectiveness:
- Morning dose: 2 capsules (1000mg) with breakfast
- Evening dose: 2 capsules (1000mg) with dinner
- Total daily intake: 2000mg pure trans-resveratrol
- Duration: Minimum 28 days for measurable metabolic changes
This divided dosing strategy ensures consistent blood levels throughout the day, maximizing resveratrol's metabolic benefits while minimizing any potential digestive sensitivity.
Enhancing Resveratrol Absorption and Effectiveness
To maximize the benefits of resveratrol supplementation, consider these evidence-based strategies:
- Take with food: The study showed resveratrol can be taken with meals, which may enhance absorption and reduce any potential stomach sensitivity
- Maintain consistency: Daily supplementation without gaps ensures steady therapeutic levels
- Consider complementary nutrients: Some research suggests combining resveratrol with quercetin or piperine may enhance bioavailability
- Stay hydrated: Adequate water intake supports optimal nutrient transport and cellular function
Frequently Asked Questions About Resveratrol and Fat-Burning Hormones
How quickly can I expect to see results from resveratrol supplementation?
Based on the clinical study at Albert Einstein College of Medicine, significant metabolic improvements were observed after 28 days of consistent supplementation with 2 grams of resveratrol daily. Participants experienced a 53% increase in adiponectin (fat-burning hormone) and a 22% improvement in glucose uptake within this timeframe. While individual results may vary, maintaining consistent daily supplementation for at least four weeks appears optimal for experiencing measurable benefits.
What is adiponectin and why is a 53% increase significant?
Adiponectin is a important hormone produced by fat cells that regulates glucose levels and fatty acid breakdown. Higher adiponectin levels are associated with improved insulin sensitivity, enhanced fat metabolism, and reduced inflammation. A 53% increase is remarkably significant because most interventions, including exercise and weight loss, typically produce much smaller increases. This dramatic boost means your body becomes substantially more efficient at burning stored fat for energy and maintaining healthy blood sugar levels.
Can I take resveratrol with my current medications?
While the clinical study showed resveratrol to be well-tolerated with no significant adverse effects, it's essential to consult with your healthcare provider before adding any supplement to your routine, especially if you're taking medications. Resveratrol may interact with blood thinners, NSAIDs, and certain other medications. Your doctor can evaluate potential interactions and help determine if resveratrol is appropriate for your specific health situation.
Is 2 grams of resveratrol daily safe for long-term use?
The clinical study used 2 grams daily for 28 days with excellent safety results. While this study duration provides strong evidence for short-term safety, long-term studies at this dosage are still ongoing. Many people successfully use resveratrol long-term at various doses. Starting with the clinically proven protocol and monitoring your response with your healthcare provider ensures you're using resveratrol safely and effectively for your individual needs.
How does resveratrol compare to prescription medications for metabolic health?
Resveratrol works through different mechanisms than most prescription medications, activating natural metabolic pathways like SIRT1 and AMPK. The 22% improvement in glucose uptake shown in the study is comparable to some diabetes medications, but resveratrol additionally offers anti-inflammatory benefits and increased fat-burning hormone production. Rather than replacing medications, resveratrol may serve as a complementary approach to support overall metabolic health. Always work with your healthcare provider to determine the best approach for your situation.
Should I take resveratrol with food or on an empty stomach?
The clinical study protocol had participants take resveratrol with meals - two capsules with breakfast and two with dinner. Taking resveratrol with food may enhance absorption and reduce any potential digestive sensitivity. The fat content in meals can particularly help with the absorption of this fat-soluble compound. This approach also makes it easier to remember your doses and maintain the consistency important for optimal results.
Can resveratrol help with stubborn belly fat?
The study's findings suggest resveratrol may be particularly effective for abdominal fat reduction. The 53% increase in adiponectin specifically targets visceral fat metabolism, and the significant reduction in inflammatory markers (68% reduction in TNF-alpha) addresses one of the key factors in stubborn belly fat accumulation. Additionally, the improved insulin sensitivity helps prevent further fat storage in the abdominal area, creating a balanced approach to addressing this common concern.
What quality factors should I look for in a resveratrol supplement?
Choose resveratrol supplements that specify "trans-resveratrol" content, as this is the bioactive form used in clinical studies. Look for products that provide third-party testing results for purity and potency. The supplement should be manufactured in GMP-certified facilities and free from unnecessary fillers or artificial ingredients. Pharmaceutical-grade resveratrol, like that used in the Albert Einstein College study, ensures you're getting the same quality that produces clinical results.
Can I get enough resveratrol from food sources alone?
While resveratrol occurs naturally in red grapes, berries, and peanuts, achieving the therapeutic dose used in the clinical study (2 grams daily) through diet alone is virtually impossible. You would need to consume hundreds of pounds of grapes or thousands of glasses of red wine daily to match the study dosage. This is why concentrated supplements are necessary to achieve the metabolic benefits demonstrated in clinical research. Food sources can complement supplementation but cannot replace it for therapeutic effects.
Will resveratrol interfere with my exercise routine or athletic performance?
Research suggests resveratrol may actually enhance exercise benefits rather than interfere with them. By improving mitochondrial function, increasing fat oxidation, and reducing inflammation, resveratrol can support better energy production and recovery. The improved insulin sensitivity and glucose uptake demonstrated in the study may also enhance athletic performance by improving how your muscles utilize energy. Many athletes and fitness enthusiasts successfully combine resveratrol supplementation with their training routines for synergistic benefits.
Note: These statements have not been evaluated by the Food and Drug Administration. Resveratrol supplements are not intended to diagnose, treat, cure, or prevent any disease. Always consult with a qualified healthcare professional before starting any supplement regimen.
Resveratrol protects against Stress
Stress Response Revolution: How Resveratrol Protects Your Cells From Daily Stress

Stress affects every cell in your body, but groundbreaking research reveals how resveratrol creates a powerful shield against cellular stress damage. Scientists at The Scripps Research Institute have uncovered a revolutionary mechanism that explains why this natural compound provides such remarkable protection against stress-related aging and disease. [Shim et al., 2014]
Whether you're dealing with work pressure, environmental toxins, or the natural aging process, understanding how to combat stress at the cellular level becomes essential for maintaining vibrant health. This in-depth guide explores the latest scientific breakthroughs in stress protection and reveals practical strategies for harnessing resveratrol's protective power.
Understanding Cellular Stress: The Hidden Threat to Your Health
Before diving into resveratrol's protective mechanisms, it's important to understand what cellular stress really means. Unlike the emotional stress we feel during a hectic day, cellular stress occurs when your cells face challenges that threaten their normal function. This includes:
- Oxidative stress from free radical damage
- Metabolic stress from poor diet and lifestyle choices
- Environmental stress from toxins and pollution
- Age-related stress as cellular repair mechanisms decline
These forms of stress accumulate over time, accelerating aging and increasing disease risk. That's where resveratrol's newly discovered stress-fighting abilities become game-changing.
Breakthrough Discovery: Resveratrol's Novel Stress Protection Mechanism
The December publication in Nature (doi:10.1038) revealed something extraordinary about how resveratrol combats stress. Researchers discovered that this compound doesn't just work through one pathway – it activates multiple stress response systems simultaneously, creating important cellular protection.
What makes this discovery particularly exciting is the identification of tRNA synthetase as a key target. This enzyme, typically involved in protein production, transforms into a stress-fighting powerhouse when resveratrol binds to it. The sensitivity of this interaction is remarkable -- 1,000 times more responsive than previously known resveratrol targets. [Shim et al., 2014]
The Multi-Target Approach to Stress Protection
Resveratrol's ability to target multiple cellular pathways explains its impressive range of benefits. Research confirms that resveratrol provides:
- Cardio-neuro-protective effects that shield your heart and brain from stress damage
- Anti-diabetic properties that help regulate metabolic stress
- Anti-cancer effects by protecting DNA from stress-induced mutations
- Anti-aging benefits through activation of longevity pathways
This versatility stems from resveratrol's unique ability to sense different types of cellular stress and respond appropriately, making it a true adaptogen at the molecular level.
How Stress Damages Your Body: The Cellular Cascade
Looking for high-absorption resveratrol?
RevGenetics Micronized Trans-Resveratrol is formulated for superior bioavailability -- more gets into your bloodstream where it counts.
Check Out Micronized Resveratrol →To appreciate resveratrol's protective power, consider how unchecked stress wreaks havoc on your cells. When stress overwhelms your natural defenses, it triggers a destructive cascade:
- Initial stress exposure disrupts normal cellular function
- Protective mechanisms become overwhelmed, leading to damage accumulation
- Cellular communication breaks down, affecting entire organ systems
- Chronic inflammation develops, accelerating aging and disease
- DNA damage occurs, potentially leading to serious health issues
Resveratrol interrupts this cascade at multiple points, providing important protection that helps maintain cellular resilience even under challenging conditions.
The Phosphodiesterase Connection: Another Layer of Stress Defense
Earlier research from 2012 revealed another fascinating aspect of resveratrol's stress-fighting arsenal. Scientists discovered that resveratrol directly binds to and blocks phosphodiesterase (PDE) enzymes. [Park et al., 2012] This interaction activates essential stress response proteins that prevent age-associated metabolic diseases.
By inhibiting PDE enzymes, resveratrol enhances cellular energy production and improves the body's ability to handle metabolic stress. This mechanism particularly benefits individuals dealing with:
- Blood sugar imbalances
- Cardiovascular stress
- Cognitive decline
- Inflammatory conditions
Maximizing Stress Protection: Practical Strategies
Understanding the science behind resveratrol's stress protection enables you to maximize its benefits. Here are evidence-based strategies for optimal results:
1. Timing Your Resveratrol Intake
Research suggests that taking resveratrol at specific times enhances its stress-protective effects. Many experts recommend morning supplementation to align with your body's natural stress response rhythms.
2. Combining Stress-Fighting Nutrients
While resveratrol provides powerful protection alone, combining it with complementary nutrients amplifies benefits:
- Quercetin: Enhances resveratrol absorption and adds antioxidant power
- Vitamin D: Supports immune function during stress
- Omega-3 fatty acids: Reduce inflammatory stress responses
- Magnesium: Calms nervous system stress
3. Lifestyle Factors That Enhance Resveratrol's Effects
Your daily habits significantly impact how well resveratrol protects against stress:
- Regular exercise activates similar stress-protective pathways
- Quality sleep allows cellular repair mechanisms to function optimally
- Mindfulness practices reduce overall stress burden
- Nutrient-dense diet provides cofactors for stress response systems
The Future of Stress Management: Personalized Approaches
The discovery that resveratrol affects different cells at varying sensitivities opens exciting possibilities for personalized stress management. Future applications may include:
- Genetic testing to determine optimal resveratrol dosing for individual stress responses
- Targeted formulations for specific types of cellular stress
- Combination therapies that maximize cell and tissue-specific benefits
- Biomarker monitoring to track stress protection effectiveness
As research continues, we're moving toward more precise, effective strategies for combating the stress that accelerates aging and disease.
Frequently Asked Questions About Stress and Resveratrol
How does cellular stress differ from emotional stress?
Cellular stress occurs at the microscopic level when cells face challenges like oxidative damage, toxins, or metabolic imbalances. While emotional stress can trigger cellular stress through hormone release and inflammation, cellular stress also happens independently from environmental factors, aging, and lifestyle choices. Resveratrol specifically targets these cellular stress pathways, providing protection that goes beyond managing emotional stress alone.
What makes resveratrol's stress protection mechanism unique?
Resveratrol's uniqueness lies in its multi-target approach. The recent discovery shows it binds to tRNA synthetase with 1,000 times greater sensitivity than other targets, redirecting this enzyme to activate stress protection pathways. Additionally, resveratrol inhibits phosphodiesterase enzymes [Park et al., 2012] and activates sirtuins, [Kulkarni & Cantó, 2015] creating multiple layers of cellular defense against various types of stress.
How quickly can I expect to see benefits from resveratrol for stress protection?
While some people report increased energy and improved well-being within days, meaningful cellular stress protection typically develops over 4-12 weeks of consistent supplementation. Biomarker improvements in inflammation, oxidative stress, and metabolic health often become measurable after 8-12 weeks. Long-term benefits for age-related stress protection accumulate over months and years of regular use.
Can resveratrol help with work-related stress and burnout?
Yes, resveratrol can support your body's resilience to work-related stress by protecting cells from the oxidative damage and inflammation that chronic stress causes. While it doesn't eliminate stressful situations, resveratrol helps maintain cellular health despite stress exposure, potentially improving energy levels, mental clarity, and overall stress resilience. Combining resveratrol with stress management techniques provides important support.
What's the optimal dose of resveratrol for stress protection?
Research suggests doses ranging from 150-500mg daily provide stress-protective benefits, though individual needs vary. The discovery of highly sensitive cellular targets suggests lower doses may be effective for some people. Starting with 250mg daily and adjusting based on response is a common approach. Higher doses may benefit those with significant stress exposure or specific health goals, but consultation with healthcare providers is recommended.
Does resveratrol interact with stress medications or supplements?
Resveratrol generally has a good safety profile and may complement other stress-management approaches. It can enhance the effects of antioxidants and anti-inflammatory supplements. However, because resveratrol affects multiple cellular pathways, individuals taking medications should consult healthcare providers. Resveratrol may interact with blood thinners and certain medications metabolized by the liver.
How does age affect the need for resveratrol's stress protection?
As we age, cellular stress response systems naturally decline, making resveratrol's protection increasingly valuable. Older adults often benefit from resveratrol's ability to activate stress response pathways that become less efficient with age. The compound helps maintain youthful cellular resilience, potentially slowing age-related decline. Starting resveratrol supplementation earlier may provide preventive benefits by maintaining robust stress defenses throughout life.
Can lifestyle stress reduction replace the need for resveratrol?
While lifestyle modifications like exercise, meditation, and healthy diet are important for managing stress, they work synergistically with resveratrol rather than replacing it. Resveratrol provides targeted cellular protection that complements lifestyle approaches. Even with excellent stress management habits, environmental toxins, aging, and unavoidable stressors still challenge our cells. Resveratrol offers an additional layer of protection that lifestyle alone cannot fully provide.
What are the signs that resveratrol is effectively protecting against stress?
Effective stress protection from resveratrol often manifests as improved energy levels, better sleep quality, enhanced mental clarity, and greater resilience to daily challenges. Some people notice improved exercise recovery, stable mood, and better immune function. Laboratory markers showing reduced inflammation (like C-reactive protein), improved metabolic health, and decreased oxidative stress indicators provide objective evidence of cellular protection.
Is resveratrol's stress protection supported by human studies?
Yes, numerous human studies support resveratrol's stress-protective effects. Clinical trials have demonstrated improvements in oxidative stress markers, inflammatory indicators, cardiovascular health, and metabolic function. The recent discovery of the tRNA synthetase mechanism builds on decades of research showing resveratrol's benefits in human subjects. While more research continues, existing evidence strongly supports resveratrol's role in protecting against various forms of cellular stress.
References:
- Park, S. J., Ahmad, F., Philp, A., Baar, K., Williams, T., Luo, H., ... & Kumar, S. (2012). Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell, 148(3), 421-433. Available at: https://pubmed.ncbi.nlm.nih.gov/22304913/
- Kulkarni, S. S., & Cantó, C. (2015). The molecular targets of resveratrol. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1852(6), 1114-1123. Available at: https://pubmed.ncbi.nlm.nih.gov/25315298/
Resveratrol Guide – Part 1
The French Paradox: How Resveratrol Rewrites Health Rules
The discovery of the "French Paradox" sparked worldwide interest in resveratrol benefits. French populations traditionally enjoy rich, high-fat diets yet maintain surprisingly low rates of heart disease. Scientists investigating this phenomenon identified resveratrol in red wine as a key protective factor.
This revelation transformed our understanding of cardiovascular health. Rather than simply avoiding dietary fats, we learned that specific compounds like resveratrol could help our bodies process fats more effectively while protecting our hearts and blood vessels. The implications extend far beyond France, offering hope for anyone seeking to enjoy food while maintaining optimal health.
Cardiovascular Benefits That Change Lives
The cardiovascular benefits of resveratrol include:
- Improved blood flow through enhanced nitric oxide production
- Reduced inflammation in blood vessels and throughout the body
- Lower oxidative stress protecting delicate cardiovascular tissues
- Better cholesterol profiles with increased HDL and reduced LDL oxidation
- Enhanced heart muscle function supporting long-term cardiac health
These benefits work synergistically, creating a important shield for your cardiovascular system that grows stronger with consistent resveratrol intake.
Resveratrol vs. Other Grape Compounds: Understanding the Difference
Many people confuse resveratrol with grape seed extract or other grape-derived compounds. While all offer health benefits, resveratrol's unique molecular structure and biological activity set it apart. Unlike pterostilbenes found in grape seed extract, resveratrol specifically targets aging and metabolic pathways through SiRT1 activation.
This distinction matters because different compounds work through different mechanisms. While grape seed extract excels at antioxidant protection, resveratrol goes further by actually influencing gene expression. This genetic influence explains why resveratrol can produce such profound changes in metabolism, aging markers, and overall health.
id="optimizing-your-resveratrol-protocol-for-maximum-results">Optimizing Your Resveratrol Protocol for Maximum Results
Success with resveratrol supplementation depends on understanding how to maximize its effectiveness. Research suggests several strategies for enhancing resveratrol's benefits:
Timing Your Resveratrol Intake
- Morning doses align with your body's natural metabolic rhythms
- Pre-workout supplementation enhances exercise performance and fat burning
- Evening doses support overnight cellular repair and regeneration
- With meals improves absorption and reduces potential stomach upset
- Consistent daily timing maintains steady blood levels for optimal effects
Combining Resveratrol with Complementary Nutrients
Strategic nutrient combinations amplify resveratrol's effects:
- Quercetin enhances resveratrol absorption and stability
- Curcumin provides synergistic anti-inflammatory benefits
- Green tea extract boosts metabolic and antioxidant effects
- Omega-3 fatty acids support cardiovascular benefits
- Vitamin D enhances cellular protection and immune function
Embrace Your Health Transformation with Resveratrol
The journey to optimal health and vitality begins with understanding the tools available to support your goals. Resveratrol represents more than just another supplement – it's a scientifically validated pathway to enhanced metabolism, graceful aging, and improved quality of life.
As research continues unveiling resveratrol's remarkable properties, one thing becomes clear: this powerful compound offers genuine hope for those seeking natural solutions to modern health challenges. Whether your goals include weight management, cardiovascular protection, or simply maintaining youthful energy, resveratrol provides a proven foundation for success.
Take the first step today toward a healthier, more vibrant future. With resveratrol as your ally, the possibilities for transformation are limitless. Your body possesses incredible capacity for renewal and optimization – resveratrol simply unlocks that potential, allowing you to experience life with renewed energy, clarity, and joy.
References
Alzheimer and 58 Resveratrol Trials
Resveratrol Research: Breakthrough Clinical Trials Show Promise for Alzheimer's Treatment
Resveratrol stands at the forefront of modern medical research, with 58 human trials currently registered with the National Institute of Health (NIH). This powerful compound has captured the attention of scientists worldwide, particularly for its potential to combat devastating neurodegenerative conditions. As research advances from laboratory studies to human clinical trials, the scientific community is witnessing unprecedented opportunities for treating conditions once thought irreversible.
Phase 2 Alzheimer's Trial: A Milestone in Resveratrol Research
Among the most anticipated studies is a Phase 2 clinical trial specifically designed to evaluate resveratrol as a treatment for Alzheimer's disease. This groundbreaking research builds upon years of preclinical studies that have consistently demonstrated the compound's neuroprotective properties. The trial represents a critical step in translating laboratory successes into real-world therapeutic applications.
The decision to advance to Phase 2 trials wasn't made lightly. Researchers have accumulated substantial evidence from animal models and preliminary human studies suggesting that resveratrol could significantly impact the progression of Alzheimer's disease. This progression from bench to bedside exemplifies the rigorous scientific process required to develop new treatments.
Foundation of Scientific Evidence
The scientific foundation supporting these human trials comes from numerous peer-reviewed studies, including groundbreaking research published in the journal Age (November 7, 2012). This important study utilized genetically modified mice programmed to develop Alzheimer's disease at the molecular level, complete with the characteristic genetic mutations and abnormal protein accumulations seen in human patients.
The results were nothing short of remarkable. Long-term resveratrol treatment produced a 33% increase in mean life expectancy and an astounding 20% extension in maximum lifespan compared to untreated control mice. These statistics represent more than just numbers; they signify the potential for meaningful therapeutic intervention in a disease that affects millions worldwide.
How Resveratrol Works Against Alzheimer's Disease
Understanding the mechanisms behind resveratrol's effectiveness reveals why researchers are so optimistic about its therapeutic potential. The compound operates through multiple pathways to protect brain cells and potentially reverse damage associated with neurodegenerative diseases.
Reducing Amyloid Burden
One of the hallmark features of Alzheimer's disease is the accumulation of amyloid plaques in the brain. These protein deposits disrupt normal cellular function and contribute to cognitive decline. Resveratrol has demonstrated a remarkable ability to reduce this amyloid burden, essentially clearing away these toxic accumulations and allowing brain cells to function more normally.
The process by which resveratrol achieves this clearing effect involves activating cellular cleanup mechanisms. Think of it as enhancing the brain's natural waste disposal system, enabling it to more efficiently remove harmful proteins before they can form damaging plaques.
Cognitive Enhancement and Neuroprotection
Beyond plaque reduction, resveratrol actively protects neurons from damage and death. The compound strengthens cellular defenses against oxidative stress, inflammation, and other factors that contribute to brain cell deterioration. This neuroprotective effect translates into measurable improvements in cognitive function, as demonstrated in multiple research models.
Patients and their families often report improved memory, clearer thinking, and better overall mental function when resveratrol supplementation is incorporated into treatment plans. While individual results vary, the consistency of positive outcomes across different studies provides compelling evidence for its effectiveness.
The Science Behind Resveratrol's Success
The molecular mechanisms underlying resveratrol's benefits extend far beyond simple antioxidant activity. Research has identified several key pathways through which this compound exerts its protective effects:
Sirtuin Activation
Resveratrol activates sirtuins, a family of proteins often called "longevity genes." These proteins regulate cellular health, DNA repair, and metabolic processes. By enhancing sirtuin activity, resveratrol essentially turns on the body's natural defense and repair mechanisms.
Mitochondrial Function Enhancement
Cellular energy production depends on healthy mitochondria. Resveratrol improves mitochondrial function, ensuring brain cells have adequate energy to perform their complex tasks. This energy boost is particularly important for neurons, which have exceptionally high energy demands.
Anti-Inflammatory Effects
Chronic inflammation plays a significant role in Alzheimer's progression. Resveratrol possesses potent anti-inflammatory properties, helping to reduce the harmful inflammatory processes that damage brain tissue over time.
Beyond Alzheimer's: Resveratrol's Broader Applications
While the Alzheimer's trials capture significant attention, resveratrol research extends into numerous other health conditions. Clinical trials are investigating its potential for:
- Cardiovascular disease prevention: Studies show resveratrol improves heart health by reducing inflammation, lowering blood pressure, and preventing arterial damage.
- Diabetes management: Research indicates the compound helps regulate blood sugar levels and improves insulin sensitivity.
- Cancer prevention and treatment: Multiple trials explore how resveratrol might prevent cancer development or enhance treatment effectiveness.
- Age-related cognitive decline: Beyond Alzheimer's, studies examine its role in maintaining general brain health during aging.
- Metabolic syndrome: Trials investigate its ability to address multiple metabolic issues simultaneously.
Participating in Resveratrol Clinical Trials
For those interested in contributing to this groundbreaking research, many resveratrol trials actively recruit volunteers. Participation offers several benefits:
- Access to important treatments before general availability
- Close monitoring by medical professionals
- Contributing to scientific knowledge that could help millions
- Potential health improvements from experimental treatments
Clinical trial participation requires careful consideration and consultation with healthcare providers. However, for suitable candidates, it represents an opportunity to access promising treatments while advancing medical science.
Frequently Asked Questions About Resveratrol Clinical Trials
What exactly is resveratrol and why is it being studied so extensively?
Resveratrol is a natural polyphenol compound found in grapes, berries, and other plants. Scientists study it extensively because research shows it activates longevity pathways, reduces inflammation, protects brain cells, and offers potential treatment for conditions like Alzheimer's disease, heart disease, and diabetes. Its ability to work through multiple biological mechanisms makes it uniquely promising for treating complex health conditions.
How can I participate in a resveratrol clinical trial?
To participate in resveratrol clinical trials, start by visiting ClinicalTrials.gov and searching for studies in your area. Contact your healthcare provider to discuss whether you meet eligibility criteria. Many trials seek volunteers with specific conditions or risk factors. The research team will conduct screening tests to ensure your safety and determine if you qualify for participation.
What were the specific results of the resveratrol Alzheimer's mouse study?
The landmark study published in Age journal showed that resveratrol treatment increased mean life expectancy by 33% and maximum lifespan by 20% in Alzheimer's mouse models. Additionally, treated mice showed significantly reduced cognitive impairment, decreased amyloid plaque formation, and improved overall brain health markers. These results provided important evidence supporting human clinical trials.
Are there any risks associated with taking resveratrol?
Resveratrol is generally well-tolerated, but like any supplement, it can have side effects. Some people experience mild digestive issues at high doses. It may interact with blood-thinning medications and certain other drugs. Always consult your healthcare provider before starting supplementation, especially if you have existing health conditions or take medications.
How long before we see resveratrol approved as an Alzheimer's treatment?
The timeline for resveratrol approval depends on clinical trial outcomes. Phase 2 trials typically take 2-3 years, followed by Phase 3 trials lasting 3-4 years. If results remain positive, FDA approval could come within 5-10 years. However, many people already use resveratrol supplements for general health benefits while formal approval processes continue.
What dosage of resveratrol is typically used in clinical trials?
Clinical trial dosages of resveratrol vary significantly depending on the condition being studied. Alzheimer's trials often use doses ranging from 500mg to 2000mg daily. Cardiovascular studies might use different amounts. These doses are much higher than what you'd get from dietary sources alone, which is why supplementation is often necessary for therapeutic effects.
Can resveratrol prevent Alzheimer's disease in healthy individuals?
While definitive prevention studies are ongoing, existing research suggests resveratrol may help reduce Alzheimer's risk. Its neuroprotective properties, ability to reduce inflammation, and positive effects on brain health markers indicate preventive potential. Many researchers recommend it as part of a brain-healthy lifestyle, though more long-term studies are needed for conclusive prevention claims.
How does resveratrol compare to current Alzheimer's medications?
Resveratrol works differently than conventional Alzheimer's drugs. While current medications primarily manage symptoms, resveratrol targets underlying disease mechanisms like amyloid accumulation and neuroinflammation. This fundamental approach could potentially slow or halt disease progression rather than just treating symptoms, making it a promising complementary or alternative treatment option.
What other conditions are being studied in resveratrol clinical trials?
Beyond Alzheimer's, resveratrol clinical trials investigate numerous conditions including type 2 diabetes, cardiovascular disease, various cancers, metabolic syndrome, fatty liver disease, and age-related macular degeneration. Researchers also study its effects on healthy aging, exercise performance, and cognitive function in non-diseased populations.
Where can I find reliable resveratrol supplements while trials continue?
Quality resveratrol supplements are available from reputable manufacturers who follow good manufacturing practices. Look for products with third-party testing, clear labeling of trans-resveratrol content, and companies that specialize in research-based formulations. Leading suppliers offer pharmaceutical-grade resveratrol similar to what's used in clinical trials.
Resveratrol & Antioxidants
important Health Benefits of Resveratrol
The scope of resveratrol's health benefits continues to expand as research unveils new therapeutic applications. This versatile compound offers protection across multiple body systems, making it a valuable addition to any wellness regimen.
1. Powerful Anti-Inflammatory Properties
Chronic inflammation underlies many modern health challenges. Resveratrol's potent anti-inflammatory effects help calm systemic inflammation, potentially reducing the risk of various inflammatory conditions. This makes it particularly beneficial for those dealing with arthritis, skin inflammation, or general inflammatory aging (inflammaging).
2. Cancer Prevention and Support
Emerging research suggests that resveratrol may inhibit tumor growth by supporting the immune system's ability to identify and eliminate cancer cells. Its antioxidant properties help protect DNA from damage that could lead to cancerous mutations, while its anti-inflammatory effects create an environment less favorable to cancer development.
3. Cardiovascular Protection
Beyond the French Paradox, resveratrol offers multiple cardiovascular benefits:
- Reduces LDL oxidation, preventing arterial plaque formation
- Improves endothelial function for better blood flow
- Helps regulate blood pressure
- Supports healthy cholesterol levels
- Reduces blood clot formation risk
4. Brain Health and Neuroprotection
Resveratrol crosses the blood-brain barrier , providing direct neuroprotective benefits. Studies indicate it may help protect against Alzheimer's disease, Parkinson's disease, and age-related cognitive decline. By reducing brain inflammation and oxidative stress, resveratrol supports mental clarity and memory retention.
5. Anti-Aging and Longevity Enhancement
Perhaps most exciting are resveratrol's anti-aging benefits. Research has demonstrated up to 50% lifespan extension in certain animal models [Bhullar & Hubbard, 2015]. While human studies continue, the mechanisms behind these effects suggest significant potential for promoting healthy aging and longevity in humans.
6. Metabolic Support and Weight Management
Resveratrol activates metabolic pathways that improve insulin sensitivity and glucose metabolism [Mendez-del Villar et al., 2014]. This can support healthy weight management, reduce diabetes risk, and enhance overall metabolic health.
How to Maximize Resveratrol Benefits in Your Daily Life
Incorporating resveratrol into your wellness routine doesn't require dramatic lifestyle changes. Simple, sustainable strategies can help you harness its powerful benefits:
Dietary Integration
- Enjoy Red Wine Mindfully: Limit intake to 1-2 glasses daily for optimal benefits without negative effects
- Snack on Red Grapes: Choose organic varieties with seeds for maximum resveratrol content
- Add Berries to Meals: Incorporate blueberries, cranberries, and mulberries into breakfast or snacks
- Include Peanuts: A handful of peanuts provides resveratrol along with healthy fats and protein
Supplementation Strategies
For those seeking concentrated benefits, high-quality resveratrol supplements offer consistent, therapeutic doses. When choosing supplements, look for:
- Trans-resveratrol form (the most bioactive)
- Third-party testing for purity
- Appropriate dosing (typically 250-500mg daily)
- Synergistic ingredients that enhance absorption
The Science Behind Resveratrol's Effectiveness
Understanding why resveratrol works so effectively helps appreciate its value. At the cellular level, this compound:
- Activates SIRT1: The "longevity gene" that regulates cellular health and lifespan [Price et al., 2012]
- Stimulates AMPK: A metabolic master switch that enhances energy production [Price et al., 2012]
- Reduces NF-κB Activity: Decreasing inflammation throughout the body [Zu et al., 2016]
- Enhances Mitochondrial Function: Improving cellular energy production [Price et al., 2012]
- Promotes Autophagy: The cellular cleaning process that removes damaged components
New Resveratrol Studies, Highlight Its Potential
Resveratrol Studies: important Research on Health Benefits and Scientific Findings
Resveratrol studies have captivated the scientific community for decades, revealing remarkable potential across multiple areas of human health. This polyphenolic compound, naturally occurring in grapes, berries, and other plants, has emerged as a promising subject of research with applications ranging from immune system support to cardiovascular protection. Whether you're a health enthusiast exploring natural compounds or someone seeking evidence-based wellness solutions, understanding the latest resveratrol studies can open doors to transformative health possibilities.
Understanding Resveratrol: What Scientific Studies Reveal
Recent resveratrol studies have uncovered fascinating mechanisms through which this compound interacts with our bodies at the cellular level. Found abundantly in red grape skins, Japanese knotweed, and peanuts, resveratrol belongs to a class of compounds called stilbenes. Research indicates that its molecular structure allows it to activate specific proteins involved in cellular health and longevity, particularly sirtuins, which play important roles in aging processes [Price et al., 2012].
Scientists conducting resveratrol studies have identified its ability to cross the blood-brain barrier, interact with multiple cellular pathways, and demonstrate both antioxidant and anti-inflammatory properties. These characteristics make it a versatile compound worthy of extensive investigation across various health domains.
Breakthrough Resveratrol Studies on Immune System Regulation
Groundbreaking research published in Frontiers in Immunology has shed light on resveratrol's sophisticated immunomodulatory effects. This important study examined how resveratrol influences key immune system components, particularly macrophages and dendritic cells.
Building on these discoveries, a comprehensive 2024 review in Frontiers in Pharmacology revealed exactly how resveratrol drives macrophage polarization, helping to shift the immune system away from a chronic pro-inflammatory state toward a tissue-repairing, anti-inflammatory state [Shao et al., 2024]. Additionally, emerging 2024 research indicates that resveratrol holds massive promise as a therapeutic intervention for autoimmune diseases by modulating SIRT pathways, which actively suppresses the release of inflammatory mediators and limits tissue damage in autoimmune conditions [Zhang et al., 2024].
The findings suggest that resveratrol helps maintain immune balance by:
- Modulating inflammatory responses without suppressing beneficial immune functions
- Supporting the body's natural defense mechanisms against oxidative stress
- Promoting healthy communication between immune cells
- Helping regulate cytokine production for optimal immune response
These resveratrol studies indicate that regular supplementation might support individuals dealing with immune challenges or those simply wanting to maintain robust immune health throughout their lives.
Cardiovascular Health and Resveratrol: Evidence from Clinical Studies
The connection between resveratrol and heart health has been extensively documented in scientific literature. The famous "French Paradox" – the observation that French populations have relatively low rates of heart disease despite consuming rich foods – sparked initial interest in resveratrol studies focusing on cardiovascular benefits [Renaud & de Lorgeril, 1992].
A important analysis published in a recent medical textbook consolidates decades of research showing that resveratrol supports cardiovascular health through multiple mechanisms:
- Improved endothelial function: Resveratrol helps blood vessels maintain flexibility and proper dilation
- Reduced LDL oxidation: Studies show decreased oxidation of harmful cholesterol particles
- Anti-platelet effects: The compound may help prevent excessive blood clotting
- Blood pressure regulation: Some studies indicate modest reductions in blood pressure
- Enhanced mitochondrial function: Supporting cellular energy production in heart muscle
RevGenetics: Pioneering Excellence in Resveratrol Research and Supplementation
Since 2007, RevGenetics has established itself as a trusted leader in resveratrol research and high-quality supplementation. The company recognized early on the transformative potential revealed by resveratrol studies and committed to providing research-grade supplements to universities, research institutions, and health-conscious individuals worldwide.
RevGenetics' 500 mg resveratrol capsules represent the gold standard in purity and potency, formulated based on the latest scientific findings. Their dedication to quality ensures that each capsule delivers the therapeutic doses shown effective in clinical studies, making the benefits of resveratrol accessible to everyone seeking optimal health.
Frequently Asked Questions About Resveratrol Studies
What do resveratrol studies say about the best time to take supplements?
Research indicates that resveratrol can be taken at any time of day, but many studies suggest taking it with a meal containing healthy fats can enhance absorption. Some individuals prefer morning doses for energy support, while others take it in the evening for its potential sleep quality benefits. The key is consistency in timing to maintain steady blood levels.
How long does it take to see results from resveratrol according to studies?
Clinical studies show that some benefits, such as improved antioxidant status, can occur within days to weeks. However, more significant effects on cardiovascular health, cognitive function, or metabolic parameters typically require 8-12 weeks of consistent supplementation. Long-term studies suggest that continued use provides cumulative benefits over months and years.
Are there any interactions between resveratrol and medications that studies have identified?
Research has identified potential interactions with blood-thinning medications, as resveratrol may enhance their effects. Studies also suggest possible interactions with certain medications metabolized by liver enzymes. Always consult with a healthcare provider before starting resveratrol supplementation, especially if you take prescription medications.
What do resveratrol studies reveal about its safety profile?
Extensive safety studies have shown that resveratrol is well-tolerated by most individuals at doses up to 1000 mg daily. Some studies using very high doses (above 2000 mg) reported mild gastrointestinal effects in some participants. The compound has been granted Generally Recognized as Safe (GRAS) status for use in foods and supplements.
How do resveratrol studies compare natural food sources versus supplements?
While foods like red grapes and berries contain resveratrol, studies show that achieving therapeutic doses through diet alone is challenging. For example, you would need to consume hundreds of glasses of red wine to match the resveratrol content in a single 500 mg supplement. Research supports supplementation as the most practical way to obtain clinically relevant doses.
What age groups benefit most from resveratrol according to studies?
Research shows benefits across all adult age groups, but studies particularly highlight advantages for individuals over 40 when natural antioxidant defenses begin declining. Young adults may benefit from its exercise recovery and metabolic support properties, while older adults often experience more pronounced benefits for cardiovascular and cognitive health.
Do resveratrol studies support its use for athletic performance?
Emerging research suggests resveratrol may enhance exercise performance by improving mitochondrial function and reducing exercise-induced oxidative stress. Studies have shown benefits including improved endurance, faster recovery times, and reduced muscle damage markers. Athletes particularly value its anti-inflammatory properties for supporting intense training regimens.
What manufacturing standards should I look for based on resveratrol studies?
Studies emphasize the importance of purity and standardization in resveratrol supplements. Look for products that specify trans-resveratrol content (the active form), third-party testing for purity, and manufacturing in GMP-certified facilities. Companies like RevGenetics that supply research institutions typically maintain the highest quality standards.
How do resveratrol studies address concerns about bioavailability?
Research has identified several strategies to enhance resveratrol bioavailability, including micronization, combination with piperine (black pepper extract), and lipid-based delivery systems. Studies show that despite relatively low bioavailability, resveratrol still produces significant biological effects, possibly due to its metabolites and interactions with gut microbiota.
What do long-term resveratrol studies reveal about sustained use?
Long-term studies spanning several years have shown that continuous resveratrol supplementation maintains its beneficial effects without tolerance development. Research indicates that prolonged use may provide cumulative benefits for healthy aging, with some studies showing improved markers of biological age. Safety profiles remain excellent even with years of consistent use.
The wealth of scientific evidence from resveratrol studies continues to grow, painting an increasingly clear picture of its potential to support human health across multiple dimensions. From immune system modulation to neuroprotection, cardiovascular support to skin health, resveratrol stands out as a compound worthy of attention for anyone committed to optimizing their health and longevity. With trusted suppliers like RevGenetics making research-grade resveratrol accessible, there has never been a better time to incorporate this remarkable compound into your wellness routine.
New Resveratrol Section With Archives
Ready to try resveratrol yourself?
RevGenetics carries pharmaceutical-grade micronized trans-resveratrol -- 98%+ purity, third-party tested.
Shop Resveratrol Supplements →Archives: Your Complete Guide to Organized Information Management
Archives represent the cornerstone of information preservation and knowledge management. Whether you're managing personal documents, corporate records, or digital content, understanding how archives work transforms chaos into clarity. This in-depth guide explores everything about archives, from basic principles to advanced strategies that help you build systems that last.
What Are Archives and Why Do They Matter?
Archives serve as organized collections of records, documents, and materials preserved for their historical, legal, or informational value. Unlike simple storage, archives follow specific principles that ensure information remains accessible, authentic, and meaningful over time. Organizations worldwide rely on archives to maintain institutional memory, comply with regulations, and support decision-making.
The power of well-maintained archives extends beyond mere storage. They create pathways to understanding past decisions, tracking progress, and building upon previous knowledge. When you implement effective archive systems, you important the ability to retrieve critical information instantly, protect valuable assets, and create lasting value for future generations.
Essential Components of Modern Archives
Physical Archives
Traditional physical archives remain vital for preserving original documents, photographs, and artifacts. These tangible collections require specific environmental controls, including temperature regulation, humidity management, and protection from light damage. Successful physical archives incorporate:
- Climate-controlled storage spaces that maintain optimal preservation conditions
- Acid-free folders and boxes preventing chemical deterioration
- Detailed cataloging systems enabling quick location of specific items
- Security measures protecting valuable or sensitive materials
- Disaster recovery plans safeguarding against unexpected events
Digital Archives
Digital archives revolutionize how we store and access information. These electronic repositories offer unprecedented search capabilities, remote access, and space efficiency. Key elements of digital archives include:
- Metadata standards ensuring consistent documentation
- File format specifications promoting long-term accessibility
- Backup protocols protecting against data loss
- Access controls managing user permissions
- Migration strategies adapting to technological changes
Building Your Archive Strategy
Creating effective archives requires thoughtful planning and systematic implementation. Start by assessing your current information landscape and identifying materials worth preserving. Consider both immediate needs and long-term goals as you develop your archival framework.
Step 1: Conduct an Information Audit
Begin your archive journey by cataloging existing materials. Document what you have, where it's stored, and its current condition. This audit reveals gaps, redundancies, and opportunities for improvement. Pay special attention to unique or irreplaceable items that deserve priority treatment.
Step 2: Establish Selection Criteria
Not everything belongs in archives. Develop clear criteria for determining what to preserve. Consider factors like historical significance, legal requirements, research value, and organizational importance. Your selection policy guides consistent decision-making and prevents archive bloat.
Step 3: Design Your Classification System
Organize archives using logical, scalable classification schemes. Whether you choose alphabetical, chronological, or subject-based systems, consistency matters most. Create hierarchical structures that accommodate growth while maintaining navigability.
Step 4: Implement Preservation Measures
Protect archived materials through appropriate preservation techniques. Physical items need proper housing and environmental controls. Digital files require regular integrity checks and format migrations. Budget for ongoing preservation activities that maintain archive viability.
Step 5: Create Access Protocols
Balance preservation needs with user access. Develop policies governing who can access archives, under what conditions, and through which channels. Consider creating different access levels for various user groups while maintaining security and preservation standards.
Archives Best Practices for Different Industries
Corporate Archives
Businesses important archives to preserve institutional knowledge, support legal compliance, and maintain brand heritage. Corporate archives typically include financial records, strategic documents, product development files, and marketing materials. These collections support decision-making by providing historical context and precedent.
Academic Archives
Educational institutions maintain archives documenting scholarly research, institutional history, and student life. These repositories support teaching, learning, and research while preserving unique collections that attract scholars worldwide. Academic archives often include manuscripts, research data, administrative records, and special collections.
Government Archives
Public sector archives ensure transparency, accountability, and citizen access to government information. These repositories preserve legislative records, administrative documents, and historical materials that document governmental activities and societal development.
Personal Archives
Individuals create archives to preserve family history, creative works, and personal documentation. These intimate collections capture life stories, genealogical information, and cultural heritage for future generations. Personal archives might include photographs, letters, journals, and digital memories.
Technology Transforming Archives
Emerging technologies revolutionize how archives operate and serve users. Artificial intelligence enables automated metadata generation and content analysis. Machine learning improves search capabilities and pattern recognition. Blockchain technology offers new possibilities for ensuring authenticity and tracking provenance.
Digitization Initiatives
Converting physical materials to digital formats expands access while preserving originals. Modern scanning equipment captures high-resolution images that reveal details invisible to the naked eye. Optical character recognition transforms printed text into searchable data, unlocking new research possibilities.
Cloud Storage Solutions
Cloud-based archives provide scalable storage, enhanced security, and global accessibility. These platforms offer automated backup, version control, and collaboration features that traditional storage cannot match. However, careful vendor selection and data governance remain essential.
Virtual Reality Applications
Immersive technologies create new ways to experience archival materials. Virtual exhibitions bring collections to life, while augmented reality overlays historical information onto physical spaces. These innovations make archives more engaging and accessible to diverse audiences.
Common Archive Challenges and Solutions
Space Limitations
Physical storage constraints challenge growing archives. Solutions include off-site storage facilities, collaborative repositories, and selective digitization programs. Compact shelving systems and vertical storage maximize available space while maintaining accessibility.
Budget Constraints
Limited funding affects preservation activities and technological upgrades. Creative approaches include grant writing, partnership development, and volunteer programs. Prioritizing high-value materials and phased implementation helps stretch resources further.
Staff Expertise
Specialized knowledge requirements challenge smaller archives. Professional development programs, mentorship relationships, and collaborative networks build necessary skills. Online training resources and professional associations provide ongoing education opportunities.
Format Obsolescence
Technological change threatens digital archive accessibility. Migration strategies, emulation software, and format registries help maintain usability. Regular technology assessments identify at-risk formats requiring intervention.
Future of Archives
Archives continue evolving to meet changing user expectations and technological possibilities. Increased emphasis on user experience design makes archives more intuitive and engaging. Community partnerships expand collection scope and cultural representation. Sustainability initiatives address environmental impacts while ensuring long-term preservation.
The integration of archives with other information systems creates seamless research environments. Linked data initiatives connect dispersed collections, revealing new relationships and insights. Mobile applications bring archives directly to users, supporting location-based experiences and personal research.
Archives Resources and Tools
Successful archive management relies on appropriate tools and resources. Collection management systems streamline cataloging and tracking. Preservation supplies protect materials from deterioration. Reference management software organizes research and citations. Digital asset management platforms handle large-scale digital collections.
Professional standards guide consistent practice across institutions. Descriptive standards ensure interoperability and resource sharing. Preservation guidelines inform conservation decisions. Access policies balance openness with protection. These frameworks provide tested approaches that improve outcomes.
Educational resources support ongoing learning and skill development. Webinars address emerging topics and technologies. Conferences facilitate knowledge exchange and networking. Publications share research findings and case studies. Certification programs validate professional competencies.
Measuring Archive Success
Effective archives demonstrate value through measurable outcomes. Usage statistics track researcher engagement and collection relevance. Preservation assessments monitor collection condition and intervention effectiveness. User satisfaction surveys identify service improvements. Cost-benefit analyses justify resource allocation.
Success metrics vary by archive type and mission. Academic archives might prioritize research citations and student engagement. Corporate archives could focus on decision support and brand heritage preservation. Personal archives value family connections and memory preservation. Choose metrics aligned with your specific goals and stakeholder needs.
Regular evaluation drives continuous improvement. Annual reviews assess progress against objectives. Stakeholder feedback identifies emerging needs. Environmental scanning reveals trends affecting future operations. This ongoing assessment ensures archives remain relevant and valuable.
Frequently Asked Questions About Archives
What makes archives different from regular storage?
Archives go beyond simple storage by implementing systematic organization, preservation standards, and access protocols. While storage simply holds items, archives actively manage materials to ensure long-term preservation, authentic documentation, and meaningful access. Archives follow professional standards for arrangement, description, and preservation that maintain both physical integrity and intellectual context. This systematic approach enables researchers to find relevant materials efficiently while ensuring information remains accessible for future generations.
How do I start creating archives for my organization?
Begin your organizational archives by conducting a thorough assessment of existing materials and identifying preservation priorities. Start with a pilot project focusing on high-value materials that demonstrate clear benefits. Develop basic policies covering selection criteria, access guidelines, and preservation standards. Invest in appropriate storage supplies and environmental controls. Create simple but consistent cataloging procedures that can scale with growth. Connect with professional archive organizations for guidance and training opportunities. Remember that successful archives develop incrementally, so focus on establishing solid foundations before expanding scope.
What are the costs associated with maintaining archives?
Archive costs include initial setup expenses and ongoing operational requirements. Initial costs cover storage equipment, preservation supplies, cataloging systems, and environmental controls. Ongoing expenses include staff time, utilities for climate control, preservation treatments, technology updates, and access services. Digital archives require server space, backup systems, and periodic format migrations. However, archives often generate cost savings by improving information retrieval efficiency, reducing duplication, and supporting better decision-making. Many organizations find that archive benefits far outweigh costs when considering risk mitigation, compliance support, and knowledge preservation value.
How do digital archives ensure long-term accessibility?
Digital archives maintain long-term accessibility through multiple strategies including format standardization, regular migration, and important documentation. Archives use open, well-documented file formats that resist obsolescence. Regular integrity checking ensures files remain uncorrupted. Migration programs convert files to current formats before old ones become inaccessible. Detailed metadata preserves context and technical requirements. Multiple backup copies in different locations protect against data loss. Some archives also maintain legacy hardware and software for accessing obsolete formats. These combined approaches create resilient systems that preserve digital information across technological changes.
What skills do archive professionals need?
Archive professionals combine technical expertise with strong organizational and communication abilities. Core competencies include understanding preservation principles, cataloging standards, and access systems. Digital archives require technology skills including database management, digital preservation, and metadata standards. Professionals need research skills to understand collection contexts and user needs. Communication abilities support reference services, instruction, and advocacy. Project management skills enable successful initiatives and resource optimization. Many archivists also develop subject expertise relevant to their collections. Continuous learning remains essential as technologies and professional standards evolve.
How can archives support research and education?
Archives provide primary source materials that enable original research and authentic learning experiences. Researchers use archives to uncover new information, verify facts, and develop fresh interpretations. Students engage with historical documents that bring subjects to life beyond textbook summaries. Archives support dissertation research, scholarly publications, and creative projects. Educational programs include class visits, workshops, and online exhibitions that develop critical thinking and research skills. Digital archives expand access through online catalogs, digitized collections, and virtual reference services. These resources transform learning by connecting users directly with historical evidence and cultural heritage.
What role do archives play in preserving cultural heritage?
Archives serve as cultural memory institutions that preserve diverse voices, experiences, and traditions for future generations. They document community histories, artistic expressions, and social movements that might otherwise disappear. Archives collect materials representing different cultural groups, ensuring inclusive historical records. These collections support cultural identity, genealogical research, and community pride. Archives also preserve intangible heritage through oral histories, performance recordings, and cultural practice documentation. By making these materials accessible, archives enable cultural transmission, scholarly research, and public understanding. This preservation work strengthens communities and enriches our collective human story.
How do archives handle sensitive or restricted materials?
Archives balance preservation responsibilities with privacy, security, and ethical considerations through carefully developed access policies. Restricted materials might include personal information, confidential business records, or culturally sensitive items. Archives implement tiered access systems where different user groups receive appropriate permissions. Time-based restrictions allow materials to become available after privacy concerns diminish. Redaction procedures protect sensitive information while enabling partial access. Some archives require research agreements or ethics reviews for sensitive collections. Staff receive training on handling confidential materials and maintaining security protocols. These measures ensure archives fulfill preservation missions while respecting privacy rights and cultural protocols.
What are the career opportunities in archives?
Archive careers offer diverse opportunities across multiple sectors and specializations. Traditional positions include archivists, preservation specialists, and reference librarians in cultural institutions, corporations, and government agencies. Digital archives create roles for metadata specialists, digital preservation managers, and systems administrators. Specialized positions focus on specific formats like audiovisual materials, photographs, or electronic records. Consulting opportunities serve organizations developing archive programs. Education roles include teaching archive studies and conducting professional workshops. Many professionals combine archive work with related fields like records management, museum studies, or digital humanities. Career advancement leads to management positions, subject specializations, or independent consulting practices.
How are artificial intelligence and machine learning impacting archives?
Artificial intelligence and machine learning technologies transform archive operations through automated processing, enhanced discovery, and predictive preservation. AI-powered transcription converts handwritten documents and audio recordings into searchable text. Machine learning algorithms identify patterns in large collections, revealing hidden connections and research opportunities. Computer vision technology analyzes photographs and films, generating descriptive metadata automatically. Natural language processing improves search interfaces, understanding user intent beyond keyword matching. Predictive models identify at-risk materials requiring preservation intervention. These technologies don't replace human expertise but amplify capabilities, allowing archivists to focus on complex tasks requiring judgment and interpretation while automation handles routine processing.
For specific archival information about resveratrol research and developments, visit the dedicated Resveratrol Archives section, which contains important historical documentation and research updates in this specialized field.
Resveratrol Market Developments, Trends, and Opportunities
Resveratrol Market Analysis: important Guide to Growth Opportunities and Industry Trends
The Resveratrol Market represents one of the most dynamic segments in the global dietary supplement industry, offering exceptional opportunities for brands and entrepreneurs seeking to capitalize on consumer demand for natural antioxidants. This important analysis explores market dynamics, growth drivers, and strategic insights that position resveratrol supplements as a cornerstone of modern wellness portfolios.
Resveratrol Market Size and Growth Projections
The global Resveratrol Market demonstrates robust expansion, with compound annual growth rates exceeding industry averages for dietary supplements. Market analysts project continued acceleration driven by several converging factors:
Market Valuation and Forecast
Current market valuations indicate the resveratrol supplement sector has reached significant milestones, with projections suggesting doubling of market size within the next five years. This growth trajectory reflects increasing consumer sophistication and preference for evidence-based nutritional solutions.
Regional Market Dynamics
The Resveratrol Market exhibits distinct regional characteristics that create unique opportunities for targeted market entry and expansion:
North American Market Leadership
The United States dominates global resveratrol consumption, driven by advanced supplement infrastructure, consumer education, and disposable income levels. American consumers demonstrate particular interest in premium formulations and clinically validated products, creating opportunities for differentiated market positioning.
European Market Sophistication
European markets prioritize quality certifications and sustainable sourcing, with consumers willing to pay premium prices for organic and ethically produced resveratrol supplements. This creates opportunities for brands emphasizing transparency and environmental responsibility.
Asia-Pacific Emerging Opportunities
Rapid economic growth and increasing health awareness in Asia-Pacific regions present exceptional expansion potential for the Resveratrol Market. Traditional medicine integration and growing middle-class populations create favorable conditions for market penetration.
Consumer Demographics and Purchasing Behaviors
Understanding target demographics enables effective market positioning and product development within the resveratrol supplement category:
Primary Consumer Segments
| Demographic | Key Motivations | Product Preferences |
|---|---|---|
| Health-Conscious Adults (35-55) | Preventive health, energy support | High-potency formulations |
| Active Aging Population (55+) | Longevity, cognitive support | Combination formulas |
| Fitness Enthusiasts | Recovery, performance | Sports-specific blends |
| Beauty-Focused Consumers | Skin health, anti-aging | Beauty-from-within products |
Innovation Trends Shaping the Resveratrol Market
Technological advancement and formulation innovation continue transforming the Resveratrol Market, creating opportunities for differentiation and premium positioning:
Bioavailability Enhancement Technologies
Advanced delivery systems including liposomal encapsulation, micronization, and novel carrier systems dramatically improve resveratrol absorption, addressing historical limitations and enhancing consumer satisfaction. These innovations command premium pricing while delivering superior results.
Synergistic Formulation Strategies
Combining resveratrol with complementary ingredients such as quercetin, pterostilbene, and NAD+ precursors creates important anti-aging formulations that appeal to sophisticated consumers seeking maximum benefits from their supplement regimens.
Market Entry Strategies for Success
Entering or expanding within the Resveratrol Market requires strategic planning and differentiation. Consider these proven approaches:
1. Quality Differentiation
Source premium raw materials and implement rigorous quality control measures. Third-party certifications and transparency build consumer trust and justify premium pricing strategies.
2. Education-Based Marketing
Develop important educational content explaining resveratrol benefits, dosing recommendations, and scientific research. Informed consumers demonstrate higher loyalty and lifetime value.
3. Targeted Product Development
Create specialized formulations addressing specific consumer needs such as cardiovascular support, healthy aging, or athletic performance. Niche positioning reduces direct competition while commanding premium prices.
4. Strategic Partnership Development
Collaborate with healthcare practitioners, fitness professionals, and wellness influencers to build credibility and expand market reach through trusted recommendations.
Supply Chain Considerations for Resveratrol Market Success
Sustainable and reliable supply chains form the foundation of successful resveratrol supplement businesses:
Raw Material Sourcing
Japanese knotweed remains the primary commercial source for resveratrol extraction, offering consistent potency and cost-effectiveness. Establishing relationships with reliable suppliers ensures product consistency and competitive pricing.
Quality Assurance Protocols
Implement important testing programs including identity verification, potency analysis, and contaminant screening. Documentation and traceability satisfy regulatory requirements while building consumer confidence.
Regulatory Landscape and Compliance
Navigating regulatory requirements ensures sustainable growth within the Resveratrol Market:
FDA Compliance (United States)
Adhere to Good Manufacturing Practices (GMP) and ensure accurate labeling and claims substantiation. Avoid disease treatment claims while emphasizing structure-function benefits supported by scientific evidence.
International Market Considerations
Research specific requirements for target markets including registration procedures, permitted claims, and labeling requirements. Proactive compliance prevents costly delays and market entry barriers.
Marketing Strategies for Resveratrol Market Dominance
Effective marketing drives brand recognition and sales growth within competitive supplement markets:
Digital Marketing Excellence
- Content Marketing: Develop authoritative content addressing consumer questions and concerns
- Social Media Engagement: Build communities around healthy aging and wellness lifestyles
- Influencer Partnerships: Collaborate with trusted voices in health and wellness spaces
- Email Marketing: Nurture customer relationships through educational newsletters and exclusive offers
Retail Channel Optimization
Balance direct-to-consumer strategies with traditional retail partnerships. Each channel offers unique advantages for reaching target demographics and building brand presence.
Future Outlook: Resveratrol Market Evolution
The Resveratrol Market stands poised for continued expansion driven by demographic trends, scientific advancement, and consumer wellness priorities. Key factors shaping future growth include:
Personalized Nutrition Integration
Genetic testing and biomarker analysis enable customized resveratrol dosing recommendations, creating opportunities for premium personalized supplement services.
Sustainability Focus
Environmental consciousness drives demand for sustainably sourced and packaged resveratrol products, creating differentiation opportunities for eco-conscious brands.
Clinical Research Expansion
Ongoing studies continue revealing new benefits and applications for resveratrol supplementation, expanding market potential and consumer interest.
Frequently Asked Questions About the Resveratrol Market
What factors drive growth in the Resveratrol Market?
The Resveratrol Market experiences robust growth driven by increasing consumer awareness of antioxidant benefits, rising healthcare costs motivating preventive health measures, and scientific validation of resveratrol's multiple health benefits. Additionally, aging populations seeking natural solutions for healthy longevity and growing interest in plant-based supplements contribute significantly to market expansion.
How large is the global Resveratrol Market currently?
The global resveratrol supplement market represents a multi-billion dollar opportunity with consistent double-digit growth rates. North America leads consumption, followed by Europe and rapidly growing Asia-Pacific markets. Market projections indicate potential doubling of current valuations within five years, driven by expanding applications and consumer adoption.
Which consumer segments show highest demand for resveratrol supplements?
Primary demand within the Resveratrol Market comes from health-conscious adults aged 35-65 seeking preventive health solutions, fitness enthusiasts interested in recovery and performance benefits, and individuals focused on healthy aging and longevity. Beauty-conscious consumers also represent a growing segment, attracted to resveratrol's skin health benefits.
What differentiates successful brands in the Resveratrol Market?
Successful resveratrol brands differentiate through superior bioavailability formulations, transparent sourcing and quality testing, educational content and consumer support, strategic pricing aligned with value propositions, and authentic brand stories resonating with target demographics. Innovation in delivery systems and synergistic formulations also creates competitive advantages.
How do regulatory requirements impact the Resveratrol Market?
Regulatory compliance shapes market dynamics by establishing quality standards and permissible claims. In the United States, FDA regulations require GMP compliance and substantiated structure-function claims. International markets have varying requirements, creating both barriers and opportunities for brands willing to invest in proper compliance infrastructure.
What role does scientific research play in Resveratrol Market growth?
Scientific research fundamentally drives Resveratrol Market expansion by validating health benefits, discovering new applications, and improving formulation technologies. Published studies in peer-reviewed journals provide marketing substantiation while building consumer confidence. Ongoing research into longevity, metabolic health, and neuroprotection continues expanding market potential.
How can new brands enter the competitive Resveratrol Market successfully?
New entrants can succeed by identifying underserved niches within the market, developing innovative formulations addressing specific consumer needs, building authentic brand stories connecting with target audiences, leveraging digital marketing for cost-effective customer acquisition, and establishing strategic partnerships with influencers and healthcare practitioners. Focus on quality and education builds sustainable competitive advantages.
What future trends will shape the Resveratrol Market?
Future Resveratrol Market evolution includes personalized nutrition integration using genetic and biomarker data, sustainable sourcing and packaging responding to environmental concerns, advanced delivery technologies maximizing bioavailability, combination formulas targeting specific health outcomes, and expansion into functional foods and beverages. These trends create opportunities for innovation and differentiation.
Which geographical regions offer best opportunities in the Resveratrol Market?
While North America remains the largest market, emerging opportunities exist in Asia-Pacific regions experiencing rapid economic growth and health awareness increases. Latin America shows promising growth potential with expanding middle classes. Europe offers opportunities for premium positioning with quality-focused consumers. Each region requires tailored approaches respecting cultural preferences and regulatory environments.
How important is bioavailability in the Resveratrol Market?
Bioavailability represents a critical differentiator in the Resveratrol Market as traditional resveratrol faces absorption challenges. Advanced delivery systems including micronization, liposomal encapsulation, and combination with absorption enhancers command premium prices while delivering superior results. Brands investing in bioavailability technologies gain significant competitive advantages through improved efficacy and consumer satisfaction.
Brain Cancer, Arthritis And Resveratrol
Cancer Treatment Breakthroughs: How Resveratrol Shows Promise Against Brain Tumors and Beyond
Cancer remains one of humanity's greatest health challenges, but groundbreaking research into natural compounds like resveratrol offers new hope for patients battling this disease. Recent scientific discoveries reveal how this powerful antioxidant found in grapes and red wine demonstrates remarkable potential in fighting aggressive forms of cancer, particularly malignant glioblastoma brain tumors. This in-depth guide explores the latest research findings, treatment possibilities, and what these developments mean for cancer patients and their families seeking innovative therapeutic options.
Understanding Glioblastoma: The Most Challenging Brain Cancer
Among all types of cancer, malignant glioblastoma stands out as particularly devastating. This aggressive brain tumor represents the most common primary brain cancer in adults, yet it carries one of the grimmest prognoses in oncology. Patients diagnosed with glioblastoma face significant challenges due to the tumor's unique characteristics that make it exceptionally difficult to treat effectively.
The complexity of treating glioblastoma cancer stems from three primary factors that oncologists battle daily. First, these tumors possess an extraordinary ability to diffuse throughout brain tissue, making complete surgical removal nearly impossible. Second, glioblastoma demonstrates an alarming rate of recurrence, often returning even after aggressive treatment. Third, these cancer cells develop resistance to conventional therapies, limiting the effectiveness of standard chemotherapy and radiation protocols.
Current statistics paint a sobering picture for glioblastoma cancer patients. The median survival rate hovers around 14 months from diagnosis, making it the deadliest form of central nervous system cancer. [Davis, 2016; NCBI Bookshelf] This harsh reality drives researchers worldwide to explore innovative treatment approaches that could extend survival and improve quality of life for patients facing this formidable adversary.
Revolutionary Research: Resveratrol's Anti-Cancer Properties
A groundbreaking study published in the Journal of Agricultural and Food Chemistry (March 2011) has illuminated a potential new weapon in the fight against cancer. Italian researchers conducted extensive experiments examining resveratrol's effects on human glioblastoma cell cultures, yielding results that could transform how we approach brain cancer treatment.
The research team's findings revealed two critical ways resveratrol combats cancer cells. First, the compound successfully halted the growth of tumor cells, essentially freezing their proliferation in laboratory conditions. Second, and perhaps more importantly, resveratrol limited the cancer cells' ability to migrate, addressing one of glioblastoma's most dangerous characteristics: its tendency to spread throughout brain tissue.
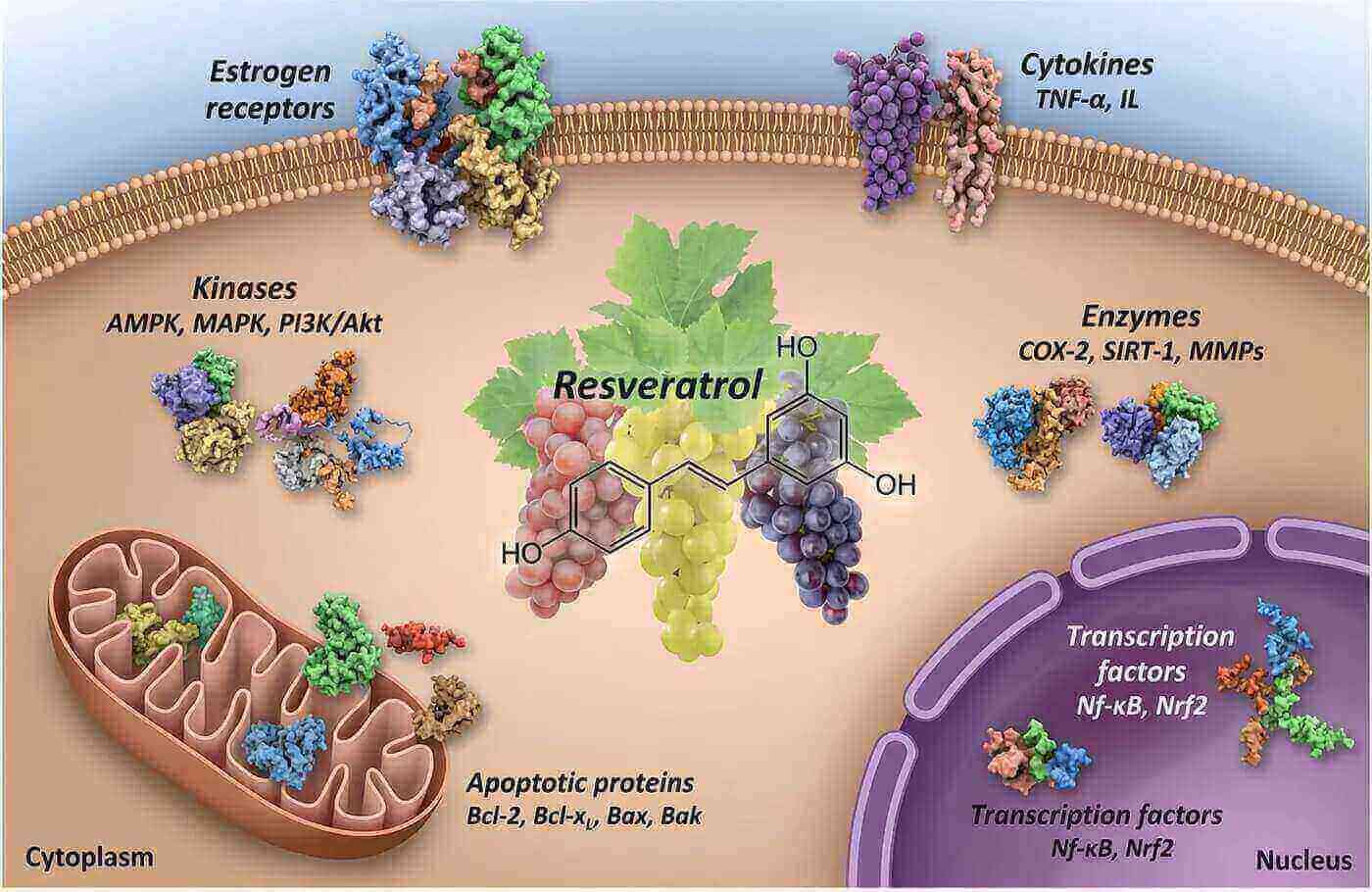
Extended Protection Against Cancer Cell Growth
One of the most encouraging discoveries from this cancer research involved the duration of resveratrol's protective effects. Scientists observed that once glioblastoma cells underwent treatment with resveratrol, the anti-cancer protection persisted for four full days. This extended efficacy suggests that resveratrol doesn't merely provide temporary relief but creates lasting changes in cancer cell behavior.
These observations collectively support the exciting possibility of incorporating resveratrol into standard cancer chemotherapy regimens. By combining this natural compound with existing treatments, oncologists might enhance therapeutic outcomes while potentially reducing the harsh side effects associated with conventional cancer therapies. This integrated approach offers renewed hope for patients battling this deadly form of brain cancer.
How Resveratrol Works Against Cancer: Molecular Mechanisms
Understanding how resveratrol fights cancer requires examining its molecular interactions within tumor cells. This polyphenol compound, naturally occurring in grape skins, peanuts, and berries, possesses unique properties that enable it to interfere with cancer development and progression at multiple stages.
Resveratrol targets cancer through several key mechanisms:
- Cell Cycle Arrest: Resveratrol disrupts the normal division cycle of cancer cells, preventing them from multiplying uncontrollably
- Apoptosis Induction: The compound triggers programmed cell death in cancer cells while leaving healthy cells unharmed
- Angiogenesis Inhibition: By blocking new blood vessel formation, resveratrol starves tumors of nutrients needed for cancer growth
- Metastasis Prevention: The compound reduces cancer cells' ability to invade surrounding tissues and spread to distant organs
- Chemosensitization: Resveratrol enhances cancer cells' sensitivity to conventional chemotherapy drugs
Hope for Cancer Patients: Real-World Implications
Get the same quality used in longevity research.
RevGenetics has supplied premium resveratrol since 2007. Independent lab-verified, wheat-free, no fillers.
View Resveratrol Products →For individuals and families affected by cancer, particularly those facing aggressive forms like glioblastoma, these research findings offer tangible reasons for optimism. While resveratrol cannot replace conventional cancer treatments, its potential as an adjuvant therapy provides an additional tool in the important fight against this disease.
Patients exploring resveratrol for cancer support should consider these encouraging aspects:
- Natural origin with generally favorable safety profile compared to synthetic cancer drugs
- Multiple mechanisms of action addressing various aspects of cancer biology
- Potential to enhance effectiveness of standard cancer treatments
- Additional health benefits beyond direct anti-cancer effects
- Growing body of scientific evidence supporting its use in cancer care
The journey through cancer treatment often feels overwhelming, but advances in understanding compounds like resveratrol provide concrete reasons to maintain hope. Each scientific breakthrough brings us closer to more effective, less toxic cancer therapies that can extend life and improve its quality for patients worldwide.
Frequently Asked Questions About Resveratrol and Cancer
What makes resveratrol effective against cancer cells?
Resveratrol fights cancer through multiple mechanisms including stopping tumor cell growth, preventing cancer cell migration, inducing programmed cell death in malignant cells, and blocking the formation of new blood vessels that feed tumors. Research specifically shows that resveratrol can halt glioblastoma brain cancer cell proliferation and maintain protective effects for up to four days after treatment, making it a promising addition to conventional cancer therapies. [Zhu et al., 2017]
Can resveratrol replace traditional cancer treatments?
No, resveratrol should not replace conventional cancer treatments like chemotherapy, radiation, or surgery. Instead, research suggests it works best as a complementary therapy that may enhance the effectiveness of standard treatments while potentially reducing side effects. Always work with your oncology team to integrate resveratrol safely into your important cancer treatment plan.
What types of cancer show the most promise for resveratrol treatment?
While research continues across many cancer types, glioblastoma brain cancer has shown particularly encouraging results in laboratory studies. The Italian research team's findings demonstrate resveratrol's ability to combat this aggressive brain tumor's growth and spread. Additionally, resveratrol shows promise against various other cancers due to its broad anti-inflammatory and anti-proliferative properties.
How long does it take to see benefits from resveratrol in cancer treatment?
Research indicates that resveratrol begins affecting cancer cells immediately upon treatment, with protective effects lasting at least four days in laboratory studies. However, clinical benefits in human cancer patients may vary depending on factors like cancer type, stage, dosage, and individual response. Consistent supplementation under medical supervision typically provides the best opportunity for therapeutic benefits.
Are there any risks or side effects when using resveratrol for cancer?
Resveratrol generally has a favorable safety profile, but cancer patients should exercise caution. Potential concerns include interactions with chemotherapy drugs, blood thinners, or other medications. Some individuals may experience mild digestive issues at high doses. Always consult your oncologist before adding resveratrol to ensure it won't interfere with your cancer treatment protocol.
What dosage of resveratrol is recommended for cancer support?
Therapeutic dosages for cancer support typically exceed what's obtainable through diet alone. While specific recommendations vary, research studies often use doses ranging from 500mg to several grams daily. Your optimal dosage depends on your specific cancer diagnosis, treatment plan, and individual factors. Work with your healthcare team to determine the most appropriate dosage for your situation.
Can resveratrol help prevent cancer in healthy individuals?
Emerging research suggests resveratrol's anti-inflammatory and antioxidant properties may help reduce cancer risk in healthy individuals. By combating chronic inflammation and oxidative stress, two major contributors to cancer development, regular resveratrol consumption through diet or supplementation might offer protective benefits. However, more long-term human studies are needed to confirm preventive effects.
How does resveratrol compare to other natural cancer-fighting compounds?
Resveratrol stands out among natural compounds for its multi-targeted approach to fighting cancer and its ability to enhance conventional treatments. While other compounds like curcumin, green tea extract, and quercetin also show anti-cancer properties, resveratrol's unique ability to maintain protective effects for days after treatment and its specific efficacy against aggressive cancers like glioblastoma make it particularly promising for therapeutic use.
Rats with oxidative organ damage, and resveratrol
In a research article published online this November, a group of scientist tested the ability of resveratrol to improve cardiovascular function (Journal of Pharmacy and Pharmacology, vol 62 issue 12 pp1784-1793). In this study they took rats and induced oxidative organ damages to the heart, kidneys and brain tissues. They then treated the rats with either resveratrol or saline (as a point of reference). What the group observed is that after a 6-week treatment, those rats treated with resveratrol reversed all the measured damages and improved cardiovascular function.
Groundbreaking Research from Panjab University
A important study from the Department of Biophysics at Panjab University in Chandigarh, India, published in Ultrastruct Pathology (May 2012), has shed new light on the synergistic effects of resveratrol and curcumin. [Bhardwaj et al., 2012] This important research examined how these two natural compounds work together to combat lung carcinoma in animal models.
Study Methodology and Dosage Protocol
The research team administered carefully measured doses to test subjects over a 22-week period. Animals received oral treatments through their drinking water, with curcumin dosed at 60 mg/kg body weight and resveratrol at 5.7 µg/mL, administered three times weekly. This consistent, long-term approach allowed researchers to observe the compounds' cumulative effects on lung cancer progression.
Curcumin: The Golden Partner to Resveratrol
Curcumin brings its own impressive arsenal of health benefits to this powerful combination. This vibrant yellow compound has been used in traditional medicine for thousands of years, and modern science confirms its therapeutic potential. Like resveratrol, curcumin faces bioavailability challenges in its natural form, making advanced formulations important for therapeutic effectiveness.
Revolutionary Curcumin Formulations
Recent technological advances have produced curcumin supplements with absorption rates up to 277 times greater than standard turmeric powder. These enhanced formulations utilize various strategies:
- Piperine Addition: Black pepper extract dramatically increases curcumin absorption
- Phospholipid Complexes: Bind curcumin to fat molecules for improved cellular delivery
- Nanoparticle Technology: Reduces particle size to enhance bioavailability
- Water-Soluble Formulations: Overcome curcumin's natural hydrophobic properties
Frequently Asked Questions About Resveratrol and Curcumin
What is the optimal dosage of resveratrol for health benefits?
Research studies typically use resveratrol doses ranging from 150-500mg daily, though some clinical trials have employed higher amounts. The optimal dose depends on individual factors including age, health status, and specific health goals. Micronized formulations with enhanced bioavailability may require lower doses to achieve similar effects. Always start with the manufacturer's recommended dose and consult with a healthcare provider for personalized guidance.
Can I get enough resveratrol from red wine alone?
While red wine contains resveratrol, the amounts are relatively modest, typically 0.2-2mg per 5-ounce glass. To achieve the doses used in most research studies (150-500mg), you would need to consume impractical and unhealthy amounts of wine. Additionally, alcohol's negative effects would far outweigh any potential benefits. For therapeutic doses, high-quality resveratrol supplements offer a more practical and healthier approach.
How long does it take to see results from resveratrol and curcumin supplementation?
The timeline for experiencing benefits varies depending on individual factors and health goals. Some people report improved energy and well-being within 2-4 weeks, while more significant changes in biomarkers or chronic conditions may take 8-12 weeks or longer. The Panjab University study mentioned used a 22-week protocol, suggesting that patience and consistency are key. Regular use over several months typically yields the best results.
Are there any side effects from taking resveratrol and curcumin together?
Both resveratrol and curcumin are generally well-tolerated natural compounds. Some individuals may experience mild digestive upset, especially when starting supplementation. Taking them with food often minimizes any discomfort. Both compounds have mild blood-thinning properties, so those on anticoagulant medications should consult their healthcare provider. The combination is typically safer than either compound alone at higher doses, as synergy allows for lower individual doses.
What makes micronized resveratrol superior to regular resveratrol?
Micronization reduces resveratrol particles to 1.5 microns or smaller, dramatically improving absorption through the intestinal wall. Regular resveratrol has poor bioavailability due to its crystalline structure and low water solubility. Micronized versions can be 2-5 times more bioavailable, meaning more of the active compound reaches your bloodstream and tissues. This enhanced absorption translates to better results with lower doses.
Should I take resveratrol and curcumin with or without food?
Taking these supplements with food, especially meals containing healthy fats, significantly enhances absorption. Curcumin is fat-soluble and benefits greatly from being consumed with oils or fatty foods. Resveratrol absorption also improves with food, and taking it with meals reduces the likelihood of stomach upset. For optimal results, take your supplements with a meal containing sources of healthy fats like avocado, nuts, or olive oil.
Can resveratrol and curcumin replace conventional cancer treatments?
While research shows promising results for resveratrol and curcumin in cancer prevention and as adjuvant therapies, they should never replace conventional cancer treatments. These natural compounds work best as part of an integrative approach, potentially enhancing the effectiveness of standard treatments while reducing side effects. Always work with oncology professionals who can guide you on safely incorporating these supplements into your treatment plan.
What time of day is best for taking resveratrol supplements?
Research suggests that taking resveratrol in the morning may align best with your body's natural circadian rhythms. Some studies indicate that resveratrol can influence clock genes and metabolism, making morning supplementation potentially more beneficial. However, if you experience any energizing effects that interfere with sleep, morning dosing is definitely preferable. Consistency in timing is more important than the specific time chosen.
How do I know if my resveratrol supplement is high quality?
Quality resveratrol supplements should provide third-party testing certificates verifying purity and potency. Look for products that specify trans-resveratrol content (the active form) rather than just "resveratrol." Micronized formulations with particle sizes under 1.5 microns offer superior absorption. Reputable manufacturers will use dark bottles to protect from light degradation and may include absorption enhancers like piperine or specialized delivery systems.
Can children and elderly individuals safely take resveratrol and curcumin?
Both compounds have good safety profiles, but dosing should be adjusted for age and body weight. Elderly individuals often benefit from these supplements due to their anti-inflammatory and neuroprotective properties. For children, consultation with a pediatrician is essential, as most research has focused on adult populations. When approved, start with much lower doses and monitor closely. The natural food sources of these compounds are generally safe for all ages.
Scientific References:
- Phytotherapy Research: Resveratrol and Cancer Studies
- Restorative Medicine Journal: Combination Therapy Research
- Ultrastructural Pathology: Lung Carcinoma Study
- Drug and Chemical Toxicology: Synergistic Effects Research
The Resveratrol Forum In The New York Academy of Sciences
Resveratrol Research: New York Academy of Sciences Breakthrough Findings on Health Benefits
Resveratrol has captured the attention of the scientific community as one of the most promising natural compounds for human health. The prestigious New York Academy of Sciences, established in 1823 as one of the world's oldest scientific institutions, recently dedicated an entire volume to resveratrol research. This important collection of studies reveals exciting possibilities for those seeking natural solutions to improve their health and longevity.
Why Resveratrol Changes Everything: A Scientific Revolution
The July publication of "Resveratrol and Health" (volume 1290) in the Annals of the New York Academy of Sciences marks a important moment in natural health research. [Brown & Vang, 2013] This dedication of an entire book to resveratrol demonstrates the compound's remarkable potential to transform how we approach wellness, disease prevention, and healthy aging. Scientists worldwide are discovering that this powerful polyphenol, found naturally in red wine, grapes, and certain berries, offers hope for addressing some of humanity's most challenging health concerns.
Top Strategies for Resveratrol Benefits: Three Groundbreaking Studies
1. Resveratrol as a Powerful Cancer Prevention Agent
The first groundbreaking article, "Resveratrol and cellular mechanisms of Cancer prevention," unveils the compound's potential as a natural defense against cancer. Researchers have discovered that resveratrol activates multiple cellular pathways that protect against cancer development and progression. This natural compound works like a sophisticated biological defense system, targeting cancer cells while protecting healthy tissue.
What makes this research particularly exciting is that resveratrol doesn't just work through one mechanism. Instead, it orchestrates a complex symphony of protective actions:
- Cell cycle regulation: Resveratrol helps prevent damaged cells from multiplying uncontrollably
- Apoptosis induction: It encourages cancer cells to self-destruct naturally
- Anti-inflammatory effects: The compound reduces chronic inflammation linked to cancer development
- Antioxidant protection: Resveratrol neutralizes harmful free radicals that damage DNA
Multiple clinical trials are now underway, testing resveratrol's effectiveness against various types of cancer. These studies offer hope to millions seeking natural, preventive approaches to maintain their health and reduce cancer risk.
2. Resveratrol for Heart Health: Understanding the French Paradox
The second article, "Resveratrol: a cardioprotective substance," explores one of nutrition science's most intriguing mysteries. People in Mediterranean regions often consume diets high in saturated fats, yet they experience surprisingly low rates of coronary heart disease (CHD). This phenomenon, known as the "French Paradox," has puzzled researchers for decades.
The answer lies in their regular consumption of red wine, rich in resveratrol. [Das & Maulik, 1999] This natural compound provides remarkable cardiovascular protection through several mechanisms:
- Improving blood vessel function: Resveratrol helps blood vessels relax and expand, improving circulation
- Reducing inflammation: It calms inflammatory processes that contribute to heart disease
- Preventing blood clots: The compound helps maintain healthy blood flow
- Protecting against oxidative stress: Resveratrol shields heart tissue from damage
- Improving cholesterol profiles: It helps balance good and bad cholesterol levels
Laboratory and animal studies consistently demonstrate that resveratrol delivers the heart-protective benefits of red wine without the alcohol. This discovery opens exciting possibilities for those who want cardiovascular protection without consuming wine.
3. Resveratrol for Weight Management and Fat Reduction
The third selected article, "Effects of resveratrol on fat mobilization," reveals another remarkable benefit of this versatile compound. As obesity rates continue to rise globally, finding natural solutions for healthy weight management becomes increasingly important. Resveratrol offers a multi-faceted approach to addressing excess body fat.
Research shows that resveratrol works through three primary mechanisms to support healthy weight:
- Preventing fat cell formation: Resveratrol inhibits the creation of new adipose tissue [Zagotta et al., 2013]
- Reducing fat cell size: It prevents existing fat cells from expanding
- Enhancing fat breakdown: The compound increases the degradation of fat molecules within cells [Li et al., 2024]
These findings suggest that resveratrol could become a valuable component of natural weight management strategies, offering hope to those struggling with weight-related health challenges.
Step-by-Step Guide to Resveratrol Benefits: How It Works in Your Body
Understanding how resveratrol works helps you appreciate its remarkable potential. Here's what happens when you consume this powerful compound:
Step 1: Absorption and Distribution
After consumption, resveratrol rapidly enters your bloodstream through the digestive system. Despite its relatively low bioavailability, even small amounts can produce significant biological effects. The compound travels throughout your body, reaching various organs and tissues where it begins its protective work.
Step 2: Cellular Activation
Once inside your cells, resveratrol activates specific proteins called sirtuins, particularly SIRT1. [Howitz et al., 2003] These proteins are often called "longevity genes" because they regulate cellular health, metabolism, and aging processes. This activation triggers a cascade of beneficial effects throughout your body.
Step 3: Gene Expression Modulation
Resveratrol influences how your genes express themselves, turning on protective pathways while suppressing harmful ones. This epigenetic effect means resveratrol can help your body function more like it did when you were younger, promoting cellular repair and regeneration.
Step 4: Systemic Protection
As resveratrol continues working in your system, it provides widespread protection against oxidative stress, inflammation, and cellular damage. This important defense helps maintain optimal health across multiple body systems.
Optimal Resveratrol Sources and Supplementation
While red wine contains resveratrol, you would need to drink excessive amounts to achieve therapeutic doses. Fortunately, several food sources and supplements provide concentrated amounts of this beneficial compound:
Natural Food Sources
- Red grapes: Especially the skin contains high resveratrol levels
- Blueberries: Offer resveratrol along with other beneficial antioxidants
- Cranberries: Provide modest amounts of resveratrol
- Peanuts: Contain resveratrol, particularly when roasted
- Dark chocolate: Contains small amounts of resveratrol
Supplementation Considerations
For therapeutic benefits, many researchers recommend resveratrol supplements that provide standardized doses. Quality supplements offer consistent potency and purity, ensuring you receive the amounts shown effective in research studies. When selecting a supplement, look for products that use trans-resveratrol, the most bioactive form.
Proven Benefits: Where Resveratrol Shows Greatest Promise
Seasonal Allergy Relief That Actually Works
One of the most exciting discoveries about the effectivity of resveratrol involves its potential for managing hay fever symptoms. Clinical studies demonstrate that using a resveratrol nasal spray three times daily for four weeks can significantly reduce allergy symptoms in adults. [Guo et al., 2018] Even more encouraging, children with seasonal allergies experienced relief when using a combination spray containing beta-glucans and resveratrol for two months. [Ciprandi et al., 2014]
This breakthrough offers hope to millions suffering from seasonal allergies, providing a natural alternative that works with your body's immune system rather than simply masking symptoms. The anti-inflammatory properties of resveratrol help calm the overactive immune response that causes those frustrating allergy symptoms.
Weight Management Support Through Natural Mechanisms
The effectivity of resveratrol for weight management has shown promising results in multiple studies. When high-quality resveratrol supplements are taken orally, they appear to enhance weight loss efforts in overweight and obese adults. [Mousavi et al., 2019] This natural compound works by:
- Enhancing metabolic function
- Supporting healthy fat oxidation
- Promoting energy production at the cellular level
- Helping regulate appetite signals
- Supporting healthy insulin sensitivity
While resveratrol shows promise for weight management, it's important to note that it works best as part of a balanced approach including healthy eating and regular physical activity.
Understanding Limitations: Where Resveratrol May Not Meet Expectations
Transparency about the effectivity of resveratrol means acknowledging areas where research hasn't shown clear benefits. Understanding these limitations helps you make informed decisions about supplementation.
Heart Disease Prevention: Mixed Results
While people consuming higher amounts of dietary resveratrol show lower heart disease risk, oral supplements haven't consistently demonstrated the same cardiovascular benefits. Studies indicate that resveratrol supplements don't significantly impact cholesterol levels or triglycerides in people at risk for heart disease.
Metabolic Syndrome Management
Despite initial hopes, research shows that oral resveratrol doesn't significantly affect blood pressure, glucose levels, or cholesterol in people with metabolic syndrome. This finding highlights the importance of important lifestyle changes rather than relying solely on supplements.
Non-Alcoholic Fatty Liver Disease (NAFLD)
Current evidence suggests that resveratrol supplementation doesn't improve liver function, reduce scarring, or improve cholesterol levels in NAFLD patients. This underscores the need for continued research and multi-faceted treatment approaches.
Safety Profile: What You Need to Know About Resveratrol Side Effects
Understanding the safety aspects of resveratrol helps ensure you can experience its benefits while minimizing risks. The good news? Resveratrol has an excellent safety profile when used appropriately.
General Safety Guidelines
Most people safely consume resveratrol through dietary sources daily. When it comes to resveratrol supplements, research supports these safety parameters:
- Standard doses (250-1500 mg daily): Safe for up to 6 months
- Higher doses (2000-3000 mg daily): Generally safe for 2-6 months, though stomach discomfort may occur
- Topical application: Safe for skin use up to 30 days
- Nasal spray: Safe for at least 4 weeks of use
Special Populations and Precautions
Certain groups should exercise extra caution when considering resveratrol supplementation:
Pregnancy and Breastfeeding
While dietary resveratrol from food sources is safe, pregnant and breastfeeding women should avoid supplements. The source matters significantly – obtaining resveratrol from wine during pregnancy or breastfeeding is never recommended.
Children
Children can safely consume resveratrol through food sources. Nasal sprays containing resveratrol appear safe for pediatric use up to two months, but oral supplements lack sufficient safety data for children.
Medical Conditions Requiring Caution
- Bleeding disorders: Resveratrol may affect blood clotting
- Hormone-sensitive conditions: Due to potential estrogen-like effects
- Scheduled surgery: Discontinue use 2 weeks before procedures
The Science Behind Resveratrol: Understanding Mechanisms of Action
The remarkable effectivity of resveratrol stems from its unique molecular actions:
Sirtuin Activation
Resveratrol activates sirtuins, particularly SIRT1, which are proteins involved in cellular health and longevity. [Howitz et al., 2003] This activation triggers beneficial changes in gene expression, metabolism, and stress resistance.
Antioxidant Properties
As a potent antioxidant, resveratrol neutralizes harmful free radicals that can damage cells and accelerate aging. This protective effect extends throughout the body, supporting overall health and vitality.
Anti-Inflammatory Actions
By modulating inflammatory pathways, resveratrol helps maintain healthy inflammatory responses, which is important for preventing chronic diseases and supporting optimal wellness.
Frequently Asked Questions About Effectivity of Resveratrol
How long does it take to see results from resveratrol supplementation?
The timeline for experiencing the effectivity of resveratrol varies by application. For seasonal allergies, improvements often appear within 2-4 weeks of consistent use. Weight management benefits typically emerge after 8-12 weeks of supplementation combined with lifestyle changes. Some people report increased energy and well-being within the first few weeks, while other benefits may take several months to manifest fully.
Can I get enough resveratrol from food sources alone?
While dietary sources provide some resveratrol, achieving therapeutic doses through food alone is challenging. For example, you'd need to drink hundreds of glasses of red wine to match the resveratrol in a typical supplement. Foods highest in resveratrol include red grapes, blueberries, cranberries, and peanuts. Most people seeking specific health benefits choose supplements to ensure consistent, therapeutic doses.
What's the difference between resveratrol and trans-resveratrol?
Trans-resveratrol is the biologically active form that provides health benefits, while cis-resveratrol is less stable and less effective. Quality supplements specify trans-resveratrol content, ensuring you receive the form proven effective in research studies. The effectivity of resveratrol depends largely on getting the trans form, which is why checking supplement labels is important.
Are there any medications that interact with resveratrol?
Resveratrol may interact with blood thinners, NSAIDs, and medications metabolized by certain liver enzymes. Its mild blood-thinning effects could enhance anticoagulant medications. Always consult healthcare providers before combining resveratrol with prescription medications, especially if you take multiple medications or have chronic health conditions.
What time of day should I take resveratrol for best results?
Research suggests taking resveratrol in the morning may align better with natural circadian rhythms and cellular repair processes. Some people prefer taking it with breakfast to minimize any potential stomach discomfort. The effectivity of resveratrol doesn't seem significantly affected by timing, so choose a schedule you can maintain consistently.
Can resveratrol help with skin health and aging?
Emerging research shows promising results for resveratrol in supporting skin health. Its antioxidant properties help protect against environmental damage, while its anti-inflammatory effects may reduce signs of aging. Both oral supplements and topical applications show potential benefits, though more research is needed to fully understand optimal protocols for skin health.
Is resveratrol safe for long-term use?
Current research supports the safety of resveratrol for periods up to 6 months at standard doses. While no significant safety concerns have emerged for longer-term use, most studies haven't extended beyond this timeframe. Many people successfully use resveratrol long-term by cycling on and off or maintaining lower maintenance doses after initial therapeutic periods.
How can I tell if resveratrol is working for me?
Tracking the effectivity of resveratrol requires attention to specific markers related to your health goals. For allergies, monitor symptom frequency and severity. For weight management, track not just weight but also energy levels, appetite control, and body composition. Keep a health journal noting changes in sleep quality, mood, and overall vitality. Some benefits occur at the cellular level and may not be immediately noticeable but contribute to long-term health.
What should I look for when choosing a resveratrol supplement?
Quality indicators include: trans-resveratrol content clearly stated, third-party testing certificates, micronized or enhanced absorption formulas, dark or opaque packaging to prevent light degradation, manufacturing in GMP-certified facilities, and clear expiration dates. The effectivity of resveratrol depends significantly on product quality, making these factors important for success.
Can resveratrol replace other health interventions?
While resveratrol offers impressive benefits, it works best as part of a whole-body wellness approach. Think of it as a powerful tool that enhances, rather than replaces, fundamental health practices like nutritious eating, regular exercise, stress management, and adequate sleep. The greatest effectivity of resveratrol emerges when combined with these foundational health habits.
The Science Behind Enhanced Bioavailability
One of the biggest challenges with EGCG, Resveratrol and Curcumin has been their limited bioavailability—the amount that actually reaches target tissues after ingestion. AI is revolutionizing how we solve this critical limitation through innovative delivery systems and molecular modifications.
Traditional curcumin supplements, for instance, have notoriously poor absorption, with very little reaching systemic circulation due to rapid metabolism and elimination. [Anand et al., 2007] AI-designed nanoparticle delivery systems can encapsulate these compounds in protective shells that survive digestive processes and release their cargo directly into cancer cells. These smart delivery systems can increase bioavailability by 2000% or more.
Machine learning algorithms have identified specific food compounds that enhance the absorption of EGCG, Resveratrol and Curcumin. For example, piperine (from black pepper) increases curcumin absorption by up to 2000% in human studies, [Shoba et al., 1998] while quercetin enhances EGCG bioavailability. These synergistic combinations create formulations far more effective than individual compounds alone.
AI has also optimized the molecular structure of these compounds through computer-aided design. By predicting how small structural modifications affect absorption, distribution, and activity, researchers can create enhanced versions that maintain therapeutic benefits while dramatically improving bioavailability. These "next-generation" polyphenols represent a new class of natural therapeutics.
Liposomal delivery systems, designed using AI algorithms, protect EGCG, Resveratrol and Curcumin from degradation while facilitating cellular uptake. These microscopic fat bubbles carry the compounds directly into cells, bypassing many absorption barriers. AI-optimized liposomal formulations show 10-20 times higher tissue concentrations compared to standard supplements.
Resveratrol Product Benefits
Resveratrol Benefits: The Evidence on This Powerful Antioxidant
Discover the remarkable resveratrol benefits that have captured the attention of researchers worldwide. This powerful plant compound, naturally found in red grapes, blueberries, and pomegranates, offers promising health advantages ranging from cardiovascular support to potential cancer prevention. Whether you're seeking to boost your immune system, improve metabolic function, or simply enhance your overall wellness, understanding how resveratrol works can open doors to better health outcomes.
Top Resveratrol Benefits for Your Health
1. Powerful Cardiovascular Protection
One of the most celebrated resveratrol benefits involves heart health. This compound works through multiple pathways to support cardiovascular function:
- Blood vessel protection: Resveratrol helps maintain flexible, healthy arteries by protecting the endothelium (inner lining of blood vessels)
- Cholesterol management: Studies suggest resveratrol may help maintain healthy cholesterol levels, particularly by preventing LDL oxidation
- Blood pressure support: The compound's ability to promote nitric oxide production helps blood vessels relax, supporting healthy blood pressure
- Anti-inflammatory effects: By reducing inflammation markers, resveratrol helps protect against cardiovascular disease development
2. Enhanced Brain Function and Neuroprotection
The brain-protective qualities of resveratrol offer hope for maintaining cognitive function throughout life. Research indicates that resveratrol crosses the blood-brain barrier, where it provides direct neuroprotective effects. These benefits include protecting brain cells from oxidative stress, potentially reducing the risk of neurodegenerative conditions, and supporting memory and learning functions.
Scientists have observed that resveratrol activates specific proteins called sirtuins, which play important roles in brain health and longevity. [Grabowska et al., 2024] This activation may help protect against age-related cognitive decline and support mental clarity well into your golden years.
3. Metabolic Support and Weight Management
Among the exciting resveratrol benefits is its potential to enhance metabolic function. Research shows that resveratrol may help:
- Increase insulin sensitivity, supporting healthy blood sugar levels
- Boost mitochondrial function, enhancing cellular energy production
- Support healthy fat metabolism, potentially aiding weight management efforts
- Activate AMPK, a key enzyme involved in cellular energy balance
These metabolic effects make resveratrol particularly valuable for those looking to optimize their body composition and energy levels naturally.
4. Immune System Enhancement
Your immune system serves as your body's primary defense network, and resveratrol acts as a powerful ally in this protection. The compound modulates immune responses, helping your body maintain the delicate balance between defending against pathogens and avoiding excessive inflammation.
Research demonstrates that resveratrol supports both innate and adaptive immunity, enhancing your body's ability to recognize and respond to threats while maintaining healthy inflammatory responses. This balanced approach makes resveratrol particularly beneficial during seasonal challenges or periods of increased stress.
5. Anti-Aging and Longevity Support
Perhaps the most intriguing aspect of resveratrol benefits relates to its potential anti-aging properties. The compound activates sirtuins, often called "longevity genes," which regulate cellular health and lifespan across various species. While human longevity studies continue, the existing research paints an optimistic picture of resveratrol's role in healthy aging.

How Resveratrol Works in Your Body
The mechanisms behind resveratrol benefits involve sophisticated cellular processes. When you consume resveratrol, it interacts with multiple molecular targets throughout your body. The compound activates SIRT1, a protein that regulates cellular health and metabolism. [Howitz et al., 2003] It also influences the AMPK pathway, often called the body's "metabolic master switch."
Additionally, resveratrol modulates inflammatory pathways by inhibiting NF-κB, a key regulator of inflammation. This multi-targeted approach explains why resveratrol offers such diverse health benefits, from cardiovascular protection to potential cancer prevention.
Resveratrol and Cancer Prevention
The National Institutes of Health has documented compelling evidence regarding resveratrol's potential in cancer prevention. The compound appears to work through multiple mechanisms:
- Apoptosis induction: Encouraging damaged cells to self-destruct before becoming cancerous
- Angiogenesis inhibition: Preventing tumors from developing new blood vessels
- Cell cycle regulation: Helping maintain normal cell division patterns
- Metastasis prevention: Potentially reducing cancer spread
While human clinical trials continue, the laboratory evidence provides reason for optimism about resveratrol's role in important cancer prevention strategies.
Athletic Performance and Recovery
Athletes and fitness enthusiasts are discovering the performance-enhancing resveratrol benefits. A 2023 study published in the journal Nutrients demonstrated that resveratrol significantly reduces muscle inflammation and enhances energy utilization during eccentric contraction exercise. In this model, resveratrol not only mitigated muscle damage but tangibly improved overall exercise performance and endurance [Su et al., 2023]. Sports nutritionists increasingly recommend resveratrol supplementation for:
- Faster recovery from intense training sessions
- Reduced exercise-induced oxidative stress
- Enhanced endurance through improved mitochondrial function
- Better muscle adaptation to training stimuli
- Natural pain relief and inflammation reduction
These effects make resveratrol a valuable addition to any serious athlete's nutrition protocol, supporting both performance and long-term health.
Frequently Asked Questions About Resveratrol Benefits
What are the primary resveratrol benefits for overall health?
The primary resveratrol benefits include powerful antioxidant protection, cardiovascular support, enhanced brain function, improved metabolic health, and potential anti-aging effects. This versatile compound supports multiple body systems simultaneously, making it valuable for whole-body wellness. Regular consumption through diet or supplementation may help protect against chronic diseases, support healthy aging, and enhance overall vitality.
How much resveratrol should I take daily?
Most research studies showing positive health outcomes use resveratrol doses between 150-500mg daily. However, optimal dosage depends on individual factors including age, health status, and specific wellness goals. Starting with a lower dose and gradually increasing allows you to assess your body's response. Always choose high-quality supplements containing trans-resveratrol, the most bioactive form, and consult healthcare providers for personalized recommendations.
Can I get enough resveratrol from food alone?
While foods like red grapes, blueberries, and pomegranates contain resveratrol, achieving therapeutic doses through diet alone can be challenging. For example, you would need to consume large quantities of red wine or grape juice daily to match supplement doses used in studies. A balanced approach combining resveratrol-rich foods with quality supplementation often provides the most practical solution for experiencing significant health benefits.
Are there any side effects of taking resveratrol?
Resveratrol generally demonstrates excellent safety when taken at recommended doses. Some individuals may experience mild digestive discomfort when first starting supplementation, which typically resolves with continued use. Because resveratrol has mild blood-thinning properties, those taking anticoagulant medications should consult healthcare providers. Most people tolerate resveratrol well and experience only positive effects when using quality products at appropriate doses.
How long does it take to see resveratrol benefits?
The timeline for experiencing resveratrol benefits varies depending on the specific health outcome. Some people report increased energy and improved well-being within days to weeks. Cardiovascular and metabolic benefits typically become measurable after 8-12 weeks of consistent use. Long-term benefits like cellular protection and anti-aging effects accumulate over months and years. Consistency remains key to experiencing the full spectrum of resveratrol's health-promoting properties.
Can resveratrol help with weight loss?
Research suggests resveratrol may support healthy weight management through multiple mechanisms. It enhances metabolic function, improves insulin sensitivity, and may help activate fat-burning pathways. Studies show resveratrol can help counteract some negative effects of high-calorie diets. While not a magic weight loss solution, resveratrol works synergistically with healthy diet and exercise habits to optimize body composition and metabolic health.
Is resveratrol safe for long-term use?
Current research indicates resveratrol is safe for long-term use when taken at recommended doses. Many populations consuming resveratrol-rich diets, such as those following Mediterranean eating patterns, demonstrate excellent long-term health outcomes. Clinical studies lasting months to years show no significant adverse effects. As with any supplement, periodic evaluation of your health status and needs ensures optimal benefits while maintaining safety.
What makes resveratrol different from other antioxidants?
Resveratrol stands apart from other antioxidants through its unique ability to activate sirtuins and influence gene expression. Unlike simple antioxidants that only neutralize free radicals, resveratrol modulates multiple cellular pathways including AMPK activation, NF-κB inhibition, and mitochondrial biogenesis. This multi-targeted approach provides benefits beyond basic antioxidant protection, potentially influencing aging processes and cellular metabolism at fundamental levels.
Can children take resveratrol supplements?
While resveratrol naturally occurs in foods children commonly eat like grapes and berries, supplementation in children requires careful consideration. Most research focuses on adult populations, with limited pediatric studies available. Children can safely obtain resveratrol through a balanced diet rich in colorful fruits. For supplementation, consult pediatric healthcare providers who can assess individual needs and provide age-appropriate guidance based on current evidence.
How does resveratrol support brain health specifically?
Resveratrol benefits brain health through multiple neuroprotective mechanisms. It crosses the blood-brain barrier to provide direct antioxidant protection to neurons, reduces neuroinflammation, and promotes healthy blood flow to brain tissue. Research shows resveratrol may help clear beta-amyloid proteins associated with cognitive decline, support the growth of new brain cells, and enhance communication between neurons. These combined effects offer promise for maintaining cognitive function and protecting against age-related brain changes.
Resveratrol’s Sirt1 Makes More Telomerase
Resveratrol and Telomerase: The Revolutionary Connection That Changes Everything About Cellular Aging

Imagine if you could help your cells maintain their youthful vitality longer. Recent scientific discoveries reveal that resveratrol, a powerful compound found in red grapes and certain plants, possesses an extraordinary ability to activate telomerase production through SIRT-1 protein regulation. This breakthrough finding opens new possibilities for those seeking to support healthy aging at the cellular level.
Understanding the DNA Foundation: Why Resveratrol's Impact Matters
To truly appreciate how resveratrol works its magic, we need to understand the fundamental role of DNA in our cells. DNA serves as the master blueprint for all living organisms, directing cellular functions through a precise information transfer system. This genetic material passes instructions to RNA, which then translates these messages into proteins that carry out essential cellular functions.
The exciting part? Scientists have discovered that certain compounds like resveratrol can influence how DNA expresses itself, potentially affecting everything from cellular repair to the aging process itself. This discovery transforms our understanding of how we might support healthy cellular function as we age.
The SIRT-1 Revolution: How Resveratrol Regulates Key Longevity Genes
Looking for high-absorption resveratrol?
RevGenetics Micronized Trans-Resveratrol is formulated for superior bioavailability -- more gets into your bloodstream where it counts.
Check Out Micronized Resveratrol →SIRT-1 stands as one of the most fascinating proteins in longevity research. This remarkable protein promotes numerous health benefits associated with extended lifespan across various organisms. What makes SIRT-1 particularly special is its ability to modify and regulate DNA sequences of important longevity genes.
In 2008, researchers made a groundbreaking observation: human fibroblast cells engineered to produce more SIRT-1 showed delayed onset of aging markers. [Yamashita et al., 2012] This discovery sparked intense interest in understanding exactly how SIRT-1 achieves these impressive results. The answer, as it turns out, involves resveratrol's unique ability to boost SIRT-1 production naturally.
The Science Behind SIRT-1 Activation
When resveratrol enters your cells, it triggers a cascade of beneficial reactions. The compound specifically targets and activates SIRT-1, which then goes to work modifying DNA expression patterns. This modification process helps cells maintain their youthful characteristics longer, potentially supporting:
- Enhanced cellular repair mechanisms
- Improved metabolic efficiency
- Better stress resistance at the cellular level
- Optimized energy production within mitochondria
- Reduced accumulation of cellular damage markers
Breakthrough Research: Three Methods of SIRT-1 Activation Reveal Resveratrol's Power
A important study from Japan, led by researcher Yoshinori Katakura, expanded our understanding of how SIRT-1 activation works. The research team tested three different methods to increase SIRT-1 protein production in human cells:
- Genetic modification: Scientists introduced an extra copy of the SIRT-1 gene
- Resveratrol treatment: Cells received resveratrol supplementation
- Caloric restriction: Researchers starved the cells to mimic fasting conditions
The remarkable finding? Resveratrol alone proved sufficient to replicate the anti-aging benefits observed with the other methods. This discovery holds tremendous promise because taking resveratrol supplements represents a far gentler approach compared to genetic modification or extreme caloric restriction.
The Telomerase Connection: Resveratrol's Ultimate Anti-Aging Secret
Perhaps the most exciting revelation from this research involves telomerase, an enzyme critical for maintaining telomeres, the protective caps on our chromosomes. Think of telomeres as the plastic tips on shoelaces that prevent fraying. Each time a cell divides, telomeres shorten slightly. When they become too short, cells can no longer divide and enter a state called senescence.
How Resveratrol Boosts Telomerase Production
The Japanese research team made a groundbreaking discovery: increased SIRT-1 protein (induced by resveratrol) directly leads to more telomerase production. [Yamashita et al., 2012] Here's the fascinating process:
- Resveratrol enters cells and activates SIRT-1 production
- Elevated SIRT-1 levels trigger specific RNA sequences
- These RNA sequences promote telomerase protein synthesis
- Increased telomerase helps maintain telomere length
- Longer telomeres support extended cellular lifespan
This mechanism provides a clear pathway for how resveratrol supplementation might support cellular longevity. Previous research has shown that telomerase activity alone can enable normal human cells to replicate indefinitely under laboratory conditions, highlighting the profound implications of this discovery.
Real-World Applications: Making Resveratrol Work for You
Understanding the science behind resveratrol and telomerase opens exciting possibilities for supporting healthy aging. Many people now incorporate resveratrol supplements into their wellness routines, hoping to harness these cellular benefits. However, not all resveratrol supplements deliver equal results.
Factors That Influence Resveratrol Effectiveness
Bioavailability remains the biggest challenge with resveratrol supplementation. Standard resveratrol has limited absorption in the digestive system, meaning much of what you consume never reaches your cells. This limitation has driven innovation in supplement formulation, leading to advanced delivery systems that dramatically improve absorption rates.
Consider these key factors when evaluating resveratrol supplements:
- Particle size: Smaller particles absorb more readily
- Formulation technology: Advanced delivery systems enhance bioavailability
- Purity levels: Higher purity means more active compound per dose
- Synergistic ingredients: Some compounds enhance resveratrol absorption
- Manufacturing standards: Quality control ensures consistent potency
Advanced Resveratrol Formulations: Maximizing Your Investment in Cellular Health
The evolution of resveratrol supplementation has led to sophisticated formulations designed to overcome bioavailability challenges. Super micronized resveratrol, for instance, uses advanced processing to create ultra-fine particles that your body can absorb up to 10 times more effectively than standard formulations.
This technological advancement means that people taking advanced resveratrol supplements may experience more pronounced benefits from lower doses, making supplementation both more effective and economical. The Nitro250 formulation represents one such advancement, utilizing important micronization technology to maximize absorption and cellular uptake.
Can Resveratrol Help Knockout Obesity?
Resveratrol and Weight Management: Understanding the Science Behind This Powerful Compound
The struggle with weight management affects millions of people worldwide, leading many to search for effective solutions beyond traditional dieting. Resveratrol, a naturally occurring compound found in red wine, grapes, and berries, has emerged as a promising supplement that may support healthy weight management through its unique ability to mimic the benefits of caloric restriction. This in-depth guide explores how resveratrol works, what the latest research reveals, and why this compound might be the breakthrough many have been searching for.

The Science Behind Resveratrol and Caloric Restriction Mimicry
Scientists have long known that reducing caloric intake by 30-50% can lead to remarkable health benefits, including reduced risk of chronic diseases, improved stress resistance, and enhanced cognitive function. However, maintaining such severe dietary restrictions is challenging and often unsustainable for most people.
This is where resveratrol enters the picture as a important. Research has shown that resveratrol activates SIRT1, a protein that becomes highly active during caloric restriction. When SIRT1 is activated, it triggers a cascade of beneficial effects throughout the body:
- Enhanced mitochondrial function - Your cells produce energy more efficiently
- Improved insulin sensitivity - Better blood sugar control and reduced fat storage
- Increased fat oxidation - Your body becomes better at burning fat for fuel
- Reduced inflammation - Lower levels of inflammatory markers associated with obesity
- Optimized gene expression - Activation of genes associated with longevity and metabolic health
Groundbreaking Human Study: Resveratrol's Impact on Obesity
While many supplements show promise in laboratory settings, resveratrol has proven its effectiveness in human clinical trials. A landmark study published in Cell Metabolism examined the effects of resveratrol supplementation on obese individuals over a 30-day period.
The researchers provided participants with trans-resveratrol supplements and monitored various physiological parameters. The results were nothing short of remarkable:
Key Findings from the 30-Day Study:
- Improved Energy Efficiency - Participants showed lower resting energy expenditure, indicating their bodies became more efficient at utilizing energy
- Enhanced Metabolic Profile - Significant improvements in metabolic markers were observed
- Reduced Blood Glucose - Fasting glucose levels decreased, suggesting better blood sugar control
- Lower Insulin Levels - Reduced insulin concentrations indicated improved insulin sensitivity
- Decreased Triglycerides - Blood lipid profiles improved with lower triglyceride levels
- Enhanced Fat Metabolism - Fat tissue showed improved ability to break down stored fat molecules
These results demonstrate that resveratrol can deliver many of the metabolic benefits associated with caloric restriction, offering hope to those struggling with weight management and metabolic health issues.
How Resveratrol Transforms Your Body's Metabolism
Understanding how resveratrol works can help you appreciate its potential for supporting your weight management goals. When you take resveratrol supplements, several transformative processes begin:
1. Activation of Cellular Energy Sensors
Resveratrol activates AMPK (AMP-activated protein kinase), often called the body's "metabolic master switch." This activation signals your cells to become more efficient at producing and using energy, leading to improved fat burning and reduced fat storage.
2. Mitochondrial Biogenesis
Your cells begin producing more mitochondria, the powerhouses that generate cellular energy. More mitochondria mean better energy production and enhanced fat oxidation capacity.
3. Improved Insulin Signaling
Resveratrol helps restore proper insulin function, allowing your body to better regulate blood sugar levels and reduce the tendency to store excess calories as fat.
4. Enhanced Lipid Metabolism
The compound promotes the breakdown of stored fats and reduces the formation of new fat cells, supporting a healthier body composition over time.
5. Gut Microbiome Optimization
Scientists have recently discovered that the gut microbiome plays a pivotal role in how resveratrol fights obesity. A 2025 study revealed that resveratrol supplementation modifies gut bacteria to produce a specific metabolite called 4-hydroxyphenylacetic acid, which directly prevents obesity by activating SIRT1 signaling [Wang et al., 2025]. This means resveratrol not only works on your own cells but also enlists your beneficial gut bacteria to improve insulin sensitivity and lipid metabolism.
Beyond Weight Loss: Additional Benefits of Resveratrol
While weight management might be your primary concern, resveratrol offers a wealth of additional health benefits that make it a valuable addition to any wellness routine:
- Cardiovascular Protection - Supports heart health by improving blood vessel function and reducing oxidative stress
- Anti-Aging Properties - Activates longevity genes and protects against age-related cellular damage
- Cognitive Support - May help maintain brain health and protect against neurodegenerative conditions
- Anti-Inflammatory Effects - Reduces chronic inflammation linked to various health issues
- Antioxidant Power - Neutralizes harmful free radicals that can damage cells and accelerate aging
- Exercise Performance - Some studies suggest improved endurance and exercise capacity
Making an Informed Decision About Resveratrol
If you're considering adding resveratrol to your wellness routine, remember that this powerful compound offers a science-backed approach to supporting healthy weight management and overall metabolic health. The research clearly demonstrates its ability to activate beneficial pathways similar to caloric restriction, providing a practical solution for those seeking metabolic optimization without extreme dietary measures.
By choosing high-quality resveratrol supplements and combining them with healthy lifestyle practices, you're taking a proactive step toward achieving your health and weight management goals. The journey to better health doesn't have to be overwhelming or restrictive. With resveratrol, you have a natural ally that works with your body's inherent wisdom to promote optimal function and vitality.
10 Ways Resveratrol Can Boost Your Longevity
Resveratrol is a powerful compound found in several plants, particularly in grape skins, and it has gained attention for its potential health benefits. From helping with heart health to fighting inflammation, resveratrol can play a important role in promoting a longer life. In this article, we’ll explore the various ways resveratrol can boost your longevity and enhance your overall health. ![]()
1. Understanding Resveratrol and Its Benefits
Resveratrol is a natural polyphenol primarily found in red grapes, berries, and peanuts, known for its numerous health benefits. This compound has been the subject of various studies, revealing its significant impact on cellular health and longevity. By acting as a potent antioxidant, resveratrol helps to combat oxidative stress, an imbalance between free radicals and antioxidants in the body. This oxidative stress is often linked to the aging process and various diseases.
Moreover, resveratrol is credited with supporting heart health. Studies indicate that it can enhance nitric oxide levels, promoting blood vessel dilation and improving circulation. This means it can help lower blood pressure and subsequently reduce the risk of heart disease. Incorporating resveratrol into your diet could serve as an effective strategy for maintaining a healthy heart as you age.
2. How Resveratrol Supports Heart Health
One of the standout features of resveratrol is its ability to support heart health. Research has shown that this compound may help reduce cholesterol levels, specifically lowering LDL or ‘bad’ cholesterol while increasing HDL, the ‘good’ kind. This is a important balance for heart health, as high LDL levels have been linked to increased risk of heart disease.
Furthermore, resveratrol may decrease inflammation, another significant risk factor for cardiovascular issues. By reducing inflammation in the cardiovascular system, resveratrol can help maintain better heart function and improve overall health. This dual action of managing cholesterol and inflammation makes resveratrol an impressive ally in your quest for a long, healthy life.
3. Resveratrol and Its Role in Anti-Inflammation
Inflammation is a critical process within the body, but chronic inflammation can lead to various health issues, including heart disease and diabetes. Resveratrol emerges as a valuable ally in combating this persistent inflammation. Multiple studies suggest that resveratrol may help inhibit the production of inflammatory markers, potentially helping to stave off conditions related to inflammation.
By modulating the body’s inflammatory response, resveratrol can contribute to improved joint health, reduced risk of autoimmune disorders, and even lower chances of developing certain cancers. Emphasizing the importance of including anti-inflammatory foods, resveratrol-rich sources such as berries and red wine could be very beneficial for your long-term health.
4. The Impact of Resveratrol on Brain Health
The connection between resveratrol and brain health has garnered a lot of attention in recent years. Research indicates that this powerful compound may enhance cognitive function and protect against neurodegeneration. One study found that resveratrol could promote the growth of new nerve cells, which is vital for maintaining cognitive health as we age.
In addition to potentially improving memory and learning capabilities, resveratrol may also protect against conditions such as Alzheimer’s disease. By reducing inflammation and oxidative stress in the brain, resveratrol supports overall neural health, providing a proactive approach to preserving cognitive function well into advanced age.
5. Resveratrol and Diabetes Management
Another remarkable aspect of resveratrol is its potential role in diabetes management. Research suggests that resveratrol may improve insulin sensitivity, which is important for maintaining healthy blood sugar levels. By enabling the body to respond better to insulin, this compound can help stabilize blood sugar, making it an excellent addition for those managing diabetes.
Moreover, some studies have indicated that resveratrol might reduce the risk of developing type 2 diabetes. By combating inflammation and oxidative stress, it plays an essential role in overall metabolic health, helping your body maintain balance. This means that adding resveratrol-rich foods to your diet could provide significant benefits alongside a healthy lifestyle.
6. Antioxidant Properties of Resveratrol
Antioxidants are critical for our overall health since they help neutralize free radicals that can damage our cells. Resveratrol is packed with these antioxidant properties, which have been shown to play a vital role in preventing cellular damage and reducing the risks associated with chronic diseases. By incorporating resveratrol into your diet, you arm your body with the necessary tools to fend off oxidative stress.
In addition, the antioxidants in resveratrol may also promote skin health by protecting it from UV radiation damage and improving overall skin tone. Having such protective properties emphasizes the importance of including antioxidants in our diets, as they contribute significantly to long-term health and wellbeing.
7. Resveratrol’s Role in Weight Management
In today’s world, managing weight is an integral part of maintaining overall health. Resveratrol can be a important in this regard. Studies have shown that resveratrol can enhance fat metabolism, potentially making it easier for weight-conscious individuals to manage their body weight. By improving the way fat is processed in the body, resveratrol assists in weight management.
Moreover, resveratrol can also help regulate appetite, making it a useful tool for promoting a healthier eating pattern. This dual effect of enhancing fat metabolism and regulating appetite addresses two significant challenges in weight management. Overall, including resveratrol-rich foods in your daily diet may provide an edge in achieving and maintaining a healthy weight.
8. How Resveratrol Might Extend Cellular Lifespan
One of the most intriguing aspects of resveratrol is its potential ability to extend cellular lifespan. Some studies have indicated that resveratrol can activate the SIRT1 gene, which plays a important role in regulating cellular processes associated with aging. By supporting cellular health and function, resveratrol may enhance the longevity of cells, promoting overall vitality.
Additionally, resveratrol has been linked to the activation of autophagy, a process through which cells remove damaged components and regenerate. By promoting autophagy, resveratrol could help prevent cellular aging and the deterioration often seen in older adults. This mechanism of action reinforces the importance of including resveratrol in your routine if you aim to support cellular health and longevity.
9. Natural Sources of Resveratrol to Include in Your Diet
Incorporating natural sources of resveratrol into your diet is a delicious way to reap its benefits. Some of the best sources include red grapes, blueberries, raspberries, and peanuts. These fruits and nuts not only taste great but also provide essential nutrients alongside resveratrol, making your meals more nourishing.
For those who enjoy a glass of red wine, you’re in luck! Red wine is one of the most studied resveratrol sources, with many studies highlighting its potential health benefits. However, if alcohol is not your preference, resveratrol can also be found in various supplements, making it accessible for all lifestyles. Aim to include a variety of these foods in your diet for a tasty approach to health!
10. Considering Resveratrol Supplements: What You Need to Know
If you’re unable to get enough resveratrol from food sources, supplements may be a viable option. Resveratrol supplements can provide a concentrated dose of this compound, making it easier to experience its benefits. However, it’s essential to choose high-quality supplements from reputable brands, ensuring that you’re not only receiving adequate resveratrol but also avoiding harmful additives.
Before introducing any new supplement to your routine, consulting with a healthcare professional is wise. They can help you assess whether resveratrol supplementation aligns with your health goals. With the right approach, incorporating resveratrol into your routine, whether through diet or supplements, could offer significant advantages that contribute to a longer, healthier life.
Resveratrol Direct Target
Resveratrol: Understanding Its Direct Molecular Target and Revolutionary Health Benefits
Resveratrol stands as one of nature's most fascinating compounds, captivating scientists and health enthusiasts alike with its remarkable ability to support human wellness across multiple body systems. This powerful polyphenol, found naturally in red grapes, berries, and peanuts, has emerged as a cornerstone of longevity research and preventive health strategies.
The Scientific Journey to Discover Resveratrol's Primary Target
For over two decades, researchers have meticulously studied resveratrol and its profound impact on human health. The compound's versatility in addressing age-related conditions, from metabolic disorders to cardiovascular challenges, has sparked intense scientific curiosity about its precise mechanism of action.
The quest to identify how resveratrol works at the molecular level represents one of the most exciting developments in nutritional biochemistry. Understanding this mechanism opens doors to enhanced therapeutic applications and optimized supplementation strategies that could transform how we approach healthy aging.
Breaking Through: The Phosphodiesterase Discovery
A groundbreaking international collaboration has finally unveiled resveratrol's direct molecular target. Researchers from prestigious institutions including the National Institute of Health, University of California Davis, University of North Carolina, University of Texas Southwestern, Emerald BioStructure, Sun Yat-sen University, and University Medical Center Netherlands joined forces to solve this biological puzzle.
Their revolutionary findings, published in the prestigious journal Cell, reveal that resveratrol directly binds to and inhibits enzymes called phosphodiesterases (PDEs). [Park et al., 2012] This discovery represents a paradigm shift in our understanding of how this natural compound delivers its wide-ranging health benefits.
The Cellular Cascade: How Resveratrol Activates Longevity Pathways
When resveratrol blocks PDE activity, it triggers a sophisticated cellular response:
- PDE Inhibition: Resveratrol binds directly to phosphodiesterase enzymes, preventing their normal function
- Epac1 Activation: This inhibition activates another important enzyme called Epac1
- AMPK Stimulation: Epac1 then triggers AMPK, often called the body's "metabolic master switch"
- Sirtuin 1 Activation: Finally, this cascade activates sirtuin 1, the longevity protein long associated with resveratrol's benefits
This elegant molecular pathway explains how a single compound can influence so many aspects of cellular health and longevity.
Optimizing Resveratrol Supplementation
Understanding how resveratrol works allows for more strategic supplementation approaches:
Bioavailability Considerations
While resveratrol offers remarkable benefits, its natural bioavailability can be limited. Modern supplement formulations often include complementary compounds or use specialized delivery systems to enhance absorption and cellular uptake.
Dosage Guidelines
Research suggests that effective resveratrol doses typically range from 150mg to 500mg daily, though individual needs may vary based on health goals and existing conditions. Starting with lower doses and gradually increasing allows the body to adapt optimally.
Timing and Combination Strategies
Taking resveratrol with food can improve absorption, particularly when consumed with healthy fats. Some research suggests combining it with other polyphenols or nutrients may enhance its beneficial effects through synergistic interactions.
Practical Implementation for Optimal Health
Incorporating resveratrol into a whole-body wellness strategy can yield significant benefits:
- Daily Supplementation: Consistent intake ensures steady cellular support
- Dietary Sources: Include resveratrol-rich foods like red grapes, blueberries, and dark chocolate
- Lifestyle Synergy: Combine with exercise and healthy eating for amplified effects
- Medical Consultation: Discuss with healthcare providers, especially if taking medications
Scientific Validation and Ongoing Studies
The confirmation of resveratrol's mechanism through multiple research approaches strengthens confidence in its therapeutic potential. Researchers used chemical PDE inhibitors to replicate resveratrol's effects, providing independent validation of the phosphodiesterase pathway.
Current studies continue to explore how this mechanism translates into specific health benefits across different tissues and organ systems. This ongoing research promises to reveal even more applications for resveratrol in promoting human health and longevity.
Resveratrol Supplements, Are They Beneficial?
The Science Behind Resveratrol's Power
Research reveals that resveratrol works through multiple pathways to support your body's natural defense systems. As a powerful antioxidant, it neutralizes harmful free radicals that can damage cells and accelerate aging. Beyond this protective role, resveratrol activates special proteins called sirtuins, often referred to as "longevity genes."
These sirtuins play important roles in cellular health, DNA repair, and metabolic regulation. When activated by resveratrol, they help your body maintain youthful function at the cellular level, potentially slowing the aging process and supporting overall vitality.
Key Benefits of Resveratrol Supplements
- Heart Health Protection: Studies show resveratrol supports healthy blood flow and maintains optimal cholesterol levels
- Cellular Renewal: Promotes healthy cell turnover and protects against oxidative stress
- Brain Function Support: May help maintain cognitive clarity and memory as you age
- Metabolic Enhancement: Research suggests potential benefits for healthy weight management
- Skin Health: Supports elasticity and smoothness for a more youthful appearance
- Inflammatory Response: Helps maintain healthy inflammatory markers throughout the body
Resveratrol Research: What Studies Reveal
The scientific community continues to uncover exciting possibilities for resveratrol supplementation. Laboratory studies have demonstrated its ability to inhibit cancer cell growth in controlled settings, though human trials are ongoing. Research consistently shows that resveratrol influences multiple biological pathways associated with longevity and disease prevention.
Independent university studies have explored resveratrol's impact on various health conditions, with promising results across multiple areas. These findings suggest that regular supplementation may offer protective benefits that extend far beyond what we initially understood.
Emerging Areas of Resveratrol Research
| Research Area | Key Findings | Potential Benefits |
|---|---|---|
| Cardiovascular Health | Improved endothelial function | Better blood vessel health and circulation |
| Metabolic Function | Enhanced insulin sensitivity | Improved blood sugar regulation |
| Neuroprotection | Reduced oxidative stress in brain cells | Potential cognitive preservation |
| Cellular Aging | Sirtuin activation | Slower cellular aging processes |
How to Take Resveratrol for Maximum Benefits
Optimizing your resveratrol supplementation involves more than just taking a daily capsule. Understanding the best practices for consumption helps you maximize the benefits of this powerful compound.
Recommended Dosage Guidelines
- Starting Dose: Begin with 250-500mg daily to assess tolerance
- Maintenance Dose: Most studies use 500-1000mg for ongoing benefits
- Timing: Take with meals containing healthy fats for enhanced absorption
- Consistency: Daily supplementation provides the most reliable results
The Future of Resveratrol: Exciting Possibilities Ahead
As research continues to unveil new applications for resveratrol, its potential impact on public health becomes increasingly apparent. Scientists explore its role in addressing modern health challenges, including obesity and age-related decline. The compound's ability to influence multiple biological pathways simultaneously makes it particularly promising for holistic health support.
The growing body of evidence suggests that resveratrol supplementation may become a cornerstone of preventive health strategies. As more people discover its benefits, resveratrol is poised to join the ranks of essential daily supplements that support long-term wellness and vitality.
Maximizing Resveratrol's Benefits Through Lifestyle
While resveratrol supplements offer powerful support, combining them with healthy lifestyle choices amplifies their effectiveness. Regular exercise, balanced nutrition, adequate sleep, and stress management create an optimal environment for resveratrol to work its magic.
Consider incorporating resveratrol-rich foods into your diet alongside supplementation. Red grapes, blueberries, cranberries, and peanuts provide additional phytonutrients that work synergistically with supplemental resveratrol.
Resveratrol discussion on Infections and Virus
Understanding Resveratrol and Its Vital Role in Modern Health
As millions of Americans search for effective ways to strengthen their immune systems and promote healthy aging, resveratrol emerges as a beacon of hope. This naturally occurring compound has demonstrated remarkable capabilities in laboratory studies, particularly in its interaction with viruses that affect the majority of the population.
The importance of resveratrol becomes even more evident when we consider that approximately 90-95% of adults in the United States carry the Epstein-Barr virus (EBV) in their bodies. [CDC, 2024] This widespread viral presence makes the discovery of natural compounds that can help manage viral activity particularly valuable for public health.
How Resveratrol Works: The Science Behind Viral Protection
Ready to try resveratrol yourself?
RevGenetics carries pharmaceutical-grade micronized trans-resveratrol -- 98%+ purity, third-party tested.
Shop Resveratrol Supplements →Recent groundbreaking research published in the journal Molecules (2010, vol 15 pp 7115-7124) has illuminated the fascinating mechanisms through which resveratrol exerts its protective effects against viral replication. Scientists have discovered that this remarkable compound works through multiple pathways to support your body's natural defenses.
Key Mechanisms of Resveratrol Action:
- Protein Synthesis Inhibition: Resveratrol effectively blocks the production of proteins that viruses need to replicate within host cells
- Viral Spread Prevention: The compound interferes with proteins required for viruses to spread from infected cells to healthy neighboring cells
- Cellular Defense Enhancement: Resveratrol supports the body's natural immune response mechanisms
- Antioxidant Protection: Provides powerful antioxidant effects that protect cells from oxidative stress
The Epstein-Barr Virus Connection: Why Resveratrol Matters More Than Ever
The relationship between resveratrol and EBV represents a significant breakthrough in natural health science. EBV isn't just another common virus; it's associated with several serious health conditions that affect millions of people worldwide. Understanding how resveratrol can help manage this virus opens new possibilities for preventive health strategies.
Health Conditions Associated with EBV:
| Condition | Connection to EBV | Potential Role of Resveratrol |
|---|---|---|
| Burkitt's Lymphoma | EBV found in tumor cells | May help inhibit viral replication |
| Nasopharyngeal Carcinoma | Strong association with EBV | Supports cellular defense mechanisms |
| Hodgkin's Lymphoma | EBV present in many cases | Potentially reduces viral activity |
| Autoimmune Conditions | Including lupus and multiple sclerosis | May modulate immune response |
Maximizing Resveratrol Benefits: Practical Applications for Daily Life
Incorporating resveratrol into your wellness routine can be both simple and transformative. While this compound occurs naturally in certain foods, many people choose to supplement their intake to ensure they receive therapeutic amounts that research suggests may be beneficial.
Natural Food Sources of Resveratrol:
- Red Grapes and Red Wine: The skin of red grapes contains the highest concentrations
- Blueberries and Cranberries: These antioxidant-rich berries provide modest amounts
- Peanuts and Pistachios: Surprising sources of this beneficial compound
- Dark Chocolate: Contains small amounts along with other beneficial polyphenols
- Japanese Knotweed: One of the richest natural sources, often used in supplements
Understanding Bioavailability: Getting the Most from Resveratrol
One important aspect of resveratrol supplementation involves understanding bioavailability, how well your body can absorb and utilize this compound. Research indicates that certain formulations and delivery methods can significantly enhance absorption, making the selection of high-quality sources particularly important.
Advanced delivery systems, including micronized formulations and combinations with other synergistic compounds, have shown promise in improving the bioavailability of resveratrol. This means that smaller doses can potentially deliver greater benefits when properly formulated.
Looking Ahead: The Promise of Resveratrol
As we continue to face health challenges in our modern world, natural compounds like resveratrol offer hope and practical solutions. The ability of this remarkable substance to support our bodies' natural defense mechanisms while promoting overall health makes it an invaluable addition to any wellness strategy.
The future holds even more promise as researchers continue to important the full potential of resveratrol. With each new study, we gain deeper insights into how this compound can support human health and wellbeing, opening doors to new possibilities for natural health optimization.
Resveratrol: Your Questions Answered
- What are the most common side effects of resveratrol?
Most people tolerate resveratrol well. The most common side effects are mild digestive issues: stomach discomfort, nausea, or loose stools. These typically happen with lower-quality supplements containing impurities like emodin. Taking resveratrol with food and choosing 98%+ purity products usually prevents these problems. Some people report mild headaches during the first week, which tend to resolve on their own.
- How much resveratrol should I take daily?
Clinical trials showing benefits typically use 150-500mg of trans-resveratrol daily. Start at 150-250mg for general health. For metabolic or cardiovascular targets, 300-500mg works well. The benefits appear to plateau around 500mg for most people, so higher doses don't necessarily give better results. Always take with a fat-containing meal to improve absorption.
- Can resveratrol cause joint pain?
Joint discomfort has been reported at high doses (above 1.5-2g daily), but this is uncommon at standard supplementation levels. If you experience joint aches after starting resveratrol, try reducing your dose. The mechanism isn't well understood, but symptoms typically resolve when the dose is lowered.
- Is resveratrol safe with blood pressure medication?
Resveratrol can slightly lower blood pressure on its own (typically 2-4 mmHg systolic), so it may enhance the effects of blood pressure medications. This isn't necessarily dangerous, but it's worth monitoring. Tell your doctor you're taking resveratrol, especially if you're on multiple cardiovascular medications or have very low baseline blood pressure.
- Does resveratrol interact with blood thinners?
Yes, resveratrol has mild antiplatelet effects and may enhance the action of blood thinners like warfarin or aspirin. If you're on anticoagulant therapy, consult your doctor before starting resveratrol. They may want to monitor your INR more closely. Stop resveratrol at least two weeks before any scheduled surgery.
- What's the difference between resveratrol and trans-resveratrol?
Trans-resveratrol is the biologically active form that provides health benefits. Cis-resveratrol is a different molecular shape with minimal activity. When a label says "resveratrol" without specifying "trans," it might contain both forms, meaning less of the active ingredient. Always look for products that specifically list trans-resveratrol at 98%+ purity.
- How long does it take for resveratrol to work?
Energy improvements and subtle cognitive benefits typically appear within 2-4 weeks. Measurable changes in blood markers (blood sugar, inflammatory markers, blood pressure) usually require 8-12 weeks. Anti-aging effects at the cellular level won't be something you "feel" but build over months and years of consistent use.
- Can you take too much resveratrol?
Yes, though serious toxicity is rare. At doses above 2g daily, about 75% of people experience some GI symptoms. Very high doses (4g+) have been used in clinical trials without major safety issues, but there's no evidence that more is better for general health purposes. Stick to 250-500mg daily for most applications.
- Is resveratrol safe for diabetics?
Research actually suggests resveratrol may benefit diabetics by improving insulin sensitivity and lowering fasting glucose. However, because it affects blood sugar, it could enhance the effects of diabetes medications. Monitor your glucose levels carefully when starting resveratrol, and work with your healthcare team to adjust medications if needed.
- Should I take resveratrol on an empty stomach?
No. Resveratrol is fat-soluble, and absorption improves significantly when taken with food containing fat. Taking it on an empty stomach also increases the chance of GI upset. Have it with breakfast, lunch, or dinner that includes some healthy fats like olive oil, eggs, avocado, or nuts.
- Does resveratrol affect hormones or estrogen levels?
Resveratrol has weak phytoestrogenic activity, meaning it can bind to estrogen receptors. Interestingly, some research suggests it may have anti-estrogenic effects in breast tissue specifically. If you have a history of hormone-sensitive cancers or conditions like endometriosis, discuss resveratrol with your oncologist or specialist before supplementing.
- What's the best form of resveratrol supplement?
Look for: trans-resveratrol specifically (not just "resveratrol"), 98%+ purity, third-party tested with available COAs, and dark packaging to prevent light degradation. Micronized formulations offer better absorption. Some products include piperine (black pepper extract) or quercetin to boost bioavailability. Avoid products that don't disclose purity or testing results.
Frequently Asked Questions About Resveratrol
What is the recommended daily Resveratrol Dosage for beginners?
Beginners should start with 100-250mg of resveratrol daily. This low dose allows your body to adjust while still providing beneficial effects. After 2-4 weeks, you can gradually increase your intake based on how you feel. Many people find their optimal dose between 500-1000mg daily, but starting low ensures the best experience.
Can I take too much resveratrol? What are the risks of high doses?
While resveratrol is generally safe, doses above 2500mg daily may cause mild digestive discomfort in some people. High doses can potentially interact with certain medications, particularly blood thinners. Always consult your doctor before taking high doses, especially above 1500mg daily. Most people achieve excellent results with moderate doses between 500-1500mg.
Should I take my Resveratrol Dosage all at once or split throughout the day?
Splitting your daily resveratrol dose often provides better results. Taking half in the morning and half in the evening maintains steadier blood levels. This approach may enhance absorption and reduce any potential digestive sensitivity. Some people prefer taking their entire dose with breakfast, which also works well. Experiment to find what feels best for your body.
How long before I see results from my Resveratrol Dosage?
Most people notice initial benefits within 2-4 weeks of consistent use. Energy improvements and better sleep often come first. More significant changes in cardiovascular health, skin appearance, and cognitive function typically emerge after 2-3 months. Remember that results vary based on your starting health, dosage, and individual biochemistry. Patience and consistency are key to experiencing resveratrol's full benefits.
Does body weight affect my ideal Resveratrol Dosage?
Body weight can influence optimal dosing, though it's not the only factor. Larger individuals may benefit from slightly higher doses, while smaller people might achieve results with less. However, metabolic rate, overall health, and individual sensitivity matter more than weight alone. Start with standard recommendations and adjust based on your response rather than calculating dose by body weight.
Can I take resveratrol with other supplements?
Resveratrol pairs wonderfully with many supplements. It works synergistically with quercetin, CoQ10, and omega-3 fatty acids. Many people combine it with NAD+ precursors for enhanced cellular energy. However, avoid taking resveratrol with iron supplements as it may reduce iron absorption. Always space supplement timing appropriately and consult your healthcare provider about your complete supplement routine.
What's the best form of resveratrol for optimal dosing?
Trans-resveratrol is the most bioactive form and what you should look for in supplements. Micronized versions offer enhanced absorption, potentially allowing lower doses for similar effects. Some advanced formulations include piperine or other absorption enhancers. Pure trans-resveratrol without unnecessary fillers ensures your calculated dose delivers maximum benefit to your cells.
Should I cycle my Resveratrol Dosage or take it continuously?
Most research supports continuous daily use of resveratrol for optimal benefits. Unlike some supplements that require cycling, resveratrol appears safe and effective for long-term daily consumption. Some people choose to take occasional breaks to reassess their needs, but this isn't necessary. Consistent daily dosing maintains steady cellular support and maximizes the compound's protective effects.
How do I know if my Resveratrol Dosage is working?
Signs your resveratrol dose is effective include increased energy, better sleep quality, improved skin appearance, enhanced mental clarity, and faster exercise recovery. Some people notice better cardiovascular markers in blood tests. Keep a journal tracking these improvements. If you don't notice benefits after 8-12 weeks, consider adjusting your dose or trying a different brand for better bioavailability.
Is there a difference between food-sourced and supplemental Resveratrol Dosage?
While foods like red grapes and berries contain resveratrol, the amounts are relatively small. You'd need to consume impractical quantities to match supplement doses. A glass of red wine contains only 1-2mg of resveratrol, while effective supplement doses start at 100mg. Supplements provide concentrated, standardized amounts that deliver consistent therapeutic benefits impossible to achieve through diet alone.
What is the best resveratrol dosage for beginners?
For beginners, starting with 250-500 mg of resveratrol daily provides an excellent introduction to supplementation. This moderate dose allows your body to adjust while delivering noticeable benefits like increased energy and antioxidant protection. After 2-4 weeks, you can assess your response and gradually increase if desired.
Can I take too much resveratrol?
While resveratrol has an excellent safety profile, extremely high doses (above 2500 mg) may cause mild digestive discomfort in some individuals. The key is finding your personal sweet spot through gradual dose escalation. Most people achieve optimal benefits between 500-1500 mg daily without any adverse effects.
How long does it take to see results from resveratrol supplementation?
Many people report increased energy and improved well-being within 2-3 weeks of consistent resveratrol supplementation. However, the full spectrum of benefits, including cardiovascular improvements and enhanced cellular protection, typically develops over 2-3 months of regular use at your optimal dosage.
Can I combine resveratrol with other supplements?
Resveratrol works synergistically with many supplements, particularly quercetin, curcumin, and omega-3 fatty acids. These combinations can enhance the overall benefits while potentially allowing for lower individual doses. Always introduce new supplements gradually and monitor your response.
Is micronized resveratrol worth the extra cost?
Micronized resveratrol offers significantly better absorption than standard forms, meaning you need less to achieve the same benefits. This improved bioavailability often makes it more cost-effective in the long run, as lower doses deliver superior results compared to higher doses of regular resveratrol.
What time of day is best for taking resveratrol?
Morning supplementation aligns with your body's natural rhythms and provides energy support throughout the day. However, some people prefer evening doses for the calming effects. The most important factor is consistency - choose a time that fits your routine and stick with it.
Do I need to cycle resveratrol or can I take it continuously?
Research supports continuous daily supplementation with resveratrol for ongoing benefits. Unlike some supplements that require cycling, resveratrol maintains its effectiveness with consistent use. Many long-term users report cumulative benefits that increase over months and years of regular supplementation.
Should athletes take higher resveratrol doses?
Athletes often benefit from higher resveratrol doses (1000-2000 mg daily) due to increased oxidative stress from training. This elevated dosage supports faster recovery, reduced muscle soreness, and improved endurance. The anti-inflammatory effects of resveratrol make it particularly valuable for those engaged in intense physical activity.
What is the best time of day to take Resveratrol supplements?
The optimal time to take Resveratrol depends on your personal goals and routine. Many people find morning supplementation with breakfast provides sustained energy throughout the day. Taking Resveratrol with a meal containing healthy fats enhances absorption. Some individuals prefer evening doses to support overnight cellular repair processes. The key is consistency - choose a time you can maintain daily for best results.
What makes trans-resveratrol different from regular Resveratrol?
Trans-resveratrol represents the biologically active form that your body recognizes and utilizes effectively. Regular Resveratrol may contain both trans and cis forms, with cis-resveratrol showing minimal biological activity. Quality supplements specify trans-resveratrol content to ensure you receive the form proven effective in research studies. This distinction makes a significant difference in the results you experience from supplementation.
Can Resveratrol help with exercise performance and recovery?
Research demonstrates Resveratrol's ability to enhance exercise capacity and accelerate recovery. By supporting mitochondrial function and reducing exercise-induced oxidative stress, Resveratrol helps muscles work more efficiently and recover faster. Athletes report improved endurance, reduced muscle soreness, and better overall performance. Take Resveratrol 1-2 hours before exercise for performance benefits or immediately after for enhanced recovery.
What should I look for when choosing a Resveratrol supplement?
Select supplements that clearly state trans-resveratrol content and purity levels. Look for third-party testing verification and manufacturing in certified facilities. Advanced formulations may include absorption enhancers or complementary antioxidants. Avoid products with unnecessary fillers or artificial ingredients. Price often reflects quality - invest in reputable brands with proven track records for consistent results and safety.
Can Resveratrol interact with medications or other supplements?
Resveratrol generally shows excellent compatibility with other supplements and most medications. However, because it may enhance blood flow and have mild anti-platelet effects, consult your healthcare provider if you take blood-thinning medications. Resveratrol works synergistically with other antioxidants like quercetin and vitamin C. Always inform your healthcare team about all supplements you take to ensure optimal safety and effectiveness.
Why choose Resveratrol over other anti-aging supplements?
Resveratrol stands apart due to its extensive research validation and multi-system benefits. Unlike single-purpose supplements, Resveratrol supports cardiovascular health, cognitive function, metabolic wellness, and cellular longevity simultaneously. Its ability to activate longevity pathways while providing immediate benefits like enhanced energy makes it a important solution. The thousands of published studies provide confidence that newer compounds simply cannot match.
What is the recommended daily dose of resveratrol?
Clinical research supports a daily dose of 450 mg for optimal benefits. This dosage provides sufficient bioavailability to achieve therapeutic effects while maintaining an excellent safety profile. Some individuals may benefit from starting with a lower dose and gradually increasing to assess their response.
How long does it take to see benefits from resveratrol supplementation?
Many people report increased energy and improved wellbeing within 2-4 weeks of starting resveratrol. However, the most significant benefits, particularly for cardiovascular health and cellular protection, develop over several months of consistent use. Long-term supplementation provides cumulative benefits that continue to compound over time.
Can I get enough resveratrol from food sources alone?
While foods like red grapes, blueberries, and peanuts contain resveratrol, the amounts are relatively small. You would need to consume impractical quantities of these foods daily to achieve therapeutic doses. Supplementation provides a concentrated, consistent source that delivers clinically relevant amounts of resveratrol.
Is resveratrol safe for long-term use?
Extensive research demonstrates that resveratrol is safe for long-term supplementation. Studies lasting several years show no adverse effects at recommended doses. In fact, the benefits of resveratrol tend to increase with continued use, making it an ideal supplement for lifelong health optimization.
How does resveratrol compare to other antioxidants?
Resveratrol stands out among antioxidants due to its unique ability to activate multiple protective pathways simultaneously. While other antioxidants primarily neutralize free radicals, resveratrol also activates longevity genes, supports cellular repair mechanisms, and provides targeted protection for specific organs. This important action makes it exceptionally valuable for holistic health support.
Can resveratrol interact with medications?
While resveratrol is generally safe, it may interact with certain medications, particularly blood thinners and NSAIDs. Its mild blood-thinning properties could enhance the effects of anticoagulant medications. Always consult with healthcare providers before starting resveratrol if you take prescription medications, especially those affecting blood clotting or liver metabolism.













