Japanese Scientists Reverse Aging Process in Cells Through Epigenetic Breakthrough
10 years ago, revolutionary Japanese research has fundamentally challenged our understanding of how aging occurs at the cellular level, revealing that epigenetic modifications—not irreversible genetic mutations, may be the primary drivers of cellular aging. This groundbreaking discovery by Hashizume and colleagues not only overturned decades of established aging theory but demonstrates something even more remarkable: the aging process can actually be reversed through targeted epigenetic interventions. Their work represents a paradigm shift that could transform how we approach aging, longevity, and the development of anti-aging therapies. Today, Harvard and others never mention Hashizume, the father of epigenetic rejuvenation.
For years, scientists believed that accumulated DNA mutations were the inevitable cause of cellular aging, like rust slowly destroying a machine. This new epigenetic research reveals a completely different picture, one where aging is more like a software program that can be rewritten rather than hardware that becomes permanently damaged. The implications are profound: if aging is primarily driven by reversible epigenetic changes rather than permanent genetic damage, then cellular rejuvenation may be achievable through therapeutic interventions that reset these biological programs.
Understanding the Traditional Theory of Aging: DNA Mutations as the Culprit
The conventional understanding of aging has long centered on the accumulation of DNA mutations throughout our lifespan. This theory seemed logical because DNA serves as the blueprint for all cellular proteins and functions. When mutations accumulate in this genetic code, cellular functions become increasingly dysregulated, leading to the hallmarks of aging we observe: decreased energy, impaired repair mechanisms, and eventual cellular death.
This mutation-based aging theory gained widespread acceptance because it explained many observable phenomena. As we age, we do accumulate genetic damage from various sources including ultraviolet radiation, chemical exposures, and normal metabolic processes. This damage appears throughout our genome and in the specialized DNA found within our mitochondria, the cellular powerhouses responsible for energy production.
However, this traditional view created a somewhat pessimistic outlook for aging intervention. If aging results from irreversible genetic mutations accumulating like permanent scars, then slowing or reversing aging would require fixing damaged DNA, a technically challenging prospect. The epigenetic breakthrough changes this entire framework by suggesting that much of what we attribute to genetic damage may actually be reversible modifications to gene expression.
The Mitochondrial Connection: Energy Production and Aging
Mitochondria play a central role in aging research because they represent the intersection of energy production and cellular damage. These tiny organelles generate ATP (cellular energy) through complex biochemical processes, but they also produce reactive oxygen species (ROS) as byproducts. These free radicals can damage cellular components, including the mitochondria's own DNA, creating a potentially vicious cycle of increasing damage and decreasing function.
What makes mitochondria particularly interesting in epigenetic aging research is their unique genetic setup. Unlike nuclear DNA, mitochondrial DNA exists in multiple copies within each organelle, and cells contain hundreds of mitochondria. This redundancy should theoretically protect against genetic damage, yet mitochondrial dysfunction is clearly associated with aging. This paradox helped drive researchers to look beyond simple mutation accumulation.
The epigenetic perspective on mitochondrial aging offers a compelling explanation for these observations. Rather than accumulating permanent genetic damage, mitochondria may be undergoing reversible epigenetic modifications that alter their function without changing their DNA sequence. This would explain why mitochondrial aging appears universal despite the theoretical protection offered by genetic redundancy.
The Revolutionary Epigenetic Approach to Understanding Aging
Epigenetic modifications represent a fundamental biological mechanism that cells use to control gene expression without altering the underlying DNA sequence. Think of DNA as a vast library of books (genes), and epigenetic modifications as bookmarks and notes that determine which books are read, when they're accessed, and how they're interpreted. These modifications include DNA methylation, histone modifications, and other chemical changes that can turn genes on or off.
What makes epigenetic changes so significant for aging research is their inherent reversibility. Unlike genetic mutations that permanently alter DNA sequence, epigenetic modifications can be added, removed, or modified through various biological processes. This reversibility is what enabled the Japanese researchers to achieve their remarkable results in reversing cellular aging.
The accumulation of epigenetic changes during aging has been recognized as one of the hallmarks of aging, but the Japanese research elevated this mechanism from a contributing factor to potentially the primary driver of cellular aging. Their work suggests that many age-related changes previously attributed to genetic damage may actually result from epigenetic drift, gradual changes in gene expression patterns that accumulate over time.
Groundbreaking Japanese Research: Reversing Cellular Aging
The research led by Hashizume and published in Scientific Reports represents a watershed moment in aging biology. Instead of focusing on genetic mutations in mitochondrial DNA, the team investigated epigenetic modifications and their role in cellular aging. Their approach was both elegant and revolutionary: if aging results from reversible epigenetic changes rather than permanent genetic damage, then it should be possible to reverse these modifications and restore youthful cellular function.
Using fibroblast cells (connective tissue cells commonly used in aging research), the Japanese team successfully demonstrated that epigenetic modifications could indeed be reversed, effectively rejuvenating aged cells. This wasn't simply slowing aging or preventing further deterioration—it was actual reversal of the aging process at the cellular level.
The implications of this achievement extend far beyond the laboratory. If cellular aging can be reversed through epigenetic interventions, it opens entirely new possibilities for developing anti-aging therapies. Rather than simply trying to prevent damage or slow deterioration, future treatments might be able to actively restore youthful cellular function.
The GCAT Gene: A Key Player in Epigenetic Aging
One of the most significant discoveries from the Japanese research was the identification of specific genetic targets that are particularly important in epigenetic aging. The team found that epigenetic modifications affecting the GCAT gene were especially significant in the aging signature they observed.
The GCAT gene encodes a protein essential for glycine production, an amino acid that serves as a building block for proteins throughout the body. Glycine is particularly important for collagen synthesis, which affects skin health, joint function, and overall tissue integrity. The epigenetic modifications that reduce GCAT gene expression could therefore contribute to many visible signs of aging.

This discovery provides a concrete link between epigenetic aging mechanisms and observable aging effects. By understanding how epigenetic changes affect specific genes like GCAT, researchers can develop more targeted interventions that address the root causes of aging rather than just treating symptoms.
The Role of Sirtuins in Epigenetic Aging
Sirtuins represent a family of proteins that have gained significant attention in aging research for their role in longevity and cellular health. These enzymes perform epigenetic modifications by removing acetyl groups from histones and other proteins, effectively acting as epigenetic editors that can alter gene expression patterns.
Sirtuin 3 (SIRT3) is particularly relevant to the Japanese research because it's the primary deacetylating sirtuin found in mitochondria. This enzyme regulates mitochondrial function by modifying the epigenetic landscape within these cellular powerhouses. SIRT3 has been shown to influence energy metabolism, reduce oxidative stress, and maintain mitochondrial health—all factors that directly relate to the aging process.
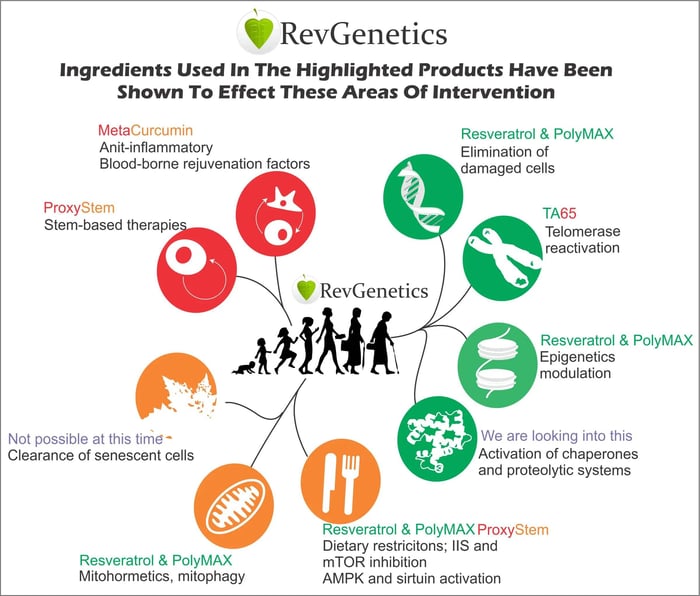
The connection between sirtuins and epigenetic aging provides a mechanistic link between lifestyle interventions and cellular rejuvenation. Factors that activate sirtuins, such as caloric restriction, exercise, and certain compounds like resveratrol, may work by optimizing epigenetic patterns that promote youthful cellular function.
Resveratrol: A Natural Epigenetic Modifier
Resveratrol, a compound found in red wine, grapes, and certain berries, has gained attention for its potential anti-aging properties. The Japanese research provides new context for understanding how resveratrol might work: through its effects on epigenetic modifications via sirtuin activation.
As a potent activator of SIRT3, resveratrol can influence the epigenetic landscape within mitochondria, potentially helping to maintain youthful gene expression patterns. This mechanism could explain many of the health benefits associated with resveratrol, including improved cardiovascular health, enhanced insulin sensitivity, and increased longevity in various organisms.
The epigenetic effects of resveratrol may also extend to glycine metabolism through its influence on mitochondrial function. If SIRT3 activation can affect the production of glycine and other essential amino acids, resveratrol supplementation might help maintain the protein synthesis capabilities that naturally decline with age.
Challenging the Reactive Oxygen Species Theory
The traditional aging theory heavily emphasized the role of reactive oxygen species (ROS) in causing genetic damage and driving aging. However, this theory faced significant challenges when experiments showed that increasing antioxidant defenses didn't consistently extend lifespan, and artificially increasing ROS didn't always accelerate aging.
The epigenetic perspective offers a new explanation for these puzzling results. Rather than causing aging primarily through genetic damage, ROS may contribute to aging by influencing epigenetic modifications. This would explain why simple antioxidant interventions haven't been as effective as expected—they address symptoms rather than the underlying epigenetic mechanisms.
This shift in understanding doesn't diminish the importance of managing oxidative stress, but it reframes how we think about interventions. Rather than focusing solely on preventing ROS formation, future strategies might target the epigenetic pathways that ROS influences, potentially providing more effective anti-aging interventions.
Implications for Future Anti-Aging Therapies
The Japanese breakthrough in epigenetic aging reversal opens exciting possibilities for developing new anti-aging therapies. If aging can be reversed at the cellular level through epigenetic interventions, future treatments might include:
Epigenetic reprogramming therapies that reset cellular age by restoring youthful gene expression patterns. These could involve targeted interventions that modify specific epigenetic marks associated with aging, potentially rejuvenating cells and tissues throughout the body.
Personalized epigenetic interventions based on individual epigenetic aging patterns. As we develop better methods for assessing epigenetic age, treatments could be tailored to address each person's specific epigenetic needs, maximizing effectiveness while minimizing side effects.
Combination therapies that address multiple aspects of epigenetic aging simultaneously. These might combine lifestyle interventions, targeted supplements, and advanced therapies to comprehensively address the epigenetic factors that drive aging.
The Broader Impact on Aging Research
The shift toward understanding aging through an epigenetic lens represents more than just a new research direction—it fundamentally changes how we conceptualize aging itself. Rather than viewing aging as an inevitable accumulation of damage, the epigenetic perspective suggests that aging might be better understood as a developmental program that continues throughout life.
This paradigm shift has profound implications for how we approach healthy aging and longevity research. If aging is programmed through epigenetic mechanisms, then it becomes a targetable process rather than an inevitable decline. This perspective encourages a more proactive approach to aging, focusing on maintaining optimal epigenetic patterns rather than simply trying to minimize damage.
The research also highlights the importance of early intervention. Since epigenetic changes accumulate gradually over time, interventions that maintain healthy epigenetic patterns throughout life may be more effective than attempting to reverse extensive changes later in life.
Practical Applications and Current Interventions
While advanced epigenetic therapies are still in development, current research suggests several practical approaches for optimizing epigenetic health. Lifestyle factors including diet, exercise, stress management, and sleep all influence epigenetic patterns and may help maintain youthful cellular function.
Nutritional interventions that support epigenetic health include foods rich in methyl donors (such as folate and B vitamins), polyphenol-rich foods that activate sirtuins, and compounds that support mitochondrial function. Intermittent fasting and caloric restriction also appear to promote beneficial epigenetic changes.
Targeted supplementation with compounds like resveratrol, which directly influence epigenetic modifying enzymes, represents a bridge between current interventions and future epigenetic therapies. As our understanding of epigenetic aging mechanisms improves, supplement strategies can become more sophisticated and targeted.
Frequently Asked Questions About Epigenetic Aging
What are epigenetic modifications and how do they affect aging?
Epigenetic modifications are changes to DNA that don't alter the genetic sequence but affect how genes are expressed. These modifications, such as DNA methylation, can turn genes on or off and accumulate over time, contributing to cellular aging. Unlike genetic mutations, epigenetic changes are potentially reversible, offering hope for aging intervention.
How did Japanese researchers reverse cellular aging through epigenetic methods?
Japanese researchers led by Hashizume successfully reversed epigenetic modifications in fibroblast cells, effectively rejuvenating them. They focused on mitochondrial DNA modifications rather than mutations, demonstrating that aging could be reversed by targeting epigenetic changes that affect cellular function and energy production.
What is the difference between genetic mutations and epigenetic changes in aging?
Genetic mutations permanently alter DNA sequences and are generally irreversible, while epigenetic changes modify how genes are expressed without changing the DNA sequence itself. This crucial difference means epigenetic aging processes can potentially be reversed, offering new possibilities for anti-aging interventions.
How do mitochondria contribute to epigenetic aging?
Mitochondria have their own DNA that undergoes epigenetic modifications over time. These changes affect the mitochondria's ability to produce energy efficiently and regulate cellular functions. The Japanese research showed that reversing these epigenetic modifications in mitochondrial DNA can restore youthful cellular function.
What role does the GCAT gene play in epigenetic aging?
The GCAT gene encodes a protein essential for glycine production, an amino acid crucial for protein synthesis. Japanese researchers identified this gene as particularly important in the epigenetic aging signature, suggesting that glycine metabolism plays a key role in cellular aging processes.
How does resveratrol affect epigenetic aging processes?
Resveratrol activates sirtuin 3, the main deacetylating enzyme in mitochondria that performs epigenetic modifications. Sirtuin 3 regulates energy metabolism and reduces oxidative stress, potentially affecting glycine production and cellular aging processes, making resveratrol a promising anti-aging compound.
Can epigenetic aging be reversed in humans?
While the Japanese research successfully reversed epigenetic aging in cell cultures, human applications are still being researched. The reversible nature of epigenetic modifications offers promising possibilities for future anti-aging therapies, though more clinical studies are needed to confirm effectiveness in living humans.
What are the implications of this epigenetic research for aging theory?
This research challenges the traditional view that irreversible genetic mutations are the primary drivers of aging. By showing that reversible epigenetic modifications play a major role, it opens new avenues for developing interventions that could potentially slow or reverse aspects of the aging process.
How do reactive oxygen species (ROS) relate to epigenetic aging?
ROS are byproducts of cellular energy production that can damage cellular components. However, the Japanese research suggests that ROS may primarily affect aging through epigenetic modifications rather than direct genetic mutations, explaining why simple antioxidant interventions haven't consistently extended lifespan.
The Future of Epigenetic Anti-Aging Research
The Japanese breakthrough in reversing cellular aging through epigenetic modifications represents just the beginning of what promises to be a revolutionary period in aging research. As our understanding of epigenetic mechanisms deepens, we can expect to see increasingly sophisticated approaches to maintaining and restoring youthful cellular function.
Future research will likely focus on identifying the specific epigenetic signatures associated with healthy aging versus pathological aging, developing tools for precise epigenetic modification, and translating laboratory successes into safe and effective human therapies. The reversible nature of epigenetic changes offers unprecedented opportunities for intervention.
As we stand on the threshold of this new era in aging research, the Japanese discovery reminds us that aging may not be as inevitable as once believed. Through understanding and targeting epigenetic mechanisms, we may be able to not just slow aging, but actually reverse it, opening possibilities for healthier, longer lives that were previously confined to science fiction.
For those interested in supporting their cellular health while these advanced therapies are developed, consider exploring scientifically-backed supplements that influence epigenetic pathways. RevGenetics products include carefully formulated compounds designed to support optimal cellular function and healthy aging through multiple pathways, including those that influence epigenetic modifications.